Nuptial Flights and Mating
The vast majority of virgin queens die within hours after leaving the mother nest. Most are destroyed by predators (Figure 3-4) and hostile workers of alien nests, with the others being variously drowned, overheated, and desiccated. In species with large nest populations, such as the leafcutter ants (Atta) and fire ants (Solenopsis), it is not uncommon for one colony to release hundreds or thousands of the young winged queens in less than an hour. If the surrounding area is dominated by stable, mature colonies, only one or two of the queens might become the progenetrices of new colonies. Most of the rest will die before they can construct a first shelter--or even before they can find a mate. In an unusual study of its kind, Whitcomb et al. (1973) have produced a catalog of the many kinds of predators that decimate young queens of the red imported fire ant Solenopsis invicta. The few individuals that navigate all the dangers must also avoid breeding with males of other species, thus producing inviable or sterile offspring.

It follows that the brief interval between leaving the home nest and settling into a new, incipient nest is a period of intense natural selection among queens, a dangerous odyssey that must be precisely timed and executed in order to succeed. We should expect to find an array of physiological and behavioral mechanisms that enable the young queens simultaneously to avoid enemies, get to the right habitat on time in order to build a secure nest, and mate with males of the same species. Field studies have shown that such specialized traits exist in abundance.
As also expected from the evolutionary argument, mating patterns vary greatly from one species to the next. However, most of the patterns thus far studied fall into one or the other of two broad classes, or "syndromes" (Hölldobler and Bartz, 1985). In the first, the female-calling syndrome, the females, which are often wingless and sometimes just fertile workers, do not travel far from the nest. Standing on the ground or low vegetation, they release sex pheromones to "call" the winged males to them (Figure 3-5). This pattern is displayed by Amblyopone and Rhytidoponera, which are members of the phylogenetically primitive subfamily Ponerinae (Haskins, 1978); presumably also by the very primitive Nothomyrmecia macrops (Hölldobler and Taylor, 1983); at least one pseudomyrmecine, the Neotropical acacia ant Pseudomyrmex ferrugineus (Janzen, 1967); and the socially parasitic species of the myrmicine genera Doronomyrmex, Formicoxenus, Harpagoxenus, and Leptothorax (Buschinger, 1968a,b, 1971a,b, 1975b; see Figure 3-6).
In general, the colonies of female-calling species are typically small at maturity, with 20 to 1,000 workers, and produce relatively few reproductives. So far as known the females mate only once. An unusual variation on this pattern is followed by the Florida harvester ant Pogonomyrmex badius. Females gather on the surface of their home nest and are inseminated by males; afterward they fly off to start new colonies. Van Pelt (1953) thought that the males came from the same nest as the females with whom they copulate, but S. D. Porter (personal communication) observed that they usually fly for about a quarter-hour first before settling on a nest different from their own. Porter observed one case in which a male mated with two females after alighting.
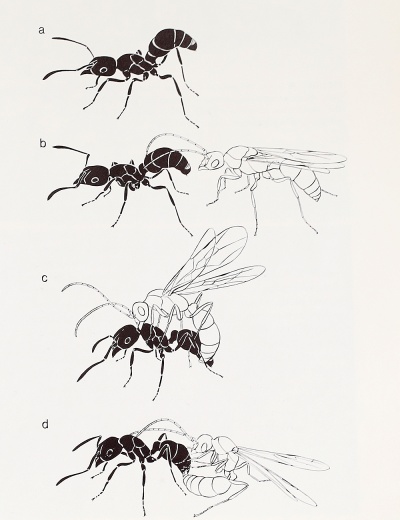
The second combination of traits during mating is the male-aggregation syndrome. Males from many colonies gather at specific mating sites, usually prominent features of the landscape such as sunflecked clearings, forest borders, hilltops, the crowns of trees, and even the tops of tall buildings. Sometimes, as in some species of Lasius and Solenopsis, the males cruise in large numbers at characteristic heights above the ground. The females fly into the swarms, often from long distances, in order to mate (see Figures 3-7 through 3-9 and Plate 2), and afterward they typically disperse widely before shedding their wings and excavating a nest. The winged queens and males of the fire ant Solenopsis invicta, for example, fly up to heights of 250 meters or more; 99 percent then descend to the ground within a 2-kilometer radius of their origin, while a very few travel as far as 10 kilometers. The ability of a single mature colony to disseminate fertile queens in many directions over long distances is one of the reasons the fire ant is so difficult to eradicate (Markin et al., 1971). Male-aggregation species typically differ from those utilizing female calling in two other key respects: the mature colonies are large, containing from several thousand to over a million workers and producing hundreds to thousands of reproductive adults yearly, and multiple insemination is common. An unusual reversal of the usual swarming procedure was recently discovered in some Pheidole species of the southwestern United States: the winged queens gather in aerial swarms, where they maintain a more or less uniform distance from each other while attracting males with pheromones. The males fly into the female swarms and mate with individual females (Hölldobler, unpublished). Swarms of variable composition, some predominantly male and others predominantly female (occasionally exclusively female), have been reported by Eberhard (1978) in the coccid-tending formicine Acropyga paramaribensis of northern South America.

Ant species can be classified another way into two broad types. When the males alight on the surface of the mating site, either in response to female calling or in swarms to compete directly with one another, they are often typically large and robust in form and possess well-developed mandibles. In contrast, males that gather in aerial swarms are usually (but not invariably) smaller relative to the queen than are males of the first type. Also, their mandibles are reduced in size and dentition, sometimes consisting of nothing more than vestigial lobate or strap-shaped organs. An example of this type is the small myrmicine Pheidole sitarches of the southwestern United States. Up to 50 males form circular swarms that hover from a few centimeters to two meters above the surface of woodland clearings. The virgin queens fly in slow, even circles through the aggregations until mounted in midair by a male, whereupon the pair cease flying and spiral to the ground together to complete the copulation (Wilson, 1957b).
The swarms of some ant species are among the more dramatic spectacles of the insect world. W. W. Froggatt (in Wheeler, 1916c) describes the flight of the giant Australian bulldog ant Myrmecia sanguinea as follows:
"On January 30th, after some very hot, stormy weather, while I was at Chevy Chase, near Armidale, N.S.W., I crossed the paddock and climbed to the top of Mt. Roul, an isolated, flat-topped, basaltic hill, which rises about 300 feet above the surrounding open, cleared country. The summit, about half an acre in extent, is covered with low "black-thorn" bushes (Bursaria spinifera). I saw no signs of bull-dog ant nests till I reached the summit. Then I was enveloped in a regular cloud of the great winged ants. They were out in thousands and thousands, resting on the rocks and grass. The air was full of them, but they were chiefly flying in great numbers about the bushes where the males were copulating with the females. As soon as a male (and there were hundreds of males to every female) captured a female on a bush, other males surrounded the couple till there was a struggling mass of ants forming a ball as large as one's fist. Then something seemed to give way, the ball would fall to the ground and the ants would scatter. As many as half a dozen of these balls would keep forming on every little bush and this went on throughout the morning. I was a bit frightened at first but the ants took no notice of me, as the males were all so eager in their endeavors to seize the females."
Donisthorpe (1915) tells of the mass flights of the abundant Myrmica rubra from the distinctively British viewpoint of an earlier observer:
"Farren-White in 1876 observed a swarm of ants near Stonehouse rising and falling over a small beech tree. The effect of those in the air--gyrating and meeting each other in their course, as seen against the deep blue sky--reminded him of the little dodder, with its tiny clustered blossoms and its network of ramifying scarlet threads, over the gorse or heather at Bournemouth. He noticed the swarm about thirty paces off, and it began to assume the appearance of curling smoke; at forty paces he could quite imagine the tree to be on fire. At fifty paces the smoke had nearly vanished into thin air."
A still different mating pattern was described in the Australian formicine species Notoncus ectatommoides by W. L. Brown (1955a):
"In a cropped lawn at Montville, numerous small holes appeared, each opened by workers and accompanied by a minute pile of dark earthen particles. From these holes, males began to issue almost immediately in numbers, until within a few minutes there had accumulated on the surface a surprisingly large number of this sex and also a few workers. The males traveled aimlessly over the sward in low, flitting flight from one blade of grass to another, never rising more than a foot or so from the ground. Movement seemed to take place at random in all directions. Suddenly, however, the males of one area all rushed simultaneously to a single focal point, which proved to be a winged female emerging from a small hole. In a few seconds, the female was surrounded by a dense swarm of males in the form of a ball, which at times must have exceeded 2 cm in diameter. This ball moved in a half-tumbling, half-dragging motion over and among the densely packed grass blades, and held together for perhaps 20 seconds, after which the female escaped, flying straight upward. She appeared not to be encumbered by a male, and no males were seen to follow her for more than a foot above the ground; she flew steadily, and soon passed out of sight.
