Pseudomyrmex gracilis
| Pseudomyrmex gracilis | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Pseudomyrmecinae |
| Genus: | Pseudomyrmex |
| Species: | P. gracilis |
| Binomial name | |
| Pseudomyrmex gracilis (Fabricius, 1804) | |
| Subspecies | |
| |
| Synonyms | |
| |
| Common Name | |
|---|---|
| Slender Twig Ant | |
| Language: | English |
A wide ranging species that is quite variable in its appearance. This ant can also be found in many different habitats. Pseudomyrmex gracilis typically nest in dead twigs or branches but will occasional be found in myrmecophytic plants. When nesting in a myrmecophyte, they play the role of a parasite of the ant-plant mutualism.
| At a Glance | • Invasive |
Photo Gallery
Identification
Similar to Pseudomyrmex hospitalis. See P. hospitalis and the nomenclature section below.
Keys including this Species
Distribution
Southern United States (introduced into Florida) to Argentina and Brazil (Ward 1993).
Latitudinal Distribution Pattern
Latitudinal Range: 31.374783° to -34.583333°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Indo-Australian Region: Hawaii.
Nearctic Region: United States.
Neotropical Region: Argentina, Barbados, Belize, Bolivia, Brazil, Colombia, Costa Rica, Ecuador, El Salvador, French Guiana, Greater Antilles, Guadeloupe, Guatemala, Guyana, Honduras, Mexico, Nicaragua, Panama, Paraguay, Peru, Suriname, Trinidad and Tobago, Uruguay, Venezuela.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
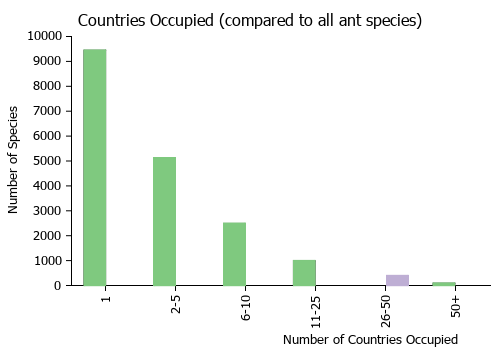
|
Biology
Ward (1993) - Befitting its wide distribution and variable phenotype, P. gracilis can be found in a variety of habitats from mangroves and thorn scrub to rainforest. It is often particularly common in disturbed situations such as old fields, roadsides, and secondary forest. Nests are usually located in dead twigs or small branches, but there are a substantial number of records of colonies occupying swollen-thorn acacias in Central America (Mexico to Panama). In a few localities P. gracilis is a common acacia inhabitant and under these circumstances it may exhibit local adaptation and phenotypic differentiation (see also Wheeler 1942:107). For example, Janzen collected a series of specimens from Acacia gentlei in Belize (15 mi. S Santa Elena) which have somewhat distinctive morphology: the workers are large, dark, abundantly hairy, and possess rather short petioles (PU 0.55), although none of these features is outside the total range of variation for the species. Janzen (1974:98) notes that the workers of this large black morph have atypically aggressive behavior. Given the kind of ecotypic variation to which P. gracilis is prone, it is not surprising to find a tendency of some populations to specialize on acacias. The ecology of this species is reminiscent of other animal species which show broad ecophenotypic variation, e.g. fish with trophic polymorphisms (Kornfield et al. 1982; Grudzien and Turner 1984; Sandlund et al. 1992).
Fonseca-Romero et al. (2019) - This ant opportunistically inhabits Turnera velutina (previously Acacia hindsii) plants, effectively exploiting the plant by using the hollow thorns, extrafloral nectaries, and beltian bodies while not providing robust defenses to the plant at the level of the aggressive ant Pseudomyrmex ferrugineus. The lack of a strong defensive functions provided by P. gracilis alters the plant through their producing thicker leaves and reducing their production of ant food rewards.
Brazil
Schmid et al. (2014) found this ant nesting in infructescences (the stem and remains of buds and fruits above the level of the water reservoir in the rosette) of the bromeliad Vriesea friburgensis on Santa Catarina Island, Brazil.
De Oliveira et al. (2015), studying ant occupancy of Cecropia trees in southwest Bahia, Brazil, found a colony of Pseudomyrmex gracilis nesting in a Cecropia pachystachya tree.
DaRocha et al. (2015) studied the diversity of ants found in bromeliads of a single large tree of Erythrina, a common cocoa shade tree, at an agricultural research center in Ilhéus, Brazil. Forty-seven species of ants were found in 36 of 52 the bromeliads examined. Bromeliads with suspended soil and those that were larger had higher ant diversity. Pseudomyrmex gracilis was found in 7 different bromeliads but was associated with twigs and bark cavities, rather than suspended soil or litter, of the plant.
Sanz-Veiga et al. (2017) observed this species visiting extrafloral nectaries of Tocoyena formosa plants in a southeastern Brazilian cerrado study-site.
Koch et al. (2018) sampled this species in Caryocar barsiliense trees, in southeastern Brazil cerrado, as part of a study examining species interactions in ant-plants.
Florida
Deyrup, Davis & Cover (2000): This introduced species is much larger than any of the seven native species of Pseudomyrmex. It might compete with these native species for food, but seems to nest in larger diameter holes in twigs and stems. It is more likely to exclude various native carpenter ants: Camponotus decipiens, Camponotus snellingi, Colobopsis impressa, Camponotus nearcticus and Camponotus discolor. It is a very abundant species in south and central Florida, and does not seem to distinguish between disturbed and undisturbed habitats. Aside from competition with native arboreal ants, this species could affect native phytophagous species, especially butterflies and moths. The buildup of populations of P. gracilis in Florida make it highly probable that it will be accidentally introduced into the West Indies, where it could have greater impact than in Florida.
Flight Period
| X | X | X | X | X | X | X | X | X | |||
| Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec |
Source: antkeeping.info.
- Check details at Worldwide Ant Nuptial Flights Data, AntNupTracker and AntKeeping.
 Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Castes
Worker
Images from AntWeb
   
| |
| Worker. Specimen code casent0103874. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
   
| |
| Worker. Specimen code casent0173760. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ALWC, Alex L. Wild Collection. |
   
| |
| Worker. Specimen code casent0173763. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ALWC, Alex L. Wild Collection. |
   
| |
| Worker. Specimen code casent0005838. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by UCDC, Davis, CA, USA. |
Queen
Images from AntWeb
   
| |
| Queen (alate/dealate). Specimen code casent0103779. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
   
| |
| Queen (alate/dealate). Specimen code casent0173762. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ALWC, Alex L. Wild Collection. |
Male
Images from AntWeb
    
| |
| Male (alate). Specimen code casent0173761. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ALWC, Alex L. Wild Collection. |
    
| |
| Male (alate). Specimen code casent0104250. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- gracilis. Formica gracilis Fabricius, 1804: 405 (w.) CENTRAL AMERICA. Wheeler, W.M. & Bailey, 1920: 256 (l.); Wheeler, G.C. & Wheeler, J. 1956: 385 (l.). Combination in Leptalea: Erichson, 1839: 309; in Pseudomyrma: Roger, 1862c: 289; in P. (Clavanoda): Enzmann, E.V. 1944: 61; in Pseudomyrmex: Kusnezov, 1953e: 214. Senior synonym of longinoda: Brown, 1949a: 43; Kempf, 1961a: 370; of variabilis: Ward, 1989: 439; of bicolor, canescens, dimidiata, glabriventris, mexicana, pilosula, sericata, velifera, volatilis and material of the unavailable name guayaquilensis referred here: Ward, 1993: 155; of atrinoda: Wild, 2007b: 55. Current subspecies: nominal plus argentinus. See also: Ward, 1999b: 521.
- bicolor. Pseudomyrma bicolor Guérin-Méneville, 1844a: 427 (w.) COLOMBIA. Combination in Pseudomyrmex: Kempf, 1958f: 434. Junior synonym of gracilis: Roger, 1862c: 289; Mayr, 1863: 452. Revived from synonymy as subspecies of gracilis: Emery, 1890b: 59; Santschi, 1925d: 223; Wheeler, W.M. 1942: 166. Junior synonym of gracilis: Ward, 1993: 155.
- sericata. Pseudomyrma sericata Smith, F. 1855c: 159 (w.) BRAZIL. Gallardo, 1932a: 50 (m.). Combination in Pseudomyrmex: Kempf, 1958f: 434. Status as species: Forel, 1912g: 19. Subspecies of gracilis: Emery, 1890b: 60; Santschi, 1916e: 370; Gallardo, 1932a: 48. Junior synonym of gracilis: Ward, 1993: 155.
- dimidiata. Pseudomyrma dimidiata Roger, 1863a: 177 (w.) COLOMBIA. Combination in Pseudomyrmex: Kempf, 1958f: 434. Subspecies of gracilis: Mayr, 1870a: 406; Forel, 1912g: 19. Junior synonym of gracilis: Ward, 1993: 155.
- mexicana. Pseudomyrma mexicana Roger, 1863a: 178 (w.) MEXICO. Wheeler, G.C. & Wheeler, J. 1956: 385 (l.); Petralia & Vinson, 1980: 381 (l.). Combination in Leptalea: Smith, M.R. 1951a: 788; in Pseudomyrmex: Kusnezov, 1953e: 214. Subspecies of gracilis: Mayr, 1870a: 409; Emery, 1890b: 45; Forel, 1907e: 7; Creighton, 1950a: 80. Revived status as species: Whitcomb, Denmark, et al. 1972: 31. Junior synonym of gracilis: Ward, 1993: 155. See also: Ward, 1985b: 225.
