Technomyrmex albipes
| Technomyrmex albipes | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Dolichoderinae |
| Genus: | Technomyrmex |
| Species group: | albipes |
| Species: | T. albipes |
| Binomial name | |
| Technomyrmex albipes (Smith, F., 1861) | |
| Synonyms | |
| |
Bolton (2007) - Technomyrmex albipes is an extremely successful tramp species that nests and forages both terrestrially and arboreally, and may enter houses. Workers may be found under stones, in and under fallen wood and in tree stumps, on the forest floor and in the leaf litter, on low vegetation and in twigs, on tree trunks and up into the canopy. A single record is known from internodes of the myrmecophyte Humboldtia laurifolia, from Gilimale in Sri Lanka. They also colonise more restricted spaces such as plant spathes and rot holes in wood and they attend a wide range of homopterous insects for honeydew. Although usually regarded as a pest or invasive species, for instance Sulaiman (1997) observes that it tends the mealybug responsible for pineapple wilt disease in Sri Lanka, albipes also has value as an important predator of the eggs of destructive insect species on coconuts in Sri Lanka (Way, Cammell, et al. (1989).
| At a Glance | • Highly invasive • Supercolonies |
Photo Gallery
Identification
Bolton (2007) - A member of the T. albipes complex in the Technomyrmex albipes group. Colour of the mandibles and antennae is variable in albipes. The mandible varies from the same dark colour as the head capsule to yellowish, with all intermediate shades present. The scape is usually about the same shade as the head capsule; the funiculus may be the same colour or entirely lighter, or the apical few segments alone may be lighter. These variations are gradient and are not considered taxonomically significant.
In the past many samples of small, darkly coloured Technomyrmex in which the gaster is setose and the tarsi are white or yellow have been misidentified as T. albipes, but in particular albipes has been confused with Technomyrmex vitiensis, Technomyrmex difficilis, Technomyrmex pallipes, Technomyrmex brunneus, Technomyrmex jocosus and Technomyrmex moerens. The first three of these are also common tramp species and vitiensis was for a while considered ajunior synonym of albipes. T. vitiensis, along with its Afrotropical relative moerens, closely resembles albipes, but both have larger eyes, longer scapes and a longer promesonotum than to albipes. Also, the mesonotal profile in albipes workers is evenly curved, whereas in both other species there tends to be an angle or step in the outline, resulting in differently sloped dorsal and declivitous mesonotal faces; see also the notes under vitiensis. As for difficilis, the presence in that species of a pair of setae on the dorsal head behind the level of the posterior margin of the eye easily distinguishes them. However, workers of difficilis in which the head is abraded are difficult to distinguish from albipes; see notes under difficilis. T. pallipes should not be confused with albipes as it always has two pairs of short stubbly setae on the dorsal head behind the level of the posterior margin of the eye. T. brunneus has mandibular grooves not seen in albipes, as well as other different characters, and jocosus has a very different arrangement of setae: neither should be confused with albipes.
Keys including this Species
- Key to Australian Technomyrmex Species
- Key to Micronesian Ants
- Key to New World Technomyrmex species
- Key to Arabian Technomyrmex
- Key to Afrotropical and west Palaearctic Technomyrmex
- Key to Malagasy Technomyrmex
- Key to Oriental Technomyrmex
- Key to Technomyrmex of Taiwan
- Key to Technomyrmex of Thailand and Laos
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 32.628611° to -9.233°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Afrotropical Region: Cameroun, Ivory Coast, Mozambique, South Africa, Sudan, United Arab Emirates, United Republic of Tanzania, Yemen.
Australasian Region: Australia.
Indo-Australian Region: Borneo, Cook Islands, Fiji, Guam, Hawaii, Indonesia (type locality), Krakatau Islands, Malaysia, Marshall Islands, Micronesia (Federated States of), New Guinea, Niue, Philippines, Pitcairn, Singapore, Solomon Islands, Sulawesi, Tokelau, Wallis and Futuna Islands.