Meanwhile, the lawn had become dotted with similar balls of frenzied males, each surrounding a female in a fashion similar to the first. Obviously, many more males than females were involved in this particular flight. On each occasion, the female left the ball after 20-30 seconds and flew straight upward."
In a similar fashion males and females of Formica obscuripes conduct nuptial swarms on the ground. Talbot (1972) observed them flying to "swarming grounds" near their nests which were maintained throughout the nuptial flight season and perhaps even from year to year. The males fly back and forth above the ground searching for females which "stand on grasses, forbs or bushes," and apparently signal their presence to the males by pheromones.
No encompassing theory exists to explain the extreme variation in the patterns of mating behavior so far observed. However, a close examination of individual species reveals details that clearly contribute to the greater success of the sexual castes. For example, flying queens of the formicine Lasius neoniger stay strictly within open fields, the exclusive habitat of the earthbound colonies. Fewer than one percent make the mistake of venturing into adjacent woodland, a habitat dominated by the otherwise closely similar Lasius alienus. In one experimental study (Wilson and Hunt, 1966), newly inseminated and flightless queens were labeled with radioactive material for easy tracking and displaced to woodland sites. They attempted to crawl out but were unable to do so. In other words the Lasius queens depend on controlled flight patterns to survive.
Like orientation, the timing of the flights is important for successful mating and colony foundation. Flights conducted as part of the female-calling syndrome do not appear to be well synchronized at the level of either the colony or the population of colonies. The search by airborne males for solitary calling females in fact resembles that of many solitary wasps (Buschinger, 1975; Haskins, 1978). In contrast, flights leading to male aggregation are tightly synchronized within the colony as well as among colonies of the same species.
The manner in which this coordination is achieved is typified by Pogonomyrmex harvester ants of the southwestern United States. The process has been described by Hölldobler (1976b). Just prior to take-off, males and females move restlessly in and out of the sandy crater nests or gather in clusters around the entrance, as shown in Figure 3-7. This preflight activity is especially pronounced in Pogonomyrmex maricopa, a morning flyer, the queens and males of which evidently need more time to warm up before taking wing. As the time of departure approaches, the reproductives run back and forth in mounting intensity. Now, in a frenzy, they climb up and down on grass leaves or small bushes around the nest. At this point many more workers pour out of the nest, running excitedly around the nest and attacking any moving object encountered (including the careless myrmecologist). When the first reproductives try to take flight, the workers at first delay many of them by pulling or carrying them back to the nest. However, once the flight is in full progress, workers cease to interfere. Although the timing of the take-off overlaps considerably between the two sexes, the males generally fly from the nest first. Once aloft both sexes appear at first to drift with the wind, but after a few seconds they take a course upwind or across the wind. Soon afterward they arrive at the swarm sites, centered on conspicuous landmarks such as tree crowns and the tops of hills or (in the case of Pogonomyrmex rugosus) merely flat local areas in the desert.
A similar marching order is observed by the carpenter ant Camponotus herculeanus, which nests in the trunks of both living and dead trees in the boreal forests of Eurasia and North America. Males leave before the queens, although the periods broadly overlap. The early departure of the winged forms is inhibited by the workers, who drag or carry many back to the nest entrance (Figure 3-10). However, when the males do succeed in taking flight, they discharge a pheromone from their mandibular gland. The concentration of this substance is highest at the peak of male activity--the gland emission can now be smelled readily by humans--enough to trigger the mass take-off of the females (Figure 3-11). Blum (1981b) reports methyl 6-methylsalicylate and mellein as two of the three components of the secretion. This pleasantly aromatic combination is shared by most other species of Camponotus, but considerable differentiation nevertheless is achieved by the addition of other substances, such as octanoic acid and methyl anthranilate, according to species (see also Lloyd et al., 1984). A similar function may be accomplished by vibrational signals rather than pheromones in Pogonomyrmex harvester ants. Both males and virgin queens stridulate just before and during take-off, running the sharp posterior rim of their postpetiole over the actively moving, striated file on the first gastric tergite (Markl et al., 1977).
Many entomologists, including especially Kannowski (1959a, 1963) and Weber (1972), have observed that each ant species, at least those displaying the male-aggregation syndrome, swarms at a precise time in the 24-hour diel cycle; and the time differs among species. Under controlled laboratory conditions, McCluskey (1958, 1965, 1967, 1974) and McCluskey and Soong (1979) demonstrated in fact that the rhythms of males are generally if not universally circadian and endogenous. Once set in a laboratory regime of 12 hours light alternating with 12 hours dark, the rhythms persist for up to a week in total darkness. They are also quite precise. McCluskey found that males of the harvester ant Messor (= Veromessor) andrei increase in movement during the last hour of darkness, then peak during the first hour of light. Throughout the remainder of the 24-hour cycle they are quiescent, usually stirring themselves only to groom, solicit food from the workers, or walk sluggishly about the nest. Males of the Argentine ant Iridomyrmex humilis, in contrast, are most active at the very end of the light period. Similarly distinctive rhythms, each spanning only one or two hours, have been documented by McCluskey and his co-workers across a wide diversity of species from four subfamilies (Ponerinae, Myrmicinae, Dolichoderinae, and Formicinae), including some that are wholly nocturnal.
Queens of at least two species, Pogonomyrmex californicus and Mesor (= Veromessor) pergandei, also display circadian rhythms, and these are more or less synchronous with those of the males (McCluskey, 1967; McCluskey and Carter, 1969). In the case of P. californicus at least, the rhythm persists even after the female has flown and lost her wings. But it ceases when she is mated.
In summary, the time of day in which flights occur is programmed by a species-specific diel rhythm. But what determines the particular day on which the flights occur? Several studies, including that by Boomsma and Leusink (1981), have shown that weather conditions play a major role in the timing of nuptial flights. One of the commonest triggering stimuli is rain, especially in species that occur in dry habitats such as deserts, grassland, and forest clearings. A typical species in this respect is Lasius neoniger, one of the most abundant ants in abandoned fields and other open environments in eastern North America. This small formicine emerges in immense swarms in the late afternoon in the second half of August or early September. The flights almost always occur within 24 hours after moderate or heavy rainfall on warm, humid days with little wind. For an hour or so the air seems filled with the winged ants. They rise from the ground like snowfall in reverse. After mating, the queens find themselves on moistened soil that is easier to excavate. They are also protected from desiccation due to overheating (Wilson, 1955a). A very similar pattern is followed by the North American leafcutter ant Atta texana, except that the flights occur well before dawn, between 0300 and 0415 hours (Moser, 1967a).
Because there are relatively few "best days" in which the young queens can be successfully launched, species belonging to the same genus are likely to swarm at the same time and location. In one respect this is a favorable result, since an apparent function of mass emergence and swarming in cicadas, termites, and other insects is the reduction of mortality by overloading predators (Wilson, 1975b). But in another respect it can be detrimental. In the tumult of the swarms, with males struggling to copulate with each female encountered, there is a strong likelihood of interspecific hybridization resulting in either sterility or the production of less viable hybrids. Applying the standard argument from natural selection theory, this circumstance favors the evolution of premating isolating mechanisms. The conventional explanation does seem compatible with a great deal of evidence. Species belonging to genera as phylogenetically diverse as Myrmecia, Pheidole, Solenopsis, and Lasius have been observed to conduct their nuptial flights within the major habitats occupied by the colonies, thus automatically avoiding sexual contact with closely related species limited to other major habitats. How widespread and efficient this isolating mechanism is among ants in general has not been determined. But it cannot be the sole device in deserts, savannas, and tropical moist forests, where large numbers of congeneric species nest closely together. To take an extreme case, in many forest localities in the Amazon Basin, thirty or more species of Pheidole can be found within a single plot of a few square kilometers. Another intrinsic isolating mechanism is differentiation in the preferred mating site within the major habitat. Among the sympatric species of Pogonomyrmex of Arizona, Pogonomyrmex desertorum and Pogonomyrmex maricopa congregate on bushes and trees, while Pogonomyrmex barbatus and Pogonomyrmex rugosus gather at different sites on the ground. In addition, males mark the sites with secretions from their mandibular glands, and apparently the females and other males are attracted by volatile pheromones contained in the material (Hölldobler, 1976b). It is possible (but not yet experimentally verified) that the pheromones are species-specific and serve as an additional isolating device.