- canescens. Pseudomyrma canescens Smith, F. 1877b: 66 (q.) BRAZIL. Combination in Pseudomyrmex: Kempf, 1958f: 434. Junior synonym of gracilis: Ward, 1993: 155.
- pilosula. Pseudomyrma pilosula Smith, F. 1877b: 62 (w.) BARBADOS. Combination in Pseudomyrmex: Kempf, 1958f: 435. Junior synonym of gracilis: Ward, 1993: 155.
- variabilis. Pseudomyrma variabilis Smith, F. 1877b: 62 (w.) BARBADOS. Combination in Pseudomyrmex: Kempf, 1958f: 435. Junior synonym of gracilis: Ward, 1989: 439.
- volatilis. Pseudomyrma volatilis Smith, F. 1877b: 65 (m.) MEXICO. Combination in Pseudomyrmex: Kempf, 1958f: 435. Junior synonym of gracilis: Ward, 1993: 155.
- glabriventris. Pseudomyrma gracilis var. glabriventris Santschi, 1922b: 345 (w.) BOLIVIA. Combination in Pseudomyrmex: Kempf, 1958f: 434. Junior synonym of gracilis: Ward, 1993: 156.
- velifera. Pseudomyrma gracilis var. velifera Stitz, 1933: 68 (q.) GUATEMALA. Combination in Pseudomyrmex: Kempf, 1958f: 434. Junior synonym of gracilis: Ward, 1993: 156.
- atrinoda. Pseudomyrma gracilis var. atrinoda Santschi, 1934c: 26 (w.q.) BRAZIL. Combination in Pseudomyrmex: Kempf, 1958f: 434. Junior synonym of gracilis: Wild, 2007b: 55.
- longinoda. Pseudomyrma gracilis var. longinoda Enzmann, E.V. 1944: 87 (w.) PERU. Junior synonym of gracilis: Brown, 1949a: 43; Kempf, 1961a: 370.
Type Material
Ward (1993):
- Pseudomyrmex gracilis Fabricius, 1804: 405. Lectotype worker, Essequibo, Guyana (Zoologisk Museum, University of Copenhagen) [Examined].
- Pseudomyrma bicolor Guerin 1844:427. Syntype queen (unique?), Colombia (Zoologische Staatssammlung, Munich) [Examined].
- Pseudomyrma sericata F. Smith 1855:159. Holotype (unique syntype) worker, Brazil (The Natural History Museum) [Examined].
- Pseudomyrma dimidiata Roger 1863a:177. Syntype workers, Colombia (not in Musee National d'Histoire Naturelle or Berlin Museum für Naturkunde der Humboldt-Universität) [Not examined].
- Pseudomyrma mexicana Roger 1863a:178. Syntype workers, Mexico (not in MNHN or ZMHB) [Not examined].
- Pseudomyrma variabilis F. Smith 1877:62. Lectotype worker, Barbadoes (BMNH) [Examined].
- Pseudomyrma pilosula F. Smith 1877:62. Two syntype workers, Barbadoes (BMNH) [Examined]. One syntype here designated LECTOTYPE.
- Pseudomyrma volatilis F. Smith 1877:65. Holotype (unique syntype) male, Mexico (BMNH) [Examined].
- Pseudomyrma canescens F. Smith 1877:66. Holotype (unique syntype) queen, Abydos, Brazil (BMNH) [Examined].
- Pseudomyrma gracilis var. glabriventris Santschi 1922:345. Syntype workers, Izozo, Bolivia (Lizer & Deletang) (NHMB) [Examined].
- Pseudomyrma gracilis mexicana var. guayaquilensis Forel 1907:7. Worker, Guayaquil, Ecuador (Buchwald) (Musee d'Histoire Naturelle Genève) [Examined].
- Pseudomyrma gracilis var. velifera Stitz 1933:68. Holotype queen, Champerico, Guatemala (Paessler) (not in Zoologisches Institut und Zoologisches Museum der Universität Hamburg; Weidner 1972) [Not examined].
- Pseudomyrma gracilis var. longinoda Enzmann 1945:87. Syntype worker, Peru (Museum of Comparative Zoology) [Examined].
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Ward (1993):
The P. gracilis complex presents one of the more taxonomically challenging problems in the genus Pseudomyrmex and the above treatment is by no means a final solution. The worker- and queen-based forms, newly synonymized under P. gracilis, fall within the bounds of the preceding diagnosis, but it is quite possible that my concept of this species will prove to be too broad. The types of P. dimidiatus, P. mexicanus and P. veliferus could not be located. They are judged to be junior synonyms on the basis of the original descriptions. The unique male holotype of P. volatilis is clearly a member of the P. gracilis group based on size (HW 1.48), mandibular dentition, pilosity, petiole shape, and shape of the parameres. In comparison with males of gracilis group species known to occur in Mexico, namely P. gracilis, P. major (see below), P. nigropilosus and P. opaciceps, the type specimen agrees best with P. gracilis.
The concept of P. gracilis adopted above encompasses an impressive amount of phenotypic variability. Collections from single regions often give the impression that this variation is distributed bimodally or multimodally, as more or less discrete morphs. For example, nest samples from Costa Rica can be segregated on the basis of worker morphology into (i) a large (HW > 1.80), usually lighter-colored form (with orange mesosoma, petiole, and postpetiole, and black head and gaster), (ii) a smaller, bicolored, usually more heavily infuscated form, and (iii) an all-black form of variable size. The first two are typically found in open or xeric habitats while the third is more common in closed forest, suggesting some ecotypic differentiation. Yet when large enough sample sizes are obtained all degrees of intermediacy in size and color are encountered, and the variation in color (less so size) can be seen among individuals (workers and alate queens) from the same nest. Thus, if there are ecotypes they do not appear to be reproductively isolated.
Left unresolved after the establishment of the above synonymy is the relationship of P. gracilis to the following nominal taxa: Pseudomyrmex alternans (Santschi), Pseudomyrma gracilis atrinoda (Santschi) , Pseudomyrmex gracilis argentinus (Santschi) and Pseudomyrmex santschii (Enzmann). But the following deserves recognition as a distinct species: Pseudomyrmex major (Forel 1899:91), stat. nov. (syntype worker, Pinos Altos, Chihuahua, Mexico (Buchan-Hepburn) (BMNH) (examined); original combination: Pseudomyrma gracilis var. major). Workers of Pseudomyrmex major can be distinguished from those of P. gracilis by their emarginated median clypeal lobe, less distinct anterior peduncle of the petiole, and larger average size. Males of P. major have broadened fore-tarsal segments. P. major is confined to western Mexico, where it occurs sympatrically with P. gracilis without showing signs of intergradation.
Ward (1989):
The lectotype worker of P. gracilis, although lacking a head, seems to correspond rather well to the concept of P. gracilis which has become prevalent in publications. The mesosoma, postpetiole, and gaster are dark brown to black, the petiole a contrasting light castaneous brown; fine, silvery (not black) pilosity covers most of the body, and the associated piligerous punctures subdue the lustre of the integument; the petiole is narrow and slender, with a long anterior peduncle; and the pronotum is margined laterally but not sharply so. I do not attach much taxonomic significance to the light-colored petiole. The P. gracilis lectotype worker agrees well with material from Kartabo, Guyana (leg. Wheeler) in which there is variable infuscation of the petiole. A second worker in the P. gracilis "type series" in ZMUC, with a red "TYPE" label, but no locality or identification label, is in fact not conspecific (it is a worker of Pseudomyrmex maculatus (F. Smith) and should be excluded from consideration as type.
There is a bewildering and variable array of forms, variously described as subspecies or "varieties" of P. gracilis, which require detailed taxonomic study. I suspect that most of these will prove to be synonyms of a single polytypic species (P. gracilis), but at this stage there is insufficient information about the intra- and inter-specific components of this variation. One unambiguous synonymy can be established here: the lectotype worker of P. variabilis (F. Smith) in BMNH agrees very closely with that of P. gracilis, the only substantial difference being that the P. variabilis petiole is black. I have designated a lectotype of P. variabilis because a second worker glued to the same card (and bearing therefore the same type label as P. variabilis) is that of a different species - P. maculatus (F. Smith)! I have printed a lectotype label for P. variabilis and marked the card shared by the two specimens in such a way that the P. maculatus worker is clearly excluded as a type specimen of P. variabilis.
Description
Ward (1993) - With the traits of the gracilis group and the following more specific features. Head broad, about as wide as long (CI 0.95-1.08); anterior margin of median clypeal lobe straight to broadly convex, rounded laterally; pronotum dorsolaterally marginate but not sharply so; in lateral view mesonotum more steeply inclined than basal face of propodeum; petiole long and slender (PU 0.46-0.57) with a distinct anterior peduncle; head densely punctulate with a subopaque to sublucid (not matte) appearance; standing pilosity abundant, fine, predominantly pale silvery-white (not black).
Size and color extremely variable (HW 1.39-2.07), varying from unicolorous black (appendages lighter) to unicolorous orange-brown, with many intermediate and bicolored combinations. In populations from Mesoamerica the gaster is typically black, or if paler (orange-brown) then it is usually accompanied by a similar light coloration of the mesosoma (and sometimes also the head).
Ward (1999) - Measurements (n=74). HL 1.38–1.99, HW 1.39–2.07, MFC 0.033–0.079, LHT1.10–1.84, CI 0.95–1.08, REL 0.54–0.65, REL2 0.54–0.65, FCI 0.02–0.04, FI 0.36–0.44, PLI 0.46–0.57, PWI 0.38–0.54.