Malagasy Region: Madagascar, Mauritius, Réunion, Seychelles.
Neotropical Region: Cayman Islands.
Oriental Region: Bangladesh, Cambodia, India, Laos, Myanmar, Sri Lanka, Taiwan, Thailand, Vietnam.
Palaearctic Region: China, Democratic Peoples Republic of Korea, Denmark, Japan, Republic of Korea.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
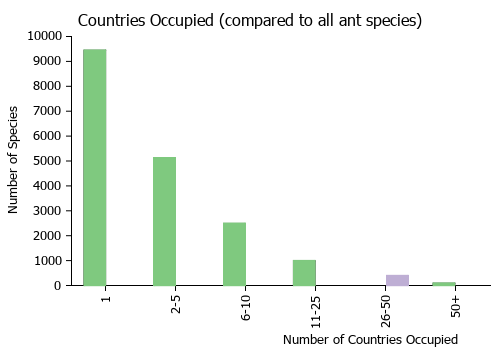
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
A member of the T. albipes complex in the Technomyrmex albipes group.
Sharaf et al. (2018): Technomyrmex albipes nests and forages in and beneath fallen wood and rocks, in tree trunks, in leaf litter, in twigs, on the forest floor, on low vegetation, and into the canopy (Bolton 2007). The species is known to feed on honeydew of a broad range of sap sucking attended hemipterans including the mealybug vectors of pineapple wilt disease (Sulaiman 1997).
Yamane et al. (2018) - Although Bolton (2007) resolved most taxonomic confusion in this species group, there still remain some unsolved problems. For example, very restricted information is available about the nesting habits of T. albipes in spite of its common occurrence as a tramp. Bolton enumerated a few instances for the nesting site of this species, namely, internodes of a myrmecophyte, plant spathes and rot holes in wood. However, some carton nests on tree leaves have been found in Borneo and Malay Peninsula for ‘this species’ as well as nests in restricted spaces such as rotting wood, dead twigs etc. (Yamane and Tsuji, personal obs.). This suggests that T. albipes (sensu Bolton) may actually be a complex of sibling species. More information on nesting site and DNA data from various localities are needed to resolve this problem.
Association with Other Organisms
 Explore: Show all Associate data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Associate data or Search these data. See also a list of all data tables or learn how data is managed.
- This species is a mutualist for the aphid Aphis clerodendri (a trophobiont) (Carver et al., 2003; Saddiqui et al., 2019).
- This species is a mutualist for the aphid Aphis gossypii (a trophobiont) (Idechiil et al., 2007; Lokeshwari et al., 2015; Saddiqui et al., 2019).
- This species is a mutualist for the aphid Hysteroneura setariae (a trophobiont) (Idechiil et al., 2007; Saddiqui et al., 2019).
- This species is a mutualist for the aphid Pentalonia nigronervosa (a trophobiont) (Idechiil et al., 2007; Saddiqui et al., 2019).
- This species is a host for the trematode Eurytrema pancreaticum (a parasitoid) (Quevillon, 2018) (encounter mode primary; indirect transmission; transmission outside nest).
- This species is a prey for the syrphid fly Bardistopus papuanum (a predator) (Quevillon, 2018).
Flight Period
| X | |||||||||||
| Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec |
- Check details at Worldwide Ant Nuptial Flights Data, AntNupTracker and AntKeeping.
 Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Castes
Bolton (2007) - Distinct morphological intercastes between workers and queens, such as are relatively common in Technomyrmex vitiensis, Technomyrmex moerens, Technomyrmex brunneus, Technomyrmex pallipes and other species of the albipes group, where ocelli are present and the mesoscutellum and metanotum are developed as separate or semi-separate sclerites in otherwise worker-like forms, appear absent from material examined of genuine albipes. Only a few basically very worker-like forms with slightly posteriorly expanded mesonota have been seen. This observation may be merely a sampling artifact indicating that most albipes material examined has been of foragers, not from nest samples, as in other species where intercastes have been confirmed they are confined to reproductive behaviour within colonies and do not undertake foraging activities. However, 24 samples provided in alcohol by Martin Pfeiffer (Universitat Ulm), collected at Banting, West Malaysia and Tawau, Sabah, East Malaysia, failed to produce a single obvious intercaste, although many of the samples included brood and fragments of nest material; one had brood and ergatoid males in it and another contained a dealate queen.
Worker
  