Many congeneric species are further separated by the timing of their mating flight, either the season of the year or the hour of the day. In Figures 3-12 and 3-13 we have presented two sets of data from army ants that suggest just such a mutually repulsing spread of flight times across the seasons and the daily cycle respectively. The males of army ants, on which the data were based, fly for an unknown distance before entering the columns or bivouacs of alien colonies belonging to the same species. If the receptiveness of the workers is synchronized by the same circadian rhythm, even the hours of flight can serve as an effective barrier to "mistakes" and interspecific hybridization. Such staggering in the diel flight schedule appears to be common among ants. In Michigan, for example, Myrmica emeryana flies between 0600 and 0800 hours, Myrmica americana between 1230 and 1630 hours, and Myrmica fracticornis between 1800 and 1930 hours (Kannowski, 1959a). Similarly, in Arizona Pogonomyrmex maricopa flies between 1000 and 1130 hours, Pogonomyrmex barbatus between 1530 and 1700 hours, and Pogonomyrmex rugosus between 1630 and 1800 hours. As morning flyers, the Pogonomyrmex maricopa queens appear to be at some disadvantage. The heat of midday prevents them from beginning nest excavation for three or four hours, during which time they are subject to higher predation than the other species (Hölldobler, 1976b). Some of the most closely related European species of Leptothorax swarm at different times of the day; others come into contact, and occasionally hybridize (Plateaux, 1978, 1987).
Another potential advantage of synchronous nuptial swarming is the increase in the numbers of colonies participating and hence the degree of outbreeding. The sparse data on allozyme variation in ants collected so far indicates that outbreeding is indeed nearly total (Craig and Crozier, 1979; Pamilo and Varvio-Aho, 1979; Pearson, 1983; Ward, 1983a). Hence mating is either effectively at random, as demonstrated in experimental choice tests with Pogonomyrmex californicus by Mintzer (1982a), or disassortative, that is, directed away from nestmates.
The glandular sources of sex pheromones produced by female ants have been identified only for a few species. The reproductive females of Rhytidoponera metallica call males with a sex attractant from the pygidial gland, an intersegmental structure between the VIth and VIIth abdominal tergites (Hölldobler and Haskins, 1977). Although some of the contents of this gland have been chemically identified (Meinwald et al., 1983), the specific behavior-releasing components have not yet been established experimentally. In several myrmicine species glands associated with the sting apparatus have been pinpointed as the sources of female sex pheromones. Virgin queens release a male attracting pheromone from the poison gland in the myrmicines Xenomyrmex floridanus (Hölldobler, 1971); Harpagoxenus sublaevis (Buschinger, 1972a); Doronomyrmex kutteri and Doronomyrmex pacis (Buschinger, 1975b; see Figure 3-6); and Formicoxenus nitidulus (Buschinger, 1976a,b).
Buschinger (1972b) was also able to demonstrate that males of Doronomyrmex kutteri and D. pacis react to the other species' female sex pheromones, and that hybridization is possible in laboratory experiments. In the field, however, both species, which occur sympatrically, appear to be sexually isolated by different diel rhythms in mating activity. In general, specificity in sexual communication is consistent with phylogenetic relationships among the leptothoracines. The Canadian slavemaker Harpagoxenus canadensis shows the same mating behavior as the European H. sublaevis, and males of both species respond to the other species' female sex pheromones. Very similar sexual behavior and responses to sex pheromones have been described in several other social parasites of the "subgenus Mychothorax" of Leptothorax, whose hosts, like those of H. canadensis and H. sublaevis, also belong to the "subgenus Mychothorax." The same is true of at least some non-parasitic members of the subgenus. In fact, there appears to be no pheromone specificity among the Leptothorax species. In contrast, Protomognathus americanus males do not respond to H. canadensis or H. sublaevis pheromones. This anomaly suggests that P. americanus may be more closely related to its host of the "subgenus Leptothorax" than to the other genus of Harpagoxenus or the "subgenus Mychothorax" (Buschinger, 1975b, 1981; Buschinger and Alloway, 1979).
Poison gland secretions of Pogonomyrmex females also elicit attraction in males (Hölldobler, 1976b). In Monomorium pharaonis, on the other hand, the female sex pheromone is derived from the Dufour's gland and the bursa pouches (Hölldobler and Wüst, 1973).
Male ants are richly endowed with exocrine glands (Hölldobler and Engel-Siegel, 1982), but little is known about their function. One important fact, noted earlier, is that Camponotus herculeanus males discharge mandibular gland contents when departing from the nest that stimulate the virgin reproductive females to launch as well the nuptial flight. A variety of compounds of the mandibular gland secretions of several Camponotus species have been identified (for review see Blum, 1981b), but it is not yet clear which substance or combination of compounds elicits the behavior. Similarly, the males of Lasius neoniger discharge their mandibular gland contents sometime during the nuptial flight (Law et al., 1965), but the precise timing and function remain unknown.
Males of Pogonomyrmex discharge mandibular gland secretions when arriving at the mating sites. The collectively discharged pheromone appears to attract the virgin females to the lek (Hölldobler, 1976b). It is possible that in other species where males have well-developed mandibular glands and distinct blends of compounds, the secretions also function in promoting aggregation and competition. Examples include Lasius' and Acanthomyops (Law et al., 1965), Camponotus (Brand et al., 1973b,c; review by Blum, 1981), Calomyrmex (Brown and Moore, 1979), Myrmecocystus (review by Blum, 1981b), Tetramorium caespitum (Pasteels et al., 1980), and Polyrhachis doddi (Bellas and Hölldobler, 1985).
A hypothesis concerning a possible novel role of male pheromones in sexual selection in army ants has recently been proposed by Franks and Hölldobler (1987). A detailed morphological examination of the reproductives has shown a close resemblance of conspecific males and females. Males are remarkably queen-like. They are large and robust, and their long, cylindrical abdomens are partially filled with an impressive battery of exocrine glands similar in form and location to those of females. Because queens are flightless and never leave their colony, males must fly between colonies and run the gauntlet of the workers before they approach the queen. For this reason, the workers can choose which males will be admitted and which virgin queens will be inseminated by the males. Army ant workers might therefore be involved in a unique form of sexual selection in which they choose both the matriarch and patriarch of new colonies. If this interpretation is correct, males resemble queens not because they are deceitful mimics; instead, under the influence of sexual selection they have come to use the same channels of communication to demonstrate their potential fitness to the workers as those used by queens.
Worker involvement in sexual selection might not be restricted to the army ants. Wheeler (1910a) noted that males of Leptogenys elongata are also accepted into alien colonies to mate with the wingless ergatoid females, and Maschwitz and Mühlenberg (1975) observed that males run along permanent foraging trails of Leptogenys ocellifera, apparently in an attempt to find access to ergatoid females. It may therefore be significant that Hölldobler and Engel-Siegel (1982) discovered very large exocrine sternal glands in Leptogenys males. Some other ponerines have ergatoid queens and therefore are not likely to engage in ordinary nuptial swarms, including species of Diacamma, Dinoponera, Megaponera, and Ophthalmopone. Longhurst and Howse (1979a) observed that males of Megaponera foetens enter the nests of alien colonies, after utilizing recruitment pheromone trails laid by workers to guide them to the nest. No information is available, however, on the exocrine glandular system of Megaponera or for that matter most other ponerine genera. Males of Ophthalmopone berthoudi also enter strange nests after dispersal flights, but so far as known do not follow odor trails--O. berthoudi workers in fact forage in an exclusively solitary manner and hence are less likely to lay recruitment trails of any kind (Peeters and Crewe, 1986a, 1987).
Male ants compete for females in a rigorous fashion, whether they are orienting to calling females in the primitive manner, flying in aerial swarms, or massing on the surface of the ground and vegetation. The competitive nature of mating is vividly illustrated by Pogonomyrmex rugosus (see Plate 2). The males gather in what can properly be called leks of the vertebrate kind. That is, the males occupy the same site year after year, use pheromones to attract other reproductives of the same species, and then compete with one another for access to the females. In the desert near Portal, Arizona, Hölldobler (1976b) was able to locate only one such site in an area of approximately 120,000 m2. The mating arena covered 4800 m2 of completely flat land unmarked by any distinctive physical features. The winged reproductives approached the arena upwind, which may suggest the presence of an olfactory cue. The first individuals to arrive (at around 1630 hours) were males, which alighted and began to race about in a frenzied manner. Soon afterward the first females alighted. They were immediately surrounded by three to ten males, as shown in Figures 3-8 and 3-9. At the height of the activity thousands of such mating clusters carpeted the ground, in densities as high as 50 per square meter. The queens actively terminated mating after several copulations, and stridulated when prevented from leaving by other suitors. This stridulatory vibration evidently served as a "female liberation signal" that communicated the female's non-receptivity to approaching males and induced them to cease pursuit (Markl et al., 1977). The females then climbed onto grass leaves to launch their flights or else flew directly from the ground. Some landed a short distance away, but others traveled at least 100 meters and possibly much farther. Each then shed her wings and began to excavate a nest chamber in the soil (Figure 3-14).
The general activity at the Pogonomyrmex rugosus mating site lasted about two hours, ending completely by 1900 hours as darkness approached. The males then withdrew into shelters around the mating site, such as crevices beneath grass clumps or little cavities in the soil. There they remained clustered overnight and through the following day until 1500-1600 hours, when they resumed activity. As on the previous day, new males flew in to the site to swell the population, and shortly afterwards females began to arrive. This cycle was repeated on three more consecutive days.