With the traits of the P. gracilis group (i.e. palp formula 6,4; masticatory margin of mandible with 7–10 teeth; median clypeal lobe laterally rounded; eyes large and elongate; standing pilosity common on mesosoma dorsum and on external faces of tibiae; pronotum dorsolaterally submarginate) and the following more specific features. Head broad, about as wide as long (CI 0.95–1.08); anterior margin of median clypeal lobe straight to broadly convex; in lateral view mesonotum more steeply inclined than dorsal face of propodeum; petiole long and slender (PLI 0.46–0.57) with a well developed anterior peduncle; head and mesosoma densely and finely punctulate-coriarious to coriarious-imbricate, subopaque to sublucid (not matte) in appearance; standing pilosity abundant, fine, predominantly pale silvery-white (not black). Size and colour extremely variable (HW 1.39–2.07), varying from unicolorous black (appendages lighter) to unicolorous orange-brown, with many intermediate and bicoloured combinations.
Karyotype
- See additional details at the Ant Chromosome Database.
 Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
- 2n = 70, karyotype = 70A (Brazil) (Sposito et al., 2006).
References
- Alatorre-Bracamontes, C.E., Vásquez-Bolaños, M. 2010. Lista comentada de las hormigas (Hymenoptera: Formicidae) del norte de México. Dugesiana 17(1): 9-36.
- Albuquerque, E., Prado, L., Andrade-Silva, J., Siqueira, E., Sampaio, K., Alves, D., Brandão, C., Andrade, P., Feitosa, R., Koch, E., Delabie, J., Fernandes, I., Baccaro, F., Souza, J., Almeida, R., Silva, R. 2021. Ants of the State of Pará, Brazil: a historical and comprehensive dataset of a key biodiversity hotspot in the Amazon Basin. Zootaxa 5001, 1–83 (doi:10.11646/zootaxa.5001.1.1).
- Baena, M.L., Escobar, F., Valenzuela, J.E. 2019. Diversity snapshot of green–gray space ants in two Mexican cities. International Journal of Tropical Insect Science 40, 239–250 (doi:10.1007/s42690-019-00073-y).
- Barassé, V., Touchard, A., Téné, N., Tindo, M., Kenne, M., Klopp, C., Dejean, A., Bonnafé, E., Treilhou, M. 2019. The peptide venom composition of the fierce stinging ant Tetraponera aethiops (Formicidae: Pseudomyrmecinae). Toxins 11, 732 (doi:10.3390/toxins11120732).
- Baty, J.W., Bulgarella, M., Dobelmann, J., Felden, A., Lester, P.J. 2020. Viruses and their effects in ants (Hymenoptera: Formicidae). Myrmecological News 30: 213-228 (doi:10.25849/MYRMECOL.NEWS_030:213).
- Brown, W. L., Jr. 1949a. Synonymic and other notes on Formicidae (Hymenoptera). Psyche (Camb.) 56: 41-49 (page 43, Senior synonym of longinoda)
- Camarota, F., Vasconcelos, H.L., Marquis, R.J., Powell, S. 2020. Revisiting ecological dominance in arboreal ants: how dominant usage of nesting resources shapes community assembly. Oecologia 194, 151–163 (doi:10.1007/s00442-020-04748-z).
- Cantone, S., Von Zuben, C.J. 2019. The hindwings of ants: A phylogenetic analysis. Psyche: A Journal of Entomology 2019, 1–11 (doi:10.1155/2019/7929717).
- DaRocha, W. D., S. P. Ribeiro, F. S. Neves, G. W. Fernandes, M. Leponce, and J. H. C. Delabie. 2015. How does bromeliad distribution structure the arboreal ant assemblage (Hymenoptera: Formicidae) on a single tree in a Brazilian Atlantic forest agroecosystem? Myrmecological News. 21:83-92.
- de Oliveira, G. V., M. M. Correa, I. M. A. Goes, A. F. P. Machado, R. J. de Sa-Neto, and J. H. C. Delabie. 2015. Interactions between Cecropia (Urticaceae) and ants (Hymenoptera: Formicidae) along a longitudinal east-west transect in the Brazilian Northeast. Annales De La Societe Entomologique De France. 51:153-160. doi:10.1080/00379271.2015.1061231
- Dekoninck, W., Wauters, N., Delsinne, T. 2019. Capitulo 35. Hormigas invasoras en Colombia. Hormigas de Colombia.
- Del-Claro, K., Rodriguez-Morales, D., Calixto, E.S., Martins, A.S., Torezan-Silingardi, H.M. 2019. Ant pollination of Paepalanthus lundii (Eriocaulaceae) in Brazilian savanna. Annals of Botany 123, 1159–1165 (doi:10.1093/aob/mcz021).
- Deyrup, M., Davis, L. & Cover, S. 2000. Exotic ants in Florida. Transactions of the American Entomological Society 126, 293-325.
- Deyrup, M.A., Carlin, N., Trager, J., Umphrey, G. 1988. A review of the ants of the Florida Keys. Florida Entomologist 71: 163-176.
- Enzmann, E. V. 1944. Systematic notes on the genus Pseudomyrma. Psyche (Camb.) 51: 59-103 (page 61, Combination in P. (Clavanoda))
- Erichson, W. F. 1839. Bericht über die Leistungen im Gebiete der Naturgeschichte während des Jahres 1838. IX. Insecten. Arch. Naturgesch. 5(2 2: 281-375 (page 309, Combination in Leptalea)
- Fabricius, J. C. 1804. Systema Piezatorum secundum ordines, genera, species, adjectis synonymis, locis, observationibus, descriptionibus. Brunswick: C. Reichard, xiv + 15-439 + 30 pp. (page 405, worker described)
- Fernandez, F., Guerrero, R.J., Sánchez-Restrepo, A.F. 2021. Sistemática y diversidad de las hormigas neotropicales. Revista Colombiana de Entomología 47, 1–20 (doi:10.25100/socolen.v47i1.11082).
- Fonseca-Romero, M. A., J. Fornoni, E. del-Val, and K. Boege. 2019. Ontogenetic trajectories of direct and indirect defenses of myrmecophytic plants colonized either by mutualistic or opportunistic ant species. Oecologia. 190:857-865. doi:10.1007/s00442-019-04469-y
- Franco, W., Ladino, N., Delabie, J.H.C., Dejean, A., Orivel, J., Fichaux, M., Groc, S., Leponce, M., Feitosa, R.M. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674, 509–543 (doi:10.11646/zootaxa.4674.5.2).
- Hill, J.G. 2015. Ants (Hymenoptera: Formicidae) of the Big Thicket Region of Texas. Midsouth Entomologist 8: 24-34.
- Kempf, W. W. 1961a. Estudos sôbre Pseudomyrmex. III. (Hymenoptera: Formicidae). Stud. Entomol. 4: 369-408 (page 370, Senior synonym of longinoda)
- Koch, E. B. A., W. Dattilo, F. Camarota, and H. L. Vasconcelos. 2018. From species to individuals: does the variation in ant-plant networks scale result in structural and functional changes? Population Ecology. 60:309-318. doi:10.1007/s10144-018-0634-5
- Koch, E.B.de A., Marques, T.E.D., Mariano, C.S.F., Neto, E.A.S., Arnhold, A., Peronti, A.L.B.G., Delabie, J.H.C. 2020. Diversity and structure preferences for ant-hemipteran mutualisms in cocoa trees (Theobroma cacao L., Sterculiaceae). Boletim do Museu Paraense Emílio Goeldi - Ciências Naturais 15, 65–81 (doi:10.46357/bcnaturais.v15i1.251).
- Kusnezov, N. 1953f. La fauna mirmecológica de Bolivia. Folia Univ. Cochabamba 6: 211-229 (page 214, Combination in Pseudomyrmex)
- Ladino, N., Feitosa, R.M. 2022. Ants (Hymenoptera: Formicidae) of the Parque Estadual São Camilo, an isolated Atlantic Forest remnant in western Paraná, Brazil. ZOOLOGIA 39: e22001 (doi:10.1590/S1984-4689.v39.e22001).
- Lau, M.K., Ellison, A.M., Nguyen, A., Penick, C., DeMarco, B., Gotelli, N.J., Sanders, N.J., Dunn, R.R., Helms Cahan, S. 2019. Draft Aphaenogaster genomes expand our view of ant genome size variation across climate gradients. PeerJ 7, e6447 (doi:10.7717/PEERJ.6447).
- Lubertazzi, D. 2019. The ants of Hispaniola. Bulletin of the Museum of Comparative Zoology, 162(2), 59-210 (doi:10.3099/mcz-43.1).
- Lutinski, J., de Filtro, M., Baucke, L., Dorneles, F., Lutinski, C., Guarda, C. 2021. Ant assemblages (Hymenoptera: Formicidae) from areas under the direct influence of two small hydropower plants in Brazil. Brazilian Journal of Environmental Sciences (Online), 1-9 (doi:10.5327/Z217694781030).
- Melo, T.S., Koch, E.B.A., Andrade, A.R.S., Travassos, M.L.O., Peres, M.C.L., Delabie, J.H.C. 2021. Ants (Hymenoptera: Formicidae) in different green areas in the metropolitan region of Salvador, Bahia state, Brazil. Brazilian Journal of Biology 82, e236269 (doi:10.1590/1519-6984.236269).
- Meurgey, F. 2020. Challenging the Wallacean shortfall: A total assessment of insect diversity on Guadeloupe (French West Indies), a checklist and bibliography. Insecta Mundi 786: 1–183.
- Moura, M.N., Cardoso, D.C., Cristiano, M.P. 2020. The tight genome size of ants: diversity and evolution under ancestral state reconstruction and base composition. Zoological Journal of the Linnean Society, zlaa135 (doi:10.1093/zoolinnean/zlaa135).