The ant leks differ from those of the sage grouse, hammerheaded bats, and Hawaiian Drosophila (see for example, Bradbury, 1985) in one important respect. Ant males are constrained in a way that vertebrates and fruit flies are not: each male ecloses from the pupa into full maturity with all of the sperm that he will ever possess. Dissections of males from phylogenetically divergent genera such as Nothomyrmecia, Camponotus, Lasius, Myrmica, and Pogonomyrmex reveal that the males' testes have degenerated and all of the sperm have migrated to the expanded vas deferens (Hölldobler, 1966, and unpublished data). When the male mates, it discharges most or all of the sperm together with the secretions of the mucus gland; he is thus incapable of additional inseminations (see Figure 3-15). As a result, reproductive success in male ants does not increase with repeated copulations, as it does with other kinds of insects whose males continuously replenish their sperm supply. Furthermore, it does not appear, from the few cases known, that males have enough sperm to inseminate more than one female. In cases where the queen is destined to produce very large numbers of offspring, one male is not even able to supply all of her needs. In Atta sexdens, for example, each newly eclosed male has between 40 and 80 million spermatozoans, while each newly mated female contains between 200 and 310 million spermatozoans in her spermathecae (Kerr, 1962). Fire ant queens (Solenopsis invicta) receive a supply of about 7 million sperm initially, which they gradually parcel out over a period of almost 7 years until the supply is exhausted (Tschinkel and Porter, 1988). Male ants are thus under strong pressure in natural selection to husband their sperm carefully.
One obvious question concerning the ultimate reproductive success of males is whether it is better for a male to invest all of his sperm in a single female or else to copulate with several females. As Hölldobler and Bartz (1985) pointed out, it is important to note that in ants, unlike other nonsocial species, a male's sperm does not all go towards effective reproduction. This is because in order for an ant colony to begin to produce any reproductive forms, it first must produce many workers. In most advanced ant societies workers are rarely reproductive, and because workers are females derived from fertilized eggs, a substantial portion of a male's sperm is used in colony growth and maintenance rather than in direct production of new queens. The consequent trade-off for a male is obvious. If he inseminates only a single female, and if she mates with no other male, then the male is certain to father any reproductives that she eventually produces. However, mortality of colony-founding females is extremely high. Hence a male that inseminates only a single female puts all of his sperm in one fragile basket. If he were to inseminate several females, on the other hand, he increases the chance that his sperm will end up in a successful foundress of a colony. In this case, however, he might decrease the chance that his sperm is used by the queen to make alates. On the other hand, if males do inseminate several females, there may be selection favoring males whose sperm mixes with other males' sperm in the females' spermathecae. Mixing sperm increases the chance that each male will have at least some offspring among the new crop of alate queens. From allozyme variation studies in multiple mating ant species, it does in fact appear that workers in colonies are fathered by several males (Pamilo, 1982b,c; Pearson, 1983; Ward, 1983a).
In species with large mature colonies, whose females must mate with several males in order to acquire sufficient sperm, males seldom attempt to monopolize females (Cole, 1983b). In many other kinds of insects, and other organisms as well, sperm competition is an important selective force, and males are often favored to ensure that no other male copulates with his mate (Parker, 1970). In multiple mating ant species, however, a male that prevents his mate from mating again may well prevent her from acquiring enough sperm to generate a mature colony, that is, one large enough to produce reproductives. Males in these species therefore should be selected to mate with females that are already mated. The active vying for position in waiting lines behind copulating pairs in Pogonomyrmex species indicates at least that males do not discriminate against previously mated females, but it does not prove the optimum multiple mating hypothesis.
To summarize, male ants are faced with two limiting resources: a restricted number of females available for mating, and a finite supply of sperm that suffices for only one or at most several matings. An expected consequence in evolution is the fierce competition of the kind observed in the Pogonomyrmex leks. Davidson (1982) observed that Pogonomyrmex barbatus and Pogonomyrmex desertorum males indiscriminately seize females and attempt to mate, while the females actively resist copulation. As a result, large males are disproportionately successful at gaining access to mates. In addition, large females mate even more disproportionately with large males. And still further, the average size of males produced by an individual colony depends on the total number of reproductives reared in a given season, which in turn is a function of the size and vigor of the colony. In short, the bigger the colony, the more likely its individual males are to succeed in the mating arenas. Why hasn't this selection pressure created ever larger males in evolution? Davidson offers two reasons: larger males mean fewer males per colony, an obvious trade-off in colony fitness, and very large males (as opposed to merely large ones) have been observed to lose some of their advantage to those slightly smaller. The result is the existence of an optimum male size in Pogonomyrmex.
A confounding bit of data reported by Davidson (1982) is that not only do larger females tend to mate with larger males, but smaller females tend to mate with smaller males. If sexual selection is operating such that females choose larger males, why do small females not also choose to mate with larger males? The answer to this question may be that males are selected to be choosy as well. As we have pointed out male ants have only so much sperm at their disposal, and they cannot afford to be profligate. Selection may favor males who compete for larger females because there is a better chance that large females will survive to produce a mature colony. A result of the competition would be that the smaller, less competitive males must settle for the smaller, less desirable females.
The evolution of male biology has been subjected to few rigorous studies, and most questions concerning trends and optimality in its evolution remain unanswered. We are in a somewhat better position with reference to both data and theory on the number of female matings. As documented in Table 3-1, which includes most or all of the information available, some fraction of the queens of fully three-quarters of all species copulate with more than one male. It is also true, as revealed by allozyme marker studies (Pamilo, 1982b,c; Pearson, 1983; Ward, 1983a), that the sperm from different fathers contribute randomly to fertilization. Cole (1983b) established that multiple matings (polyandry) occurs more frequently in species with large colony size. He concluded, as West-Eberhard (1975) and a few other previous writers had suggested earlier on more intuitive grounds, that polyandry was therefore likely to be a response to the need on the part of queens in large colonies for more spermatozoans than one male can provide. In a study of 25 species in 5 subfamilies, Tschinkel (1987a) added stronger evidence from the number of sperm acquired by queens. In comparisons across species, the number of sperm increases very rapidly with the number of ovarioles. It ranged in Tschinkel's sample from a few tens of thousands in Ponera and Hypoponera, which form small, slow-growing colonies, to 400 million in the leafcutter Atta texana, which attains populations of over a million workers at a time. At another level, the number of sperm stored per ovariole (as opposed to per queen) increased from 2,000 for queens with only six ovarioles to about 30,000 for queens with about 200 ovarioles.
Not satisfied with the intuitively simplest explanation, however, Crozier and Page (1985) went on to employ the method of multiple competing hypotheses to test the adaptiveness of polyandry. They constructed no less than eight such explanations (some admittedly very improbable) to account for the trend documented by Cole. The explanation of limited male contribution favored by Cole and earlier authors was downgraded, because "males of species with big females are generally larger than those with small females, so there is no absolute bar to male size (within reason!)." This does not seem to be a very strong counterargument. Aerial swarmers can potentially benefit from smaller size, which confers greater agility during the approach to incoming queens. Also, as we have noted with reference to mating in Pogonomyrmex, there is a trade-off between male size and male numbers, still poorly analyzed, that might contribute to the preferred production by colonies of smaller males.
Hence it is prudent to keep alive the limited-sperm hypothesis of polyandry. Crozier and Page, after discarding most of the other competing explanations, hold on to three (not counting the limited-sperm hypothesis, which we favor) as both inherently plausible and compatible with the correlation between colony size and polyandry. The first is that caste determination might be genetic, and if so polyandry would allow fuller expression of the caste system in each colony. It follows that species with more complex caste differentiation (a trait associated with large colonies) should be more polyandrous than species with simpler caste systems. As Crozier and Page note, there is no evidence for genetic caste determination in ants to the present time, although recent evidence suggests some kind of genetic predisposition toward various forms of labor specialization in honeybees (Calderone and Page, 1988; Frumhoff and Baker, 1988; Robinson and Page, 1988). All of the many substantial studies to date have implicated a single genotype with multiple developmental pathways controlled by nutritive and other environmental factors (see Chapter 8). The second surviving explanation in the Crozier-Page analysis is that polyandry maximizes the production of divergent worker genotypes, quite apart from caste phenotypes, and hence the range of environmental conditions that a colony can tolerate. Broad-niche species, most often those possessing large colonies, should be more polyandrous than species with narrow niches. This broad relationship has not yet been tested empirically. The third favored hypothesis is that multiple matings reduce the chances of disaster due to the production of diploid males. Males of Hymenoptera, it will be recalled, ordinarily come from unfertilized eggs and are determined as males simply by being haploid, that is, having only one set of sex-determining genes. When one or a very few loci are involved in the process, and recessive male-determining alleles exist, it is also possible to get males from fertilized eggs, the so-called diploid males. The queen of an ant colony can ordinarily control male egg production precisely by opening or closing her spermathecal valve "at will," thus determining whether an egg in the vaginal passage is fertilized. But she has no control whatever over the production of diploid male eggs, because the effort to produce females will still result in a fixed percentage of males by Mendelian chance alone. This circumstance does not matter much if the strategy of the colony is to produce males during early stages of colony growth (beyond the very earliest, fragile stage of colony founding), a not uncommon event in species with a small mature colony size. But it can add a substantial energetic burden on species whose strategy is to hold off production of drones until the colony is large. By mixing sperm from multiple males, the variance of such a load is reduced. In other words, more colonies are likely to have some diploid males, but on the average they are less likely to produce large numbers of diploid males.