- Przybyszewski, K.R., Silva, R.J., Vicente, R.E., Garcia Freitas, J.V., Pereira, M.J.B., Izzo, T.J., Tonon, D.S. 2020. Can baited pitfall traps for sampling dung beetles replace conventional traps for sampling ants? Sociobiology 67, 376-387 (doi:10.13102/sociobiology.v67i3.5201).
- Ramalho, M.O., Duplais, C., Orivel, J., Dejean, A., Gibson, J.C., Suarez, A.V., Moreau, C.S. 2020. Development but not diet alters microbial communities in the Neotropical arboreal trap jaw ant Daceton armigerum: an exploratory study. Scientific Reports 10, 7350 (doi:10.1038/s41598-020-64393-7).
- Roger, J. 1862c. Synonymische Bemerkungen. 1. Ueber Formiciden. Berl. Entomol. Z. 6: 283-297 (page 289, Combination in Pseudomyrma)
- Sanz-Veiga, P. A., L. R. Jorge, S. Benitez-Vieyra, and F. W. Amorim. 2017. Pericarpial nectary-visiting ants do not provide fruit protection against pre-dispersal seed predators regardless of ant species composition and resource availability. PLoS ONE. 12. doi:10.1371/journal.pone.0188445
- Schmid V.S., Langner S., Steiner J. and Zillikens A. 2014. Inflorescences of the Bromeliad Vriesea friburgensis as Nest Sites and Food Resources for Ants and Other Arthropods in Brazil. Psyche. 2014:Article ID 396095. 9 pp. doi:10.1155/2014/396095
- Souza, C., Oliveira, F.M.P., Ribeiro, E.M.S., Silva, C.H.F., Durval, J.I.F., Leal, I.R. 2022. Extrafloral nectar secretion does not mediate ant predatory activity on Pityrocarpa moniliformis plants in a seasonally dry forest. Acta Oecologica 114, 103806 (doi:10.1016/j.actao.2021.103806).
- Tibcherani, M., Aranda, R., Mello, R.L. 2020. Time to go home: The temporal threshold in the regeneration of the ant community in the Brazilian savanna. Applied Soil Ecology 150, 103451 (doi:10.1016/j.apsoil.2019.103451).
- Ulysséa, M.A., Brandão, C.R.F. 2013. Ant species (Hymenoptera, Formicidae) from the seasonally dry tropical forest of northeastern Brazil: a compilation from field surveys in Bahia and literature records. Revista Brasileira de Entomologia 57, 217–224 (doi:10.1590/s0085-56262013005000002).
- Varela-Hernández, F., Medel-Zosayas, B., Martínez-Luque, E.O., Jones, R.W., De la Mora, A. 2020. Biodiversity in central Mexico: Assessment of ants in a convergent region. Southwestern Entomologist 454: 673-686.
- Ward, P. S. 1989a. Systematic studies on pseudomyrmecine ants: revision of the Pseudomyrmex oculatus and P. subtilissimus species groups, with taxonomic comments on other species. Quaest. Entomol. 25: 393-468 (page 439, Senior synonym of variabilis)
- Ward, P. S. 1993. Systematic studies on Pseudomyrmex acacia-ants (Hymenoptera: Formicidae: Pseudomyrmecinae). J. Hym. Res. 2: 117-168 (page 155, Senior synonym of bicolor, canescens, dimidiata, glabriventris, mexicana, pilosula, sericata, vilifera and volatilis)
- Ward, P. S. 1999b. Systematics, biogeography and host plant associations of the Pseudomyrmex viduus group (Hymenoptera: Formicidae), Triplaris- and Tachigali-inhabiting ants. Zool. J. Linn. Soc. 126: 451-540 (page 521, see also)
- Wetterer, J.K. 2021. Ants (Hymenoptera, Formicidae) of St. Vincent, West Indies. Sociobiology 68, e6725 (doi:10.13102/sociobiology.v68i2.6725).
- Wheeler, G. C.; Wheeler, J. 1956. The ant larvae of the subfamily Pseudomyrmecinae (Hymenoptera: Formicidae). Ann. Entomol. Soc. Am. 49: 374-398 (page 385, larva described)
- Wheeler, W. M.; Bailey, I. W. 1920. The feeding habits of pseudomyrmine and other ants. Trans. Am. Philos. Soc. (2) 22: 235-279 (page 256, larva described)
- Xiong, Z., He, D., Guang, X., Li, Q. 2023. Novel tRNA gene rearrangements in the mitochondrial genomes of poneroid ants and phylogenetic implication of Paraponerinae (Hymenoptera: Formicidae). Life 1310, 2068 (doi:10.3390/life13102068).
- Yanoviak, S.P., Frederick, D.N. 2014. Water surface locomotion in tropical canopy ants. Journal of Experimental Biology 217, 2163–2170 (doi:10.1242/jeb.101600).
References based on Global Ant Biodiversity Informatics
- Adams B. J., S. A. Schnitzer, and S. P. Yanoviak. 2016. Trees as islands: canopy ant species richness increases with the size of liana-free trees in a Neotropical forest. Ecography doi: 10.1111/ecog.02608
- Adams B. J., S. A. Schnitzer, and S. P. Yanoviak. 2019. Connectivity explains local ant community structure in a Neotropical forest canopy: a large-scale experimental approach. Ecology 100(6): e02673.
- Alatorre-Bracamontes, C.E. and M Vasquez-Bolanos. 2010. Lista comentada de las hormigas (Hymenoptera: Formicidae) del norte de México. Dugesiana 17(1):9-36
- Andrade T., G. D. V. Marques, K. Del-Claro. 2007. Diversity of ground dwelling ants in Cerrado: an analysis of temporal variations and distinctive physiognomies of vegetation (Hymenoptera: Formicidae). Sociobiology 50(1): 121-134.
- Antoniazzi R., R. N. S. L. Garoo, W. Dattilo, S. P. Ribeiro, and F. S. Neves. 2019. Ant species richness and interactions in canopies of two distinct successional stages in a tropical dry forest. The Science of Nature 106: 20
- Arruda F. V., M. A. Pesquero, D. G. Marcelino, G. A. Leiter, J. H. C. Delabie, and R. Fagundes. 2015. Size and condition of bamboo as structural factors behind the vertical stratification of the bamboo-nesting ant community. Insectes Sociaux DOI 10.1007/s00040-015-0440-4
- Baroni Urbani C. 1977. Katalog der Typen von Formicidae (Hymenoptera) der Sammlung des Naturhistorischen Museums Basel (2. Teil). Mitt. Entomol. Ges. Basel (n.s.) 27: 61-102.
- Battirola, L.E., M.I. Marques, J. Adis and J.H.C. Delabie. 2005. Composicao da comunidade de Formicidae (Insecta, Hymenoptera) em copas de Attalea phalerata Mart. Composic?a?o da comunidade de Formicidae (Insecta, Hymenoptera) em copas de Attalea phalerata Mart. (Arecaceae), no Pantanal de Pocone?, Mato Grosso, Brasil. Revista Brasileira de Entomologia 49(1): 107-117.
- Benitez-Malvido, J., W. Dattilo, A. P. Martinez-Falcon, C. Duran-Barron, J. Valenzuela, S. Lopez, and R. Lombera. 2016. The multiple impacts of tropical forest fragmentation on arthropod biodiversity and on their patterns of interactions with host plants. Plos One 11: e0146461.
- Bestelmeyer B. T., and R. L. Schooley. 1999. The ants of the southern Sonoran desert: community structure and the role of trees. Biodiversity and Conservation 8: 643-657.
- Bezdeckova K., P. Bedecka, and I. Machar. 2015. A checklist of the ants (Hymenoptera: Formicidae) of Peru. Zootaxa 4020 (1): 101–133.
- Biagiotti G., J. A. Alves Pereira, C. Rodrigues Ribas, V. Korasaki, R. Zanetti, A. C. Medeiros de Queiroz. 2013. Richness and species composition of ants in the recovery process of a Gully erosion. Cerne, Lavras 19(4): 661-668.
- Brandao, C.R.F. 1991. Adendos ao catalogo abreviado das formigas da regiao neotropical (Hymenoptera: Formicidae). Rev. Bras. Entomol. 35: 319-412.
- Branstetter M. G. and L. Sáenz. 2012. Las hormigas (Hymenoptera: Formicidae) de Guatemala. Pp. 221-268 in: Cano E. B. and J. C. Schuster. (eds.) 2012. Biodiversidad de Guatemala. Volumen 2. Guatemala: Universidad del Valle de Guatemala, iv + 328 pp
- Bruch C. 1914. Catálogo sistemático de los formícidos argentinos. Revista del Museo de La Plata 19: 211-234.
- Byk J., K. Del-Claro. 2010. Nectar- and pollen-gathering Cephalotes ants provide no protection against herbivory: a new manipulative experiment to test ant protective capabilities. Acta Ethol. 13: 33-38.
- Bächtold A., K. Del-Claro, L. A. Kaminski, A. V. L. Freitas, and P. S. Oliveira. 2012. Natural history of an ant-plant-butterfly interaction in a Neotropical savanna. Journal of Natural History 46: 943-954.
- Caldart V. M., S. Iop, J. A. Lutinski, and F. R. Mello Garcia. 2012. Ants diversity (Hymenoptera, Formicidae) of the urban perimeter of Chapecó county, Santa Catarina, Brazil. Revista Brasileira de Zoociências 14 (1, 2, 3): 81-94.
- Calle Z., N. Henao-Gallego, C. Giraldo, and I. Armbrecht. 2013. A Comparison of Vegetation and Ground-dwelling Ants in Abandoned and Restored Gullies and Landslide Surfaces in the Western Colombian Andes. Restoration Ecology 21(6): 729-736.
- Cameron P. 1891. Appendix. Hym. Formicidae. Pp. 89-95 in: Whymper, E. Travels amongst the Great Andes. London: J. Murray, xxii + 147 pp.
- Cancino, E.R., D.R. Kasparan, J.M.A. Coronado Blanco, S.N. Myartseva, V.A. Trjapitzin, S.G. Hernandez Aguilar and J. Garcia Jimenez. 2010. Himenópteros de la Reserva El Cielo, Tamaulipas, México. Dugesiana 17(1):53-71
- Castano-Meneses, G., M. Vasquez-Bolanos, J. L. Navarrete-Heredia, G. A. Quiroz-Rocha, and I. Alcala-Martinez. 2015. Avances de Formicidae de Mexico. Universidad Nacional Autonoma de Mexico.
- Castaño-Meneses G. 2012. Primeros registros de hormigas (Hymeoptera: Formicidae) en suelos de mangle blanco (Laguncularia racemosa) en Xcalak, Quintana Roo, México. Dugesiana 19(2): 113-116.
- Cereto, C.E., G.O. Schmidt, A.G. Martins, T.T. Castellani and B.C. Lopes. 2011. Nesting of ants (Hymenoptera, Formicidae) in dead post-reproductive plants of Actinocephalus polyanthus (Eriocaulaceae), a herb of coastal dunes in southern Brazil. Insectes Sociaux DOI 10.1007/s00040-011-0165-y
- Coelho M. S., G. W. Fernandes, J. C. Santos, and J. H. C. Delabie. 2009. Ants (Hymenoptera: Formicidae) as bioindicators of land restoration in a Brazilian Atlantic forest fragment. Sociobiology 54(1): 51-63.
- Cogni R., A. V. L. Freitas, and P. S. Oliveira. 2003. Interhabitat differences in ant activity on plant foliage: Ants at extrafloral nectaries of Hibiscus pernambucensis in sandy and mangrove forests. Entomologia Experimentalis et Applicata 107: 125-131.
- Cogni, R. and A.V.L. Freitas. 2002. The ant assemblage visiting extrafloral nectaries of Hibiscus pernambucensis (Malvaceae) in a Mangrove forest in Southeast Brazil (Hymenoptera: Formicidae). Sociobiology 40(2):373-383
- Coronado-Blanco J. M., E. Ruíz-Cancino, D. A. Dubovikoff, and M. Vásquez-Bolaños. 2015. Notas sobre Pseudomyrmex gracilis (Fabricius, 1804) (Hymenoptera: Formicidae: Pseudomyrmecinae) en el noreste de México. (pp. 101-107). En: Castaño-Meneses G., M. Vásquez-Bolaños, J. L. NavarreteHeredia, G. A. Quiroz-Rocha e I. Alcalá-Martínez (Coords.). Avances de Formicidae de México. UNAM, Universidad de Guadalajara, Guadalajara, Jalisco.
- Crawley W. C. 1916. Ants from British Guiana. Ann. Mag. Nat. Hist. 8(17): 366-378.
- Cuezzo, F. 1998. Formicidae. Chapter 42 in Morrone J.J., and S. Coscaron (dirs) Biodiversidad de artropodos argentinos: una perspectiva biotaxonomica Ediciones Sur, La Plata. Pages 452-462.
- Dattilo W. et al. 2019. MEXICO ANTS: incidence and abundance along the Nearctic-Neotropical interface. Ecology https://doi.org/10.1002/ecy.2944
- De La Mora, A., and S. M. Philpott. 2010. Wood-nesting ants and their parasites in forests and coffee agroecosystems. Environmental Entomology 39: 1473-1481.
- De la Mora, A., C. J. Murnen, and S. M. Philpott. 2013. Local and landscape drivers of biodiversity of four groups of ants in Neotropical coffee landscapes. Biodiversity and Conservation 22: 871-888.
- Del Toro, I., M. Vázquez, W.P. Mackay, P. Rojas and R. Zapata-Mata. Hormigas (Hymenoptera: Formicidae) de Tabasco: explorando la diversidad de la mirmecofauna en las selvas tropicales de baja altitud. Dugesiana 16(1):1-14.
- Delabie J. H. C., and I. C. do Nascimento. 1998. As Formigas do municipio de Ilhéus (Insecta: Hymenoptera: Formicidae). Especiaria 1(2): 133-152.
- Diehl E., C. L. Gottert, and D. G. Flores. 2012. Ant communities in three species used in urban arborization in Sao Leopoldo, state of Rio Grande do Sul, Brazil. Bioikos, Campinas 20(1): 25-32.
- Diehl, E., F.Sacchett and E.Z.de Albuquerque. 2005. Riqueza de formigas de solo na praia da Pedreira, Parque Estadual de Itapuã, Viamão, RS, Brasil. Revista Brasileira de Entomologia 49(4): 552-556
- Donoso D. A., G. Onore, G. Ramon, and J. E. Lattke. 2014. Invasive ants of continental Ecuador, a first account. REMCB 35: 133-141.
- Drose W., L. R. Podgaiski, C. Fagundes Dias, M. de Souza Mendonca. 2019. Local and regional drivers of ant communities in forest-grassland ecotones in South Brazil: A taxonomic and phylogenetic approach. Plos ONE 14(4): e0215310.
- Durou S., A. Dejean, I. Olmsted, and R. R. Snelling. 2002. Ant diversity in coastal zones of Quintana Roo, Mexico, with special reference to army ants. Sociobiology 40(0): 385-402.
- Eidmann H. 1936. Ökologisch-faunistische Studien an südbrasilianischen Ameisen. [part]. Arb. Physiol. Angew. Entomol. Berl.-Dahl. 3: 26-48.
- Emery C. 1890. Studii sulle formiche della fauna neotropica. Bull. Soc. Entomol. Ital. 22: 38-8
- Emery C. 1890. Voyage de M. E. Simon au Venezuela (Décembre 1887 - Avril 1888). Formicides. Annales de la Société Entomologique de France (6)10: 55-76.
- Emery C. 1894. Estudios sobre las hormigas de Costa Rica. Anales del Museo Nacional de Costa Rica 1888-1889: 45-64.
- Enzmann E. V. 1944. Systematic notes on the genus Pseudomyrma. Psyche (Camb.) 51: 59-103.
- Escalante Gutiérrez J. A. 1993. Especies de hormigas conocidas del Perú (Hymenoptera: Formicidae). Revista Peruana de Entomología 34:1-13.
- Escalante J. A. 1975. Hormigas de la Provincia de la Convencionm Cusco. Revista Peruana de Entomologia 18:125-126.
- Fabricius, J. C. 1804. Systema Piezatorum, secundum ordines, genera, species; adjectis synonymis, locis, observationibus, descriptionibus. Reichard.
- Favretto M. A., E. Bortolon dos Santos, and C. J. Geuster. 2013. Entomofauna from West of Santa Catarina State, South of Brazil. EntomoBrasilis 6 (1): 42-63.
- Fernandes T. T., R. R. Silva, D. Rodrigues de Souza-Campana, O. Guilherme Morais da Silva, and M. Santina de Castro Morini. 2019. Winged ants (Hymenoptera: Formicidae) presence in twigs on the leaf litter of Atlantic Forest. Biota Neotropica 19(3): http://dx.doi.org/10.1590/1676-0611-bn-2018-0694
- Fernández F., E. E. Palacio, W. P. Mackay, and E. S. MacKay. 1996. Introducción al estudio de las hormigas (Hymenoptera: Formicidae) de Colombia. Pp. 349-412 in: Andrade M. G., G. Amat García, and F. Fernández. (eds.) 1996. Insectos de Colombia. Estudios escogidos. Bogotá: Academia Colombiana de Ciencias Exactas, Físicas y Naturales, 541 pp
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Forel A. 1903. Faune myrmécologique des noyers dans le canton de Vaud. Bulletin de la Société Vaudoise des Sciences Naturelles 39: 83-94.
- Forel A. 1907. Formicides du Musée National Hongrois. Ann. Hist.-Nat. Mus. Natl. Hung. 5: 1-42.
- Forel A. 1908. Ameisen aus Sao Paulo (Brasilien), Paraguay etc. gesammelt von Prof. Herm. v. Ihering, Dr. Lutz, Dr. Fiebrig, etc. Verhandlungen der Kaiserlich-Königlichen Zoologisch-Botanischen Gesellschaft in Wien 58: 340-418.
- Forel A. 1908. Fourmis de Costa-Rica récoltées par M. Paul Biolley. Bulletin de la Société Vaudoise des Sciences Naturelles 44: 35-72.
- Forel A. 1912. Formicides néotropiques. Part IV. 3me sous-famille Myrmicinae Lep. (suite). Mémoires de la Société Entomologique de Belgique. 20: 1-32.
- Franco W., N. Ladino, J. H. C. Delabie, A. Dejean, J. Orivel, M. Fichaux, S. Groc, M. Leponce, and R. M. Feitosa. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674(5): 509-543.
- Gallardo A. 1932. Las hormigas de la República Argentina. Subfamilia Mirmicinas, sección Promyrmicinae. Anales del Museo Nacional de Historia Natural de Buenos Aires 37: 37-87.