Reasoning in another mode, Woyciechowski and Lomnicki (1987) proposed that multiple matings prevent workers from producing male offspring. According to their model of kin selection, workers are at an advantage if they produce sons and care for nephews in the presence of a mother queen who mated only once, but they should avoid personal reproduction and care for brothers in the presence of a mother queen who mated several times. The existing data on queen mating patterns and worker reproduction are not adequate to test the hypothesis.
More recently, Sherman et al. (1988) have argued that the role of polyandry is to increase genetic variation within colonies, thereby reducing the likelihood that parasites or pathogens can decimate the worker force by overcoming all of its physiological and behavioral defenses at once. A balanced portfolio of investments in genetic variation, in other words, is more likely to produce the highest long-term probability of survival and successful growth. This argument is logical, but in our opinion does not accord with the remarkable correlations that exist between polyandry, sperm count, and colony size. This latter relation favors the limited-sperm hypothesis but no other.
In any case, the reproductive behavior of ants is a still poorly explored domain with rich possibilities for general evolutionary biology. More studies are needed along all fronts, including the comparative natural history of nuptial flights, the detailed analysis of individual males and females during mating, genetic studies of sex determination, and more sophisticated models of reproductive competition at the individual and colony levels.
Flight Month Data
 Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Sort by Taxon
| Taxon | Subfamily | Month | Notes |
|---|---|---|---|
| Acromyrmex versicolor | Myrmicinae | Jul • Aug | |
| Acropyga sauteri | Formicinae | Mar • Apr • May • Jun | |
| Aenictus lifuiae | Dorylinae | Aug | Taiwan |
| Amblyopone australis | Amblyoponinae | Feb • Mar | |
| Aneuretus simoni | Aneuretinae | Jul • Aug | |
| Anoplolepis gracilipes | Formicinae | Apr | |
| Aphaenogaster fulva | Myrmicinae | Aug • Sep • Oct | |
| Aphaenogaster lamellidens | Myrmicinae | May • Jun | |
| Aphaenogaster occidentalis | Myrmicinae | Jun • Jul • Aug • Sep | |
| Aphaenogaster picea | Myrmicinae | May | |
| Aphaenogaster subterranea | Myrmicinae | Jul • Aug • Sep | |
| Aphaenogaster tennesseensis | Myrmicinae | Jun | |
| Aphaenogaster texana | Myrmicinae | Jun | |
| Atta mexicana | Myrmicinae | Jun • Jul | |
| Atta texana | Myrmicinae | Apr • May • Jun | |
| Azteca instabilis | Dolichoderinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Brachymyrmex depilis | Formicinae | Mar • Apr | |
| Brachymyrmex obscurior | Formicinae | Jun | |
| Brachymyrmex patagonicus | Formicinae | Apr • May • Jun • Jul • Aug • Sep • Oct | |
| Brachyponera chinensis | Ponerinae | May • Jun • Jul | |
| Camponotus aethiops | Formicinae | Jun • Jul • Aug | |
| Camponotus americanus | Formicinae | Apr • May • Jun | |
| Camponotus anthrax | Formicinae | May | |
| Camponotus auriventris | Formicinae | Apr | |
| Camponotus caryae | Formicinae | Apr • May | |
| Camponotus castaneus | Formicinae | Mar • Apr • May • Jun • Jul | |
| Camponotus chromaiodes | Formicinae | Mar • Apr • May • Jun • Jul | |
| Camponotus cingulatus | Formicinae | May • Jun • Nov | |
| Camponotus claviscapus | Formicinae | May • Jun • Jul • Aug • Sep | |
| Camponotus consobrinus | Formicinae | Dec | |
| Camponotus curviscapus | Formicinae | May • Jun • Jul | |
| Camponotus dalmaticus | Formicinae | Apr • May | |
| Camponotus decipiens | Formicinae | May | |
| Camponotus essigi | Formicinae | May | |
| Camponotus fallax | Formicinae | May • Jun | |
| Camponotus floridanus | Formicinae | Apr • May • Jun • Jul • Aug • Sep | |
| Camponotus fragilis | Formicinae | Jul • Aug | |
| Camponotus herculeanus | Formicinae | May • Jun • Jul | |
| Camponotus hyatti | Formicinae | Mar • Apr • May | |
| Camponotus inaequalis | Formicinae | Mar | as ''Camponotus tortuganus'' (March) |
| Camponotus laevigatus | Formicinae | Mar | |
| Camponotus laevissimus | Formicinae | Apr • May | |
| Camponotus lateralis | Formicinae | Apr | |
| Camponotus ligniperda | Formicinae | May • Jun | |
| Camponotus modoc | Formicinae | Apr • May • Jun • Jul | Washington (May) |
| Camponotus mucronatus | Formicinae | Jun • Jul | |
| Camponotus nawai | Formicinae | Aug | Japan |
| Camponotus nearcticus | Formicinae | Apr • May • Jun | |
| Camponotus nicobarensis | Formicinae | Apr | |
| Camponotus novaeboracensis | Formicinae | Apr • May • Jun • Jul | Washington (May) |
| Camponotus novogranadensis | Formicinae | Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Camponotus obscuripes | Formicinae | May • Jun • Jul • Aug | in Japan, May to June in lowlands, and until August in the mountains |
| Camponotus ocreatus | Formicinae | Mar • Apr | |
| Camponotus pennsylvanicus | Formicinae | Apr • May • Jun • Jul | |
| Camponotus piceus | Formicinae | May • Jun • Jul | |
| Camponotus planatus | Formicinae | May • Jun | |
| Camponotus sanctaefidei | Formicinae | May • Jun • Jul • Aug • Sep • Oct • Nov | |
| Camponotus sansabeanus | Formicinae | Mar • Apr • May | |
| Camponotus saxatilis | Formicinae | Jul • Aug | |
| Camponotus semitestaceus | Formicinae | Mar | |
| Camponotus sexguttatus | Formicinae | Apr | |
| Camponotus socius | Formicinae | May | |
| Camponotus subbarbatus | Formicinae | Apr • May • Jun | |
| Camponotus substitutus | Formicinae | Oct • Nov | |
| Camponotus suffusus | Formicinae | Mar | |
| Camponotus texanus | Formicinae | Mar | |
| Camponotus turkestanus | Formicinae | Apr • May • Jun | |
| Camponotus vagus | Formicinae | Apr • May • Jun | |
| Camponotus vicinus | Formicinae | Mar • Apr • May • Jun • Jul | |
| Camponotus yamaokai | Formicinae | May | Japan |
| Camponotus yogi | Formicinae | Sep | |
| Cardiocondyla mauritanica | Myrmicinae | Jun | |
| Carebara diversa | Myrmicinae | Apr | |
| Cataglyphis cursor | Formicinae | Jul • Aug • Sep | |
| Cataglyphis hispanica | Formicinae | Jul • Aug • Sep | |
| Cataglyphis nodus | Formicinae | Jun • Jul | |
| Colobopsis impressa | Formicinae | Apr • May • Jun • Jul | |
| Colobopsis mississippiensis | Formicinae | Jun • Jul | |
| Colobopsis nipponica | Formicinae | Jun • Jul | Japan |
| Colobopsis obliqua | Formicinae | May • Jun | |
| Colobopsis truncata | Formicinae | Jun • Jul • Aug | |
| Crematogaster ashmeadi | Myrmicinae | Aug • Sep | |
| Crematogaster cerasi | Myrmicinae | Aug • Sep • Oct | |
| Crematogaster depilis | Myrmicinae | Jun | |
| Crematogaster lineolata | Myrmicinae | Oct | |
| Crematogaster matsumurai | Myrmicinae | Jul • Aug • Sep | Japan |
| Crematogaster mutans | Myrmicinae | Apr | |
| Crematogaster osakensis | Myrmicinae | Sep | Japan |
| Crematogaster scutellaris | Myrmicinae | Aug • Sep • Oct | |
| Crematogaster stollii | Myrmicinae | Jun • Jul • Aug • Sep • Oct | |
| Cryptopone ochracea | Ponerinae | Aug • Sep | |
| Cryptopone testacea | Ponerinae | Jan • Apr • May • Jun • Aug • Dec | |
| Cyphomyrmex costatus | Myrmicinae | Feb • Mar • Apr • Sep • Dec | |
| Cyphomyrmex minutus | Myrmicinae | May | |
| Diacamma rugosum | Ponerinae | Jan • Feb • Mar • Apr | |
| Dinomyrmex gigas | Formicinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Dolichoderus bispinosus | Dolichoderinae | Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov | |
| Dolichoderus debilis | Dolichoderinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Dolichoderus lutosus | Dolichoderinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Dolichoderus plagiatus | Dolichoderinae | May | |
| Dolichoderus quadripunctatus | Dolichoderinae | Jul • Aug • Sep | |
| Dolichoderus sibiricus | Dolichoderinae | Aug • Sep • Oct | Japan |
| Dolichoderus taschenbergi | Dolichoderinae | Jun | |
| Dolopomyrmex pilatus | Myrmicinae | Mar | |
| Dorymyrmex bicolor | Dolichoderinae | Feb • Mar • Apr | |
| Dorymyrmex bureni | Dolichoderinae | Apr • May • Jun • Jul • Aug | |
| Dorymyrmex insanus | Dolichoderinae | Jan • Feb • Mar • Apr • May • Jul • Aug • Sep • Oct • Nov • Dec | |
| Ectatomma ruidum | Ectatomminae | Jan • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Ectatomma tuberculatum | Ectatomminae | May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Forelius pruinosus | Dolichoderinae | May • Jun • Jul • Aug | |
| Formica altipetens | Formicinae | Jul • Aug | |
| Formica argentea | Formicinae | Jul • Aug | |
| Formica aserva | Formicinae | Jun • Jul | |
| Formica biophilica | Formicinae | Jun • Jul | |
| Formica bruni | Formicinae | Jul • Aug | |
| Formica brunneonitida | Formicinae | Jun • Jul • Aug | |
| Formica caucasicola | Formicinae | Jul • Aug | |
| Formica cinerea | Formicinae | Jun • Jul • Aug | |
| Formica clara | Formicinae | Jun • Jul | |
| Formica exsecta | Formicinae | Jun • Jul • Aug | |
| Formica exsectoides | Formicinae | Jul | |
| Formica foreli | Formicinae | Jul • Aug | |
| Formica forsslundi | Formicinae | Jul | |
| Formica francoeuri | Formicinae | Apr • May • Jun | |
| Formica fusca | Formicinae | Jun • Jul • Aug | |
| Formica fuscocinerea | Formicinae | Jun • Jul • Aug • Sep | |
| Formica gagates | Formicinae | Jul • Aug | |
| Formica gagatoides | Formicinae | Jul • Aug | |
| Formica glacialis | Formicinae | Jul • Aug | |
| Formica glauca | Formicinae | Jun • Jul | |
| Formica incerta | Formicinae | Jul • Aug | |
| Formica integra | Formicinae | May | |
| Formica integroides | Formicinae | Apr | |
| Formica lemani | Formicinae | Jun • Jul • Aug • Sep | |
| Formica longiceps | Formicinae | Jul • Aug | |
| Formica lugubris | Formicinae | May • Jun • Jul | |
| Formica manchu | Formicinae | Jun • Jul • Aug | |
| Formica mesasiatica | Formicinae | Jun • Jul • Aug | |
| Formica moki | Formicinae | Jun • Jul | |
| Formica obscuripes | Formicinae | Apr • May | Washington (May) |
| Formica obscuriventris | Formicinae | Jun | |
| Formica pacifica | Formicinae | Apr | |
| Formica paralugubris | Formicinae | May • Jun • Jul | |
| Formica pisarskii | Formicinae | Aug | |
| Formica polyctena | Formicinae | Apr • May • Jun | |
| Formica pratensis | Formicinae | Apr • May • Jun • Jul • Aug • Sep | |
| Formica pressilabris | Formicinae | Jun • Jul • Aug | |
| Formica ravida | Formicinae | May • Jun • Jul | |
| Formica rubicunda | Formicinae | Jul | |
| Formica selysi | Formicinae | Jun • Jul • Aug • Sep | |
| Formica subaenescens | Formicinae | Aug | |
| Formica subpolita | Formicinae | Jun | |
| Formica subsericea | Formicinae | Apr • May • Jun • Jul • Aug | |
| Formica transkaucasica | Formicinae | Jul • Aug | |
| Formica transmontanis | Formicinae | Oct • Nov | |
| Formica truncorum | Formicinae | Jun • Jul • Aug | |
| Formica ulkei | Formicinae | Jul • Aug | |
| Formica uralensis | Formicinae | Jun • Jul • Aug | |
| Formicoxenus nitidulus | Myrmicinae | Jul • Aug | |
| Gnamptogenys continua | Ectatomminae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Gnamptogenys hartmani | Ectatomminae | Jan • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct | |
| Harpagoxenus sublaevis | Myrmicinae | Jul • Aug | |
| Hypoponera nippona | Ponerinae | Aug | Japan |
| Hypoponera opacior | Ponerinae | Jul | |
| Hypoponera punctatissima | Ponerinae | Jun • Jul • Aug • Sep | |
| Iridomyrmex bicknelli | Dolichoderinae | Apr | Yass, New South Wales |
| Iridomyrmex purpureus | Dolichoderinae | Oct | |
| Lasius alienus | Formicinae | Jul • Aug • Sep | |
| Lasius austriacus | Formicinae | Jul • Aug • Sep | |
| Lasius bicornis | Formicinae | Apr • May • Jun • Jul • Aug • Sep | |
| Lasius brevicornis | Formicinae | Aug • Sep | |
| Lasius brunneus | Formicinae | May • Jun • Jul • Aug | |
| Lasius capitatus | Formicinae | Sep | Japan |
| Lasius carniolicus | Formicinae | May • Jun • Jul • Aug • Sep • Oct | |
| Lasius citrinus | Formicinae | Apr • May • Jun • Jul • Aug | |
| Lasius claviger | Formicinae | Aug • Sep • Oct • Nov • Dec | |
| Lasius distinguendus | Formicinae | Jul • Aug • Sep | |
| Lasius emarginatus | Formicinae | Jun • Jul • Aug | |
| Lasius flavus | Formicinae | Jun • Jul • Aug • Sep | |
| Lasius fuliginosus | Formicinae | Jun • Jul • Aug • Sep | |
| Lasius hayashi | Formicinae | Jul • Aug | Japan |
| Lasius interjectus | Formicinae | Apr • May • Jun | |
| Lasius japonicus | Formicinae | Jul • Aug | Japan |
| Lasius jensi | Formicinae | Jun • Jul • Aug • Sep | |
| Lasius lasioides | Formicinae | May • Jun • Jul | |
| Lasius latipes | Formicinae | Jun • Jul • Aug • Sep | |
| Lasius meridionalis | Formicinae | Jun • Jul • Aug • Sep | |
| Lasius mixtus | Formicinae | Jul • Aug • Sep | |
| Lasius morisitai | Formicinae | Jul | Japan |
| Lasius murphyi | Formicinae | Mar | |
| Lasius myops | Formicinae | May • Jun • Jul • Aug • Sep | |
| Lasius nearcticus | Formicinae | Jul • Aug • Sep | |
| Lasius neoniger | Formicinae | Jul • Aug • Sep • Oct • Nov • Dec | |
| Lasius niger | Formicinae | Jun • Jul • Aug • Sep | |
| Lasius nipponensis | Formicinae | Jun • Jul | Japan |
| Lasius orientalis | Formicinae | Jul | Japan |
| Lasius paralienus | Formicinae | Aug • Sep • Oct | |
| Lasius platythorax | Formicinae | Jun • Jul • Aug | |
| Lasius productus | Formicinae | Aug • Sep | Japan |
| Lasius psammophilus | Formicinae | Jul • Aug | |
| Lasius reginae | Formicinae | Aug • Sep | |
| Lasius sabularum | Formicinae | Aug • Sep • Oct | |
| Lasius sakagamii | Formicinae | Jun • Jul • Aug • Sep • Oct | Japan |