- Garcia-Martinez M. A., I. Landero-Torres, O. R. Leiva-Ovalle, H. E. Lee-Espinosa, and M. E. Galindo-Tovar. 2013. Composición y riqueza del ensamble de hormigas en la Barranca de Metlác perteneciente al Parque Nacional Cañón del Río Blanco, Veracruz, México. In Formicidae de Mexico (eds. M. Vasquez-Bolanos, G. Castano-Meneses, A. Cisneros-Caballero, G. A. Quiroz-Rocha, and J. L. Navarrete-Heredia) p 115-122.
- Gazzana Flores D., C. L. Goettert, E. Diehl. 2002. Ant communities in Inga marginata and Jacaranda micrantha in a sub-urban area. Acta Biologica Leopoldensia 24(2): 147-155.
- Goitia W., and K. Jaffe. 2009. Ant-Plant Associations in Different Forests in Venezuela. Neotropical Entomology 38(1): 007-031.
- Gomes E. C. F., G. T. Ribeiro, T. M. S. Souza, and L. Sousa-Souto. 2014. Ant assemblages (Hymenoptera: Formicidae) in three different stages of forest regeneration in a fragment of Atlantic Forest in Sergipe, Brazil. Sociobiology 61(3): 250-257.
- Gove, A. D., J. D. Majer, and V. Rico-Gray. 2009. Ant assemblages in isolated trees are more sensitive to species loss and replacement than their woodland counterparts. Basic and Applied Ecology 10: 187-195.
- Groc S., J. H. C. Delabie, F. Fernandez, F. Petitclerc, B. Corbara, M. Leponce, R. Cereghino, and A. Dejean. 2017. Litter-dwelling ants as bioindicators to gauge the sustainability of small arboreal monocultures embedded in the Amazonian rainforest. Ecological Indicators 82: 43-49.
- Heil, M. 2013. Let the best one stay: screening of ant defenders by Acacia host plants functions independently of partner choice or host sanctions. Journal of Ecology 101: 684-688.
- Hernandez, F. Varela and G. Castano-Meneses. 2010. Checklist, Biological Notes and Distribution of Ants (Hymenoptera: Formicidae) from Barranca de Metztitlán Biosphere Reserve, Hidalgo, Mexico. Sociobiology 56(2):397-434
- INBio Collection (via Gbif)
- Ilha C., J. A. Lutinski, D. Von Muller Pereira, F. R. Mello Garcia. 2009. Riqueza de formigas (Hymenoptera: Formicidae) de Bacia da Sanga Caramuru, municipio de Chapeco-SC. Biotemas 22(4): 95-105.
- Iop S., V. M. Caldart, J. A. Lutinski, and F. R. Mello Garcia. 2009. Formigas urbanas da cidade de Xanxerê, Santa Catarina, Brasil. Biotemas 22(2): 55-64.
- Jaffe, Klaus and Lattke, John. 1994. Ant Fauna of the French and Venezuelan Islands in the Caribbean in Exotic Ants, editor D.F. Williams. 182-190.
- Johnson R. Personnal Database. Accessed on February 5th 2014 at http://www.asu.edu/clas/sirgtools/resources.htm
- Kamura, C.M., M.S.C. Morini, C.J. Figueiredo, O.C. Bueno, and A.E.C. Campos-Farinha. 2007. Comunidades de formigas (Hymenoptera: Formicidae) em um ecossistema urbano próximo à Mata Atlântica. Brazilian Journal of Biology 67(4): 635-641
- Kautz, S., D. J. Ballhorn, J. Kroiss, S. U. Pauls, C. S, Moreau, S. Eilmus, and M. Heil. 2012. Host plant use by competing acacia-ants: mutualists monopolize while parasites share hosts. Plos One 7, e37691.
- Kempf W. W. 1961. A survey of the ants of the soil fauna in Surinam (Hymenoptera: Formicidae). Studia Entomologica 4: 481-524.
- Kempf W. W., and K. Lenko. 1976. Levantamento da formicifauna no litoral norte e ilhas adjacentes do Estado de São Paulo, Brasil. I. Subfamilias Dorylinae, Ponerinae e Pseudomyrmecinae (Hym., Formicidae). Studia Entomologica 19: 45-66.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Kusnezov N. 1953. La fauna mirmecológica de Bolivia. Folia Universitaria. Cochabamba 6: 211-229.
- Landero-Torres I., I. Madrid-Neco, J. E. Valenzuela-Gonzalez, M. E. Galindo-Tovar, O. R. Leyva-Ovalle, J. Murguia-Gonzalez, H. E. Lee-Espinosa, and M. A. Garcia-Martinez. 2014. Myrmecofuana from three ornamental agroecosystems with different management and a forest remnant in Ixtaczoquitlan, Veracruz, Mexico. Southwestern Entomologist 39(4): 783-795.
- Larsen, A., and S. M. Philpott. 2010. Twig-nesting ants: the hidden predators of the coffee berry borer in Chiapas, Mexico. Biotropica 42: 342-347.
- Lattke J. E., and M. Velez, and N. Aguirre. 2016. Survey of ants in dry forests of Southwestern Ecuador (Hymenoptera: Formicidae). Sociobiology 63(3): 909-918.
- LeBrun E. G., R. M. Plowes, and L. E. Gilbert. 2015. Imported fire ants near the edge of their range: disturbance and moisture determine prevalence and impact of an invasive social insect. Journal of Animal Ecology,81: 884–895.
- Longino J. T. 2013. Ants of Honduras. Consulted on 18 Jan 2013. https://sites.google.com/site/longinollama/reports/ants-of-honduras
- Longino J. T. 2013. Ants of Nicargua. Consulted on 18 Jan 2013. https://sites.google.com/site/longinollama/reports/ants-of-nicaragua
- Longino J. T., J. Coddington, and R. K. Colwell. 2002. The ant fauna of a tropical rain forest: estimating species richness three different ways. Ecology 83: 689-702.
- Longino J. et al. ADMAC project. Accessed on March 24th 2017 at https://sites.google.com/site/admacsite/
- Lopes D. T., J. Lopes, I. Cardoso do Nascimento, and J. H. Delabie. 2010. Epigeic ants diversity (Hymenoptera, Formicidae) in three environments in Mata dos Godoy State Park, Londrina, State of Paraná, Brazil. Iheringia, Sér. Zool., Porto Alegre, 100(1): 84-90.
- Lopez-Moreno I. R., M. E. Diaz-Betancourt, and T. Suarez Landa. 2003. Insectos sociales en ambientes antropizados: las hormigas de la ciudad de Coatepec, Veracruz, Mexico. Sociobiology 42(3): 604-622.
- Luederwaldt H. 1918. Notas myrmecologicas. Rev. Mus. Paul. 10: 29-64.
- Luna Bello G. O. 2005. Composicion y estructura de la comunidad de hormigas a lo largo de un gradiente de intensificacion agricola en zonas de bosque tropical humedo en la region autonoma Atlantico Sur, Nicaragua. PhD Universidad Centroamericana, Facultad de Ciencia y Tecnologia del Ambiente carrera de Ecologi y Desarrollo. 127 pages
- Lutinski J. A., B. C. Lopes, and A. B. B.de Morais. 2013. Diversidade de formigas urbanas (Hymenoptera: Formicidae) de dez cidades do sul do Brasil. Biota Neotrop. 13(3): 332-342.
- Lutinski J. A., F. R. Mello Garcia, C. J. Lutinska, and S. Iop. 2008. Ants diversity in Floresta Nacional de Chapecó in Santa Catarina State, Brazil. Ciência Rural, Santa Maria 38(7): 1810-1816.
- Maes, J.-M. and W.P. MacKay. 1993. Catalogo de las hormigas (Hymenoptera: Formicidae) de Nicaragua. Revista Nicaraguense de Entomologia 23.
- Marinho C. G. S., R. Zanetti, J. H. C. Delabie, M. N. Schlindwein, and L. de S. Ramos. 2002. Ant (Hymenoptera: Formicidae) Diversity in Eucalyptus (Myrtaceae) Plantations and Cerrado Litter in Minas Gerais, Brazil. Neotropical Entomology 31(2): 187-195.
- Marques G. D. V., and K. Del-Claro. 2006. The Ant Fauna in a Cerrado area: The Influence of Vegetation Structure and Seasonality (Hymenoptera: Formicidae). Sociobiology 47(1): 1-18.
- McDonald D. L., D. R. Hoffpauir, and J. L. Cook. 2016. Survey yields seven new Texas county records and documents further spread of Red Imported Fire Ant, Solenopsis invicta Buren. Southwestern Entomologist, 41(4): 913-920.
- Menozzi C. 1926. Neue Ameisen aus Brasilien. Zoologischer Anzeiger. 69: 68-72.
- Menozzi C. 1927. Formiche raccolte dal Sig. H. Schmidt nei dintorni di San José di Costa Rica. Entomologische Mitteilungen. Berlin-Dahlem. 16: 266-277.
- Menozzi C. 1935. Spedizione del Prof. Nello Beccari nella Guiana Britannica. Hymenoptera-Formicidae. Redia. 21: 189-203.
- Mirmecofauna de la reserva ecologica de San Felipe Bacalar
- Morini M. S. de C., C. de B. Munhae, R. Leung, D. F. Candiani, and J. C. Voltolini. 2007. Comunidades de formigas (Hymenoptera, Formicidae) em fragmentos de Mata Atlântica situados em áreas urbanizadas. Iheringia, Sér. Zool., Porto Alegre, 97(3): 246-252.
- Munhae C. B., Z. A. F. N. Bueno, M. S. C. Morini, and R. R. Silva. 2009. Composition of the Ant Fauna (Hymenoptera: Formicidae) in Public Squares in Southern Brazil. Sociobiology 53(2B): 455-472.
- Nascimento Santos M., J. H. C. Delabie, and J. M. Queiroz. 2019. Biodiversity conservation in urban parks: a study of ground-dwelling ants (Hymenoptera: Formicidae) in Rio de Janeiro City. Urban Ecosystems https://doi.org/10.1007/s11252-019-00872-8
- Navarro, E.V. Vergara, H. Echavarria Sanchez, F.J. Serna Cardona. 2007. Hormigas (Hymenoptera: Formicidae) asociadas al arboretum de la Universidad Nacional de Colombia, sede Medellin. Boletín Sociedad Entomológica Aragonesa 40:497-505.
- Neves F. S., K. S. Queiroz-Dantas, W. D. da Rocha, and J. H. C. Delabie. 2013. Ants of Three Adjacent Habitats of a Transition Region Between the Cerrado and Caatinga Biomes: The Effects of Heterogeneity and Variation in Canopy Cover. Neotrop Entomol 42: 258268.
- Neves F. S., R. F. Braga, M. M. do Espirito-Santo, J. H. C. Delabie, G. Wilson Fernandes, and G. A. Sanchez-Azofeifa. 2010. Diversity of Arboreal Ants In a Brazilian Tropical Dry Forest: Effects Of Seasonality and Successional Stage. Sociobiology 56(1): 1-18.
- Novais, S. M., W. D. DaRocha, N. Calderon-Cortes, and M. Quesada. 2017. Wood-boring beetles promote ant nest cavities: extended effects of a twig-girdler ecosystem engineer. Basic and Applied Ecology 24: 53-59.
- O'Keefe S. T., J. L. Cook, T. Dudek, D. F. Wunneburger, M. D. Guzman, R. N. Coulson, and S. B. Vinson. 2000. The Distribution of Texas Ants. The Southwestern Entomologist 22: 1-92.
- Oliveira-Santos L. G. R., R. D. Loyola, A. B. Vargas. 2009. Canopy Traps: a Technique for Sampling Arboreal Ants in Forest Vertical Strata. Neotropical Entomology 38(5):691-694.
- Pacheco, R., R.R. Silva, M.S. de C. Morini, C.R.F. Brandao. 2009. A Comparison of the Leaf-Litter Ant Fauna in a Secondary Atlantic Forest with an Adjacent Pine Plantation in Southeastern Brazil. Neotropical Entomology 38(1):055-065
- Pereira M. C., J. H. C. Delabie, Y. R. Suarez, and W. F. Antonialli Junior. 2013. Spatial connectivity of aquatic macrophytes and flood cycle influence species richness of an ant community of a Brazilian floodplain. Sociobiology 60(1): 41-49.
- Pergande, T. 1895. Mexican Formicidae. Proceedings of the California Academy of Sciences Ser. 2 :850-896
- Philpott S. M., I. Perfecto, and J. Vandermeer. 2006. Effects of management intensity and season on arboreal ant diversity and abundance in coffee agroecosystems. 15: 139-155.
- Philpott, S.M. and P.F. Foster. 2005. Nest-site limitation in coffee agroecosytems: Artificial nests maintain diversity of arboreal ants. Ecological Applications 15(4):1478-1485
- Philpott, S.M., P. Bichier, R. Rice, and R. Greenberg. 2007. Field testing ecological and economic benefits of coffee certification programs. Conservation Biology 21: 975-985.
- Pignalberi C. T. 1961. Contribución al conocimiento de los formícidos de la provincia de Santa Fé. Pp. 165-173 in: Comisión Investigación Científica; Consejo Nacional de Investigaciones Científicas y Técnicas (Argentina) 1961. Actas y trabajos del primer Congreso Sudamericano de Zoología (La Plata, 12-24 octubre 1959). Tomo III. Buenos Aires: Librart, 276 pp.
- Pires de Prado L., R. M. Feitosa, S. Pinzon Triana, J. A. Munoz Gutierrez, G. X. Rousseau, R. Alves Silva, G. M. Siqueira, C. L. Caldas dos Santos, F. Veras Silva, T. Sanches Ranzani da Silva, A. Casadei-Ferreira, R. Rosa da Silva, and J. Andrade-Silva. 2019. An overview of the ant fauna (Hymenoptera: Formicidae) of the state of Maranhao, Brazil. Pap. Avulsos Zool. 59: e20195938.
- Ramos L. de S., C. G. S. Marinho, R. Zanetti, and J. H. C. Delabie. 2003. Impacto de iscas formicidas granuladas sobre a mirmecofauna não-alvo em eucaliptais segundo duas formas de aplicacação / Impact of formicid granulated baits on non-target ants in eucalyptus plantations according to two forms of application. Neotropical Entomology 32(2): 231-237.
- Reis P. C. J., W. D. Darocha, L. Falcao, T. J. Guerra, and F. S. Neves. 2013. Ant Fauna on Cecropia pachystachya Trécul (Urticaceae) Trees in an Atlantic Forest Area, Southeastern Brazil. Sociobiology 60(3): 222-228.
- Reynoso-Campos J. J., J. A. Rodriguez-Garza, and M. Vasquez-Bolanos. 2015. Hormigas (Hymenoptera: Formicidae) de la Isla Cozumel, Quintana Roo, Mexico (pp. 27-39). En: Castaño Meneses G., M. Vásquez-Bolaños, J. L. Navarrete-Heredia, G. A. Quiroz-Rocha e I. Alcalá-Martínez (Coords.). Avances de Formicidae de México. UNAM, Universiad de Guadalajara, Guadalajara, Jalisco.
- Ribas C. R., J. H. Schoereder, M. Pic, and S. M. Soares. 2003. Tree heterogeneity, resource availability, and larger scale processes regulating arboreal ant species richness. Austral Ecology 28(3): 305-314.
- Ribas C. R., and J. H. Shoereder. 2007. Ant communities, environmental characteristics and their implications for conservation in the Brazilian Pantanal. Biodivers. Conserv. 16: 1511-1520.
- Rico-Gray, V. 1993. Use of plant-derived food resources by ants in the dry tropical lowlands of coastal Veracruz, Mexico. Biotropica 25(3):301-315.
- Rico-Gray,V., J.G. Garcia-Franco, M. Palacios-Rios, C. Diaz-Castelazo, V. Parra-Tabla and J.A. Navarro. 1998. Geographical and Seasonal Variation in the Richness of Ant-Plant Interactions in Mexico. Biotropica 30(2):190-200.
- Rivas-Arancibia, S. P., H. Carrillo-Ruiz, A. Bonilla-Arce, D. M. Figueroa-Castro, and A. R. Andres-Hernandez. 2014. Effect of disturbance on the ant community in a semiarid region of central Mexico. Applied Ecology and Environmental Research 12: 703-716.
- Rodrigues C. A., M. da Silva Araujo, P. I. Decurcio Cabral, R. Lima, L. Bacci, and M. A. Oliveira. 2008. Arboreal ants community associated with the Pequizeiro (Caryocar brasiliense) in the Remnant of Cerrado Goiano. Pesquisa Florestal Brasiliera 57: 39-44.
- Rosa da Silva R. 1999. Formigas (Hymenoptera: Formicidae) do oeste de Santa Catarina: historico das coletas e lista atualizada das especies do Estado de Santa Catarina. Biotemas 12(2): 75-100.
- Rosa da Silva R., and B. Cortes Lopes. 1997. Ants (Hymenoptera: Formicidae) from Atlantic rainforest at Santa Catarina Island, Brazil: two years of sampling. Rev. Biol. Trop. 45(4): 1641-1648.
- Santschi F. 1916. Formicides sudaméricains nouveaux ou peu connus. Physis (Buenos Aires). 2: 365-399.
- Santschi F. 1922. Myrmicines, dolichodérines et autres formicides néotropiques. Bulletin de la Société Vaudoise des Sciences Naturelles 54: 345-378.
- Santschi F. 1925. Nouveaux Formicides brésiliens et autres. Bulletin et Annales de la Société Entomologique de Belgique 65: 221-247.
- Santschi F. 1931. Fourmis de Cuba et de Panama. Revista de Entomologia (Rio de Janeiro). 1: 265-282.
- Santschi F. 1934. Fourmis de Misiones et du Chaco argentin. Revista de la Sociedad Entomológica Argentina. 6: 23-34.
- Schmid V. S., S. Langner, J. Steiner, and A. Zillikens. 2014. Inflorescences of the Bromeliad Vriesea friburgensis as Nest Sites and Food Resources for Ants and Other Arthropods in Brazil. Psyche http://dx.doi.org/10.1155/2014/396095
- Schoereder J. H., T. G. Sobrinho, M. S. Madureira, C. R. Ribas, and P. S. Oliveira. 2010. The arboreal ant community visiting extrafloral nectaries in the Neotropical cerrado savanna. Terrestrial Arthropod Reviews 3: 3-27.
- Sendoya S. F., A. V. L. Freitas, and P. S. Oliveira. 2009. Egg-laying butterflies distinguish predaceous ants by sight. American Naturalist 174: 134-140.
- Silvestre R., C. R. F. Brandão, and R. R. Silva da 2003. Grupos funcionales de hormigas: el caso de los gremios del cerrado. Pp. 113-148 in: Fernández, F. (ed.) 2003. Introducción a las hormigas de la región Neotropical. Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, xxvi + 424 pp.
- Silvestre R., M. F. Demetrio, and J. H. C. Delabie. 2012. Community Structure of Leaf-Litter Ants in a Neotropical Dry Forest: A Biogeographic Approach to Explain Betadiversity. Psyche doi:10.1155/2012/306925
- Smith M. R. 1936. A list of the ants of Texas. Journal of the New York Entomological Society 44: 155-170.