| Lasius sonobei | Formicinae | Aug • Sep | Japan |
| Lasius spathepus | Formicinae | Jun • Jul • Aug | Japan |
| Lasius subumbratus | Formicinae | Jul • Aug | |
| Lasius talpa | Formicinae | Aug • Sep | Japan |
| Lasius umbratus | Formicinae | Jun • Jul • Aug • Sep | |
| Leptogenys elongata | Ponerinae | May • Jun | |
| Leptogenys punctaticeps | Ponerinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Leptothorax acervorum | Myrmicinae | Jun • Jul • Aug • Sep | |
| Leptothorax gredleri | Myrmicinae | Jul • Aug | |
| Leptothorax kutteri | Myrmicinae | Jul • Aug | |
| Leptothorax muscorum | Myrmicinae | Jun • Jul • Aug • Sep | |
| Leptothorax pacis | Myrmicinae | Jul | |
| Linepithema humile | Dolichoderinae | May • Jun • Jul | |
| Liometopum luctuosum | Dolichoderinae | Mar | |
| Liometopum microcephalum | Dolichoderinae | Jun • Jul | |
| Liometopum occidentale | Dolichoderinae | Mar • Apr • May • Jun | |
| Manica rubida | Myrmicinae | May • Jun • Jul • Aug • Sep | |
| Manica yessensis | Myrmicinae | Aug | Japan |
| Mayaponera arhuaca | Ponerinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep | |
| Mayaponera constricta | Ponerinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Dec | |
| Megalomyrmex symmetochus | Myrmicinae | May • Jun • Jul • Aug | |
| Meranoplus bicolor | Myrmicinae | Jan • Feb • Apr • May • Jun • Aug • Sep • Oct • Nov • Dec | |
| Messor aciculatus | Myrmicinae | Apr • May | Japan |
| Messor barbarus | Myrmicinae | Sep • Oct • Nov | |
| Messor capitatus | Myrmicinae | Jan • Nov • Dec | |
| Messor ebeninus | Myrmicinae | Nov • Dec | |
| Messor structor | Myrmicinae | Mar • Apr • May • Jun • Jul • Aug • Sep | |
| Monomorium ergatogyna | Myrmicinae | Jun | |
| Monomorium floricola | Myrmicinae | Jun | |
| Myrmecia brevinoda | Myrmeciinae | Mar | |
| Myrmecia tarsata | Myrmeciinae | Nov | |
| Myrmecina americana | Myrmicinae | Aug • Sep • Oct | |
| Myrmecina graminicola | Myrmicinae | Aug • Sep | |
| Myrmecocystus creightoni | Formicinae | Feb | |
| Myrmecocystus kennedyi | Formicinae | Mar • Apr | |
| Myrmecocystus mendax | Formicinae | Jul | |
| Myrmecocystus mexicanus | Formicinae | Jul • Aug | |
| Myrmecocystus mimicus | Formicinae | Jun • Jul • Aug | |
| Myrmecocystus navajo | Formicinae | Jul • Aug | |
| Myrmecocystus romainei | Formicinae | Jul | |
| Myrmecocystus semirufus | Formicinae | Mar | |
| Myrmecocystus testaceus | Formicinae | Mar • Apr • May | |
| Myrmecocystus yuma | Formicinae | Jul • Aug | |
| Myrmica aloba | Myrmicinae | Jul • Aug • Sep | |
| Myrmica bibikoffi | Myrmicinae | Aug | |
| Myrmica constricta | Myrmicinae | Aug • Sep | |
| Myrmica deplanata | Myrmicinae | Jul • Aug • Sep | |
| Myrmica forcipata | Myrmicinae | Jul • Aug | |
| Myrmica gallienii | Myrmicinae | Aug • Sep • Oct | |
| Myrmica hellenica | Myrmicinae | Aug • Sep • Oct | |
| Myrmica hirsuta | Myrmicinae | Aug • Sep | |
| Myrmica jessensis | Myrmicinae | Sep | Japan |
| Myrmica karavajevi | Myrmicinae | Jul • Aug • Sep | |
| Myrmica kotokui | Myrmicinae | Aug • Sep • Oct | Japan |
| Myrmica kurokii | Myrmicinae | Aug | |
| Myrmica lobicornis | Myrmicinae | Jul • Aug • Sep | |
| Myrmica lobulicornis | Myrmicinae | Jul • Aug • Sep | |
| Myrmica lonae | Myrmicinae | Jul • Aug • Sep | |
| Myrmica pinetorum | Myrmicinae | May | |
| Myrmica pisarskii | Myrmicinae | Jul • Aug • Sep | |
| Myrmica punctiventris | Myrmicinae | Sep • Oct • Nov | |
| Myrmica ruginodis | Myrmicinae | Jul • Aug | |
| Myrmica rugulosa | Myrmicinae | Aug • Sep • Oct | |
| Myrmica sabuleti | Myrmicinae | Jul • Aug • Sep | |
| Myrmica salina | Myrmicinae | Aug | |
| Myrmica scabrinodis | Myrmicinae | Jul • Aug • Sep | |
| Myrmica schencki | Myrmicinae | Jul • Aug | |
| Myrmica specioides | Myrmicinae | Jul • Aug • Sep | |
| Myrmica sulcinodis | Myrmicinae | Jul • Aug • Sep | |
| Myrmica vandeli | Myrmicinae | Jul • Aug • Sep | |
| Myrmica wesmaeli | Myrmicinae | Aug • Sep | |
| Myrmicaria natalensis | Myrmicinae | Jan | |
| Myrmicocrypta dilacerata | Myrmicinae | Jan • Feb • Mar • Apr • May • Jun • Dec | |
| Neivamyrmex klugii distans | Dorylinae | Mar • Apr • May • Jun | |
| Neivamyrmex melshaemeri | Dorylinae | Apr • May • Jun • Jul • Aug | |
| Neivamyrmex nigrescens | Dorylinae | Jul | |
| Neivamyrmex pilosus | Dorylinae | Mar • Apr • May | late dry season to early wet season |
| Neivamyrmex pseudops | Dorylinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Dec | |
| Neivamyrmex swainsonii | Dorylinae | Mar • Apr • May | |
| Novomessor cockerelli | Myrmicinae | Jul | |
| Nylanderia faisonensis | Formicinae | Apr • May | |
| Nylanderia flavipes | Formicinae | May • Jun | Japan |
| Nylanderia parvula | Formicinae | May • Jun | |
| Nylanderia terricola | Formicinae | Feb | |
| Nylanderia vaga | Formicinae | Oct | |
| Nylanderia vividula | Formicinae | Mar • Apr | |
| Odontomachus bauri | Ponerinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Odontomachus clarus | Ponerinae | Jun • Jul | |
| Odontomachus simillimus | Ponerinae | Feb | |
| Pachycondyla harpax | Ponerinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Nov • Dec | |
| Paraparatrechina sakurae | Formicinae | Oct • Nov | Japan |
| Paraponera clavata | Paraponerinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec | |
| Paratrechina longicornis | Formicinae | Jun • Jul | |
| Pheidole bicarinata | Myrmicinae | Jun • Jul • Aug | |
| Pheidole californica | Myrmicinae | Apr • May | |
| Pheidole ceres | Myrmicinae | Jul | |
| Pheidole christopherseni | Myrmicinae | Jan • Feb • Mar • Nov • Dec | |
| Pheidole dentata | Myrmicinae | Apr • May • Jun | |
| Pheidole dentigula | Myrmicinae | May | |
| Pheidole desertorum | Myrmicinae | Jun • Jul • Aug | |
| Pheidole diversipilosa | Myrmicinae | Jun | |
| Pheidole flavens | Myrmicinae | May | |
| Pheidole floridana | Myrmicinae | Apr • May | |
| Pheidole gilvescens | Myrmicinae | Aug | |
| Pheidole hyatti | Myrmicinae | May • Jun • Jul | |
| Pheidole megacephala | Myrmicinae | Aug | |
| Pheidole moerens | Myrmicinae | May • Jun • Jul | |
| Pheidole morrisii | Myrmicinae | May • Jun | |
| Pheidole navigans | Myrmicinae | Jul • Aug | |
| Pheidole obscurithorax | Myrmicinae | May • Jun • Jul | |
| Pheidole obtusospinosa | Myrmicinae | Jul • Aug • Sep | |
| Pheidole pallidula | Myrmicinae | May • Jun • Jul • Aug | |
| Pheidole pilifera | Myrmicinae | Jun • Jul • Aug | |
| Pheidole tysoni | Myrmicinae | Aug | |
| Pheidole vistana | Myrmicinae | Jun • Jul | |
| Pheidole xerophila | Myrmicinae | Jul • Aug | |
| Plagiolepis pallescens | Formicinae | Jun • Jul • Aug | |
| Plagiolepis xene | Formicinae | Jul • Aug | |
| Pogonomyrmex badius | Myrmicinae | May • Jun • Jul | |
| Pogonomyrmex barbatus | Myrmicinae | Apr • May • Jun • Jul • Aug • Sep | |