- Sobrinho T. G., and J. H. Schoereder. 2007. Edge and shape effects on ant (Hymenoptera: Formicidae) species richness and composition in forest fragments. Biodivers Conserv 16: 14591470.
- Soto-Cardenas M. A., M. Vasquez-Bolanos, I. Chairez-Hernandez, M. M. Correa-Ramirez, and J. Lumar Reyes-Munoz. 2019. First record of Pseudomyrmex gracilis (Fabricius, 1804) (Hymenoptera: Formicidae: Pseudomyrmecinae) for Durango, Mexico. Dugesiana 26(1): 13-14.
- Suguituru S. S., D. R. de Souza, C. de Bortoli Munhae, R. Pacheco, and M. S. de Castro Morini. 2011. Diversidade e riqueza de formigas (Hymenoptera: Formicidae) em remanescentes de Mata Atlântica na Bacia Hidrográfica do Alto Tietê, SP. Biota Neotrop. 13(2): 141-152.
- Suguituru S. S., M. Santina de Castro Morini, R. M. Feitosa, and R. Rosa da Silva. 2015. Formigas do Alto Tiete. Canal 6 Editora 458 pages
- Suguituru S. S., R. Rosa Silva, D. R. de Souza, C. de Bortoli Munhae, and M. Santina de Castro Morini. Ant community richness and composition across a gradient from Eucalyptus plantations to secondary Atlantic Forest. Biota Neotrop. 11(1): 369-376.
- Ulyssea M. A., and C. R. F. Brandao. 2013. Ant species (Hymenoptera, Formicidae) from the seasonally dry tropical forest of northeastern Brazil: a compilation from field surveys in Bahia and literature records. Revista Brasileira de Entomologia 57(2): 217224.
- Ulysséa M. A., C. R. F. Brandão. 2013. Ant species (Hymenoptera, Formicidae) from the seasonally dry tropical forest of northeastern Brazil: a compilation from field surveys in Bahia and literature records. Revista Brasileira de Entomologia 57(2): 217-224.
- Valdes-Rodriguez S., P. Chacon de Ulloa, and I. Armbrecht. 2014. Soil ant species in Gorgona Island, Colombian Pacific. Rev. Biol. Trop. 62 (1): 265-276.
- Varela-Hernandez, F., M. Rocha-Ortega, R. W. Jones, and W. P. Mackay. 2016. Insectos: Hormigas (Formicidae) del estado de Queretaro, Mexico. Pages 397-404 in W. Jones., and V. Serrano-Cardenas, editors. Historia Natural de Queretaro. Universidad Autonoma de Queretaro, Mexico.
- Vargas R. P. 1964. Clave para identificar los Formicidae de la provincia de Chiclayo. Revista Peruana de Entomologia 7(1): 98-102.
- Vasconcelos H. L., J. M. S. Vilhena, W. E. Magnusson, and A. L. K. M. Albernaz. 2006. Long-term effects of forest fragmentation on Amazonian ant communities. Journal of Biogeography 33: 1348-1356.
- Vasconcelos, H.L. and J.M.S. Vilhena. 2006. Species turnover and vertical partitioning of ant assemblages in the Brazilian Amazon: A comparison of forests and savannas. Biotropica 38(1):100-106.
- Vasconcelos, H.L., J.M.S. Vilhena, W.E. Magnusson and A.L.K.M. Albernaz. 2006. Long-term effects of forest fragmentation on Amazonian ant communities. Journal of Biogeography 33:1348-1356
- Vasquez-Bolanos M. 2011. Checklist of the ants (Hymenoptera: Formicidae) from Mexico. Dugesiana 18(1): 95-133.
- Vasquez-Bolanos M. 2015. Taxonomia de Formicidae (Hymenoptera) para Mexico. Métodos en Ecología y Sistemática 10(1): 1. 53 pages.
- Vittar, F. 2008. Hormigas (Hymenoptera: Formicidae) de la Mesopotamia Argentina. INSUGEO Miscelania 17(2):447-466
- Vittar, F., and F. Cuezzo. "Hormigas (Hymenoptera: Formicidae) de la provincia de Santa Fe, Argentina." Revista de la Sociedad Entomológica Argentina (versión On-line ISSN 1851-7471) 67, no. 1-2 (2008).
- Vásquez-Bolaños M. 2011. Lista de especies de hormigas (Hymenoptera: Formicidae) para México. Dugesiana 18: 95-133
- Ward P. S. 1985. The Nearctic species of the genus Pseudomyrmex (Hymenoptera: Formicidae). Quaestiones Entomologicae 21: 209-246
- Ward P. S. 1990. The Ant Subfamily Pseudomyrmecinae (Hymenoptera: Formicidae): Generic Revision and Relationship to Other Formicids. Systematic Entomology 15: 449-489
- Ward P. S., and D. A. Downie. 2005. The ant subfamily Pseudomyrmecinae: phylogeny and evolution of big-eyed arboreal ants. Systematic Entomology 30: 310-335.
- Ward, P. S. 1989. Systematic Studies on Pseudomyrmecine Ants: Revision of the Pseudomyrmex Oculatus and P. Subtilissimus Species Groups with Taxonomic Comments on Other Species. Questiones Entomologicae 25: 393-468
- Ward, P.S. 1993. Systematic studies on Pseudomyrmex acacia-ants (Hymenoptera: Formicidae) Journal of Hymenoptera Research 2(1):117-168
- Wetterer J. K. 2010. Worldwide spread of the graceful twig ant, Pseudomyrmex gracilis (Hymenoptera: Formicidae). Florida Entomologist 93: 535-540.
- Wheeler W. M. 1916. Ants collected in British Guiana by the expedition of the American Museum of Natural History during 1911. Bulletin of the American Museum of Natural History 35: 1-14.
- Wheeler W. M. 1918. Ants collected in British Guiana by Mr. C. William Beebe. Journal of the New York Entomological Society 26: 23-28.
- Wheeler W. M. 1925. Neotropical ants in the collections of the Royal Museum of Stockholm. Arkiv för Zoologi 17A(8): 1-55.
- Wheeler W. M. 1942. Studies of Neotropical ant-plants and their ants. Bulletin of the Museum of Comparative Zoology 90: 1-262.
- Wheeler W. M., and I. W. Bailey. 1920. The feeding habits of pseudomyrmine and other ants. Transactions of the American Philosophical Society (2)22: 235-279.
- Wheeler W.M. 1935. Check list of the ants of Oceania. Occasional Papers of the Bernice Pauahi Bishop Museum 11(11):1-56.
- Wheeler, G.C. and J. Wheeler. 1985. A checklist of Texas ants. Prairie Naturalist 17:49-64.
- Wheeler, William Morton. 1934. Ants From The Islands Off The West Coast Of Lower California and Mexico. The Pan-Pacific Entomologist. 10(3):131-144.
- Whitcomb W. H., H. A. Denmark, W. F. Buren, and J. F. Carroll. 1972. Habits and present distribution in Florida of the exotic ant, Pseudomyrmex mexicanus (Hymenoptera: Formicidae). Florida Entomologist 55: 31-33.
- Wild, A. L. "A catalogue of the ants of Paraguay (Hymenoptera: Formicidae)." Zootaxa 1622 (2007): 1-55.
- Yanoviak S. P., and M. Kaspari. 2000. Community structure and the habitat templet: ants in the tropical forest canopy and litter. Oikos 89: 259-266.
- Zolessi L. C. de, Y. P. Abenante, and M. E. de Philippi. 1988. Lista sistematica de las especies de Formicidos del Uruguay. Comun. Zool. Mus. Hist. Nat. Montev. 11: 1-9.
- Zolessi L. C. de; Y. P. de Abenante, and M. E. Philippi. 1989. Catálogo sistemático de las especies de Formícidos del Uruguay (Hymenoptera: Formicidae). Montevideo: ORCYT Unesco, 40 + ix pp.
- da Silva de Oliveira A. B., and F. A. Schmidt. 2019. Ant assemblages of Brazil nut trees Bertholletia excelsa in forest and pasture habitats in the Southwestern Brazilian Amazon. Biodiversity and Conservation 28(2): 329-344.
- da Silva, R.R., C.R.F. Brandao, and R. Silvestre. 2004. Similarity Between Cerrado Localities in Central and Southeastern Brazil Based on the Dry Season Bait Visitors Ant Fauna. Studies on Neotropical Fauna and Environment 39(3):191-199.
- de Almeida Soares S., Y. R. Suarez, W. D. Fernandes, P. M. Soares Tenorio, J. H. C. Delabie, and W. F. Antonialli-Junior. 2013. Temporal variation in the composition of ant assemblages (Hymenoptera, Formicidae) on trees in the Pantanal floodplain, Mato Grosso do Sul, Brazil. Rev. Bras. entomol. 57: 84-90
- de Souza D. R., S. G. dos Santos, C. de B. Munhae, and M. S. de C. Morini. 2012. Diversity of Epigeal Ants (Hymenoptera: Formicidae) in Urban Areas of Alto Tietê. Sociobiology 59(3): 703-117.
- de Zolessi, L.C., Y.P. de Abenante and M.E. Philippi. 1987. Lista sistemática de las especies de formícidos del Uruguay. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo 11(165):1-9
- de Zolessi, L.C., Y.P. de Abenante and M.E. Phillipi. 1989. Catalago Systematico de las Especies de Formicidos del Uruguay (Hymenoptera: Formicidae). Oficina Regional de Ciencia y Technologia de la Unesco para America Latina y el Caribe- ORCYT. Montevideo, Uruguay