| Pogonomyrmex californicus | Myrmicinae | Apr • May • Jun • Jul • Aug | |
| Pogonomyrmex magnacanthus | Myrmicinae | Apr • May | |
| Pogonomyrmex maricopa | Myrmicinae | Jul • Aug | |
| Pogonomyrmex montanus | Myrmicinae | Jul | |
| Pogonomyrmex occidentalis | Myrmicinae | Aug • Sep | |
| Pogonomyrmex rugosus | Myrmicinae | Jul • Aug • Sep | |
| Pogonomyrmex subdentatus | Myrmicinae | Mar • Apr • May • Jun | |
| Pogonomyrmex subnitidus | Myrmicinae | Jun | |
| Pogonomyrmex tenuispinus | Myrmicinae | Jul | |
| Polyergus breviceps | Formicinae | Aug • Sep | |
| Polyergus rufescens | Formicinae | Jul • Aug • Sep | |
| Polyergus topoffi | Formicinae | Jul | |
| Polyergus vinosus | Formicinae | May | |
| Polyrhachis lamellidens | Formicinae | Sep • Oct • Nov | |
| Ponera coarctata | Ponerinae | Aug • Sep | |
| Ponera kohmoku | Ponerinae | Aug | Japan |
| Ponera pennsylvanica | Ponerinae | Mar • Apr • May | |
| Poneracantha triangularis | Ectatomminae | Jul | |
| Prenolepis imparis | Formicinae | Jan • Feb • Mar • Apr • May • Jun • Dec | |
| Prenolepis nitens | Formicinae | Apr • May | |
| Proatta butteli | Myrmicinae | Apr | |
| Proceratium chickasaw | Proceratiinae | Aug | |
| Proceratium silaceum | Proceratiinae | Jan | |
| Procryptocerus belti | Myrmicinae | May • Jun • Jul • Aug • Sep • Nov • Dec | |
| Pseudomyrmex apache | Pseudomyrmecinae | Apr | |
| Pseudomyrmex caeciliae | Pseudomyrmecinae | May | |
| Pseudomyrmex ejectus | Pseudomyrmecinae | May • Jun • Jul | |
| Pseudomyrmex gracilis | Pseudomyrmecinae | Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov | |
| Solenopsis amblychila | Myrmicinae | Apr | |
| Solenopsis carolinensis | Myrmicinae | May • Jun | |
| Solenopsis fugax | Myrmicinae | Aug • Sep | |
| Solenopsis geminata | Myrmicinae | Apr • May | |
| Solenopsis invicta | Myrmicinae | Apr • May • Jun • Jul | |
| Solenopsis krockowi | Myrmicinae | Jul • Aug | |
| Solenopsis molesta | Myrmicinae | Jun • Jul • Aug | |
| Solenopsis pergandei | Myrmicinae | May • Jun | |
| Solenopsis tennesseensis | Myrmicinae | Jul | |
| Solenopsis texana | Myrmicinae | Jul | |
| Stenamma debile | Myrmicinae | Aug • Sep • Oct | |
| Stenamma nipponense | Myrmicinae | Sep | Japan |
| Stigmatomma pallipes | Amblyoponinae | Aug • Sep | |
| Strongylognathus alpinus | Myrmicinae | Jul • Aug • Sep • Oct | |
| Strongylognathus bulgaricus | Myrmicinae | Jul • Aug • Sep | |
| Strongylognathus huberi | Myrmicinae | Jul • Aug • Sep | |
| Strongylognathus testaceus | Myrmicinae | Jun • Jul • Aug | |
| Strumigenys dolichognatha | Myrmicinae | Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov | |
| Strumigenys elongata | Myrmicinae | Feb • Mar • Apr • May • Jun • Jul • Aug • Sep | |
| Strumigenys gundlachi | Myrmicinae | Apr • May • Jun • Jul • Aug • Sep | |
| Strumigenys lewisi | Myrmicinae | Aug | Japan |
| Strumigenys margaritae | Myrmicinae | Jul • Aug | |
| Strumigenys membranifera | Myrmicinae | Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov | |
| Strumigenys minutula | Myrmicinae | Jun • Jul | Hong Kong |
| Strumigenys zeteki | Myrmicinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug | |
| Syscia augustae | Dorylinae | Jun • Jul • Aug | |
| Tapinoma erraticum | Dolichoderinae | May • Jun • Jul | |
| Tapinoma glabrella | Dolichoderinae | May • Jun | |
| Tapinoma insularis | Dolichoderinae | Apr | |
| Tapinoma madeirense | Dolichoderinae | Jun • Jul | |
| Tapinoma magnum | Dolichoderinae | Apr • May • Jun • Jul • Aug • Sep | April-June in Italy, Germany, and the Netherlands; August-September in Algeria |
| Tapinoma minutum | Dolichoderinae | Mar | |
| Tapinoma sessile | Dolichoderinae | Apr • May • Jun • Jul | |
| Technomyrmex albipes | Dolichoderinae | Jul | |
| Technomyrmex gibbosus | Dolichoderinae | Sep | Japan |
| Temnothorax affinis | Myrmicinae | Jul • Aug | |
| Temnothorax albipennis | Myrmicinae | Jul | |
| Temnothorax andrei | Myrmicinae | Jun • Jul | |
| Temnothorax caguatan | Myrmicinae | Jun • Jul | |
| Temnothorax congruus | Myrmicinae | Jul | Japan |
| Temnothorax corticalis | Myrmicinae | Jul • Aug • Sep | |
| Temnothorax crassispinus | Myrmicinae | Jul • Aug • Sep | |
| Temnothorax curvispinosus | Myrmicinae | Jun • Jul • Aug | |
| Temnothorax interruptus | Myrmicinae | Jun • Jul • Aug • Sep | |
| Temnothorax nigriceps | Myrmicinae | Jun • Jul • Aug • Sep | |
| Temnothorax nylanderi | Myrmicinae | Jul • Aug • Sep | |
| Temnothorax parvulus | Myrmicinae | Aug • Sep | |
| Temnothorax ravouxi | Myrmicinae | Jun • Jul • Aug | |
| Temnothorax saxonicus | Myrmicinae | Jul | |
| Temnothorax sordidulus | Myrmicinae | Jul | |
| Temnothorax spinosior | Myrmicinae | Jul | Japan |
| Temnothorax tuberum | Myrmicinae | Jun • Jul • Aug | |
| Temnothorax unifasciatus | Myrmicinae | Jun • Jul • Aug | |
| Temnothorax vivianoi | Myrmicinae | Aug | Sicily, Italy |
| Tetramorium atratulum | Myrmicinae | May • Jun • Jul • Aug • Sep | |
| Tetramorium bicarinatum | Myrmicinae | May • Jun | |
| Tetramorium caespitum | Myrmicinae | Jun • Jul • Aug | |
| Tetramorium hungaricum | Myrmicinae | Jun | |
| Tetramorium immigrans | Myrmicinae | Jul | |
| Tetramorium impurum | Myrmicinae | Jul • Aug • Sep • Oct | |
| Tetramorium moravicum | Myrmicinae | May • Jun | |
| Tetramorium semilaeve | Myrmicinae | Jul • Aug | |
| Trachymyrmex septentrionalis | Myrmicinae | May • Jun • Jul • Aug | |
| Typhlomyrmex rogenhoferi | Ectatomminae | Mar • Apr • May • Jun • Jul • Aug | |
| Veromessor andrei | Myrmicinae | Jun • Jul | |
| Veromessor julianus | Myrmicinae | Feb • Mar | |
| Veromessor pergandei | Myrmicinae | Feb • Mar • Apr | |
| Wadeura guianensis | Ponerinae | Jul • Aug • Sep • Oct • Nov | |
| Wasmannia auropunctata | Myrmicinae | Apr • May • Jun • Jul • Aug • Sep • Oct • Nov | |
| Xenomyrmex stollii | Myrmicinae | Jan • Feb • Mar • Apr • May • Jun • Jul • Aug • Sep • Oct • Nov • Dec |
Sort by Month
References
- Boomsma, J.J., Leusink, A. 1981. Weather conditions during nuptial flights of four European ant species. Oecologia 50, 236-241.
- Feitosa, R.M., da Silvab, R.R., Aguiarc, A.P. 2016. Diurnal flight periodicity of a Neotropical ant assemblage (Hymenoptera, Formicidae) in the Atlantic Forest. Revista Brasileira de Entomologia 60: 241-247 (doi:10.1016/j.rbe.2016.05.006).
- Franks, N.R., Sendova-Franks, A.B., Sendova-Vassileva, M., Vassilev, L. 1991. Nuptial flights and calling behaviour in the ant Leptothomx acervorum (Fabr.). Insectes Sociaux 38, 327–330 (doi:10.1007/BF01314918).
- Hölldobler, B., Wilson, E.O. 1990. The Ants. Cambridge, Mass. Harvard University Press.
- Torres, J.A., Snelling, R., Canals, M. 2001. Seasonal and nocturnal periodicities in ant nuptial flights in the Tropics (Hymenoptera: Formicidae). Sociobiology 37: 601-626.
| ||||||||||||||||||||||||||