| |
| . | Owned by Museum of Comparative Zoology. |
Images from AntWeb
   
| |
| Worker. Specimen code casent0104343. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0188900. Photographer Erin Prado, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0125198. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0055965. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0101467. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MNHN, Paris, France. |
    
| |
| Syntype of Technomyrmex albipes. Worker. Specimen code casent0102952. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by OUM, Oxford, UK. |
    
| |
| Worker. Specimen code casent0103002. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by OUM, Oxford, UK. |
Queen
Images from AntWeb
   
| |
| Queen (alate/dealate). Specimen code casent0071195. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Queen (alate/dealate). Specimen code casent0125208. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Queen (alate/dealate). Specimen code casent0060419. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
    
| |
| Queen (alate/dealate). Specimen code casent0125325. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
Male
Images from AntWeb
    
| |
| Male (alate). Specimen code casent0055727. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- detorquens. Formica detorquens Walker, 1859: 372 (q.) SRI LANKA. Combination in Technomyrmex: Donisthorpe, 1932b: 575. Senior synonym of forticulus: Donisthorpe, 1932b: 575. Nomen oblitum, synonym of albipes: Bolton, 2007a: 68. See also: Wilson & Taylor, 1967: 84.
- forticulus. Crematogaster forticulus Walker, 1859: 372 (q., not w.) SRI LANKA. Junior synonym of detorquens: Donisthorpe, 1932b: 576; nomen oblitum, synonym of albipes: Bolton, 2007a: 68.
- albipes. Formica (Tapinoma) albipes Smith, F. 1861b: 38 (w.) INDONESIA (Sulawesi). Forel, 1891b: 98 (q.); Forel, 1908a: 21 (ergatoid m.); Karavaiev, 1926d: 441 (m.); Wheeler, G.C. & Wheeler, J. 1951: 205 (l.); Crozier, 1969: 245 (k.). Combination in Tapinoma: Mayr, 1863: 455; in Technomyrmex: Emery, 1888d: 392. Senior synonym of nigrum: Mayr, 1872: 147; Mayr, 1876: 83; of albitarse: Emery, 1893f: 249; of bruneipes, detorquens, forticulus, wedda: Bolton, 2007a: 68.
- nigrum. Tapinoma nigrum Mayr, 1862: 703 (w.) SRI LANKA. Junior synonym of albipes: Mayr, 1872: 147; Mayr, 1876: 83; Bolton, 2007a: 68.
- albitarse. Tapinoma albitarse Motschoulsky, 1863: 14 (w.q.) SRI LANKA. Junior synonym of albipes: Emery, 1893f: 249.
- bruneipes. Technomyrmex albipes var. bruneipes Forel, 1895e: 466 (w.q.m.) INDIA. Raised to species: Collingwood & Agosti, 1996: 361. Junior synonym of albipes: Bolton, 2007a: 68.
- wedda. Technomyrmex albipes r. wedda Forel, 1913e: 663 (w.q.) SRI LANKA. Junior synonym of albipes: Bolton, 2007a: 68.
Type Material
Bolton (2007) - Syntype workers, Indonesia: Sulawesi, “Tond” (= Tondano) (A.R. Wallace) (Oxford University Museum of Natural History) [examined].
- Formica (Tapinoma) albipes: Syntype, 2 workers, Tondano, Sulawesi, Indonesia, Oxford University Museum of Natural History.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Bolton (2007) - TL 2.4 - 2.9, HL 0.56 - 0.63, HW 0.52 - 0.58, SL 0.48 - 0.58, PW 0.35 - 0.42, WL 0.66 - 0.78 (50 measured). Indices: CI 87 - 95, SI 91 - 102, or 24 - 27, EPI 70 - 88, DTI 110 - 124.
Frontal carina with 2 (very rarely 3) setae: in profile the anteriormost seta at the torulus, the posteriormost seta approximately at the level of the anterior margin of the eye. Dorsum of head posterior to this entirely lacks setae. With head in full-face view the anterior clypeal margin with a very weak, shallow median indentation; sides of head shallowly convex and the posterior margin with a small shallow indentation medially. Eyes located in front of midlength, EPI < 90; outer margin of eye usually just fails to break, or sometimes just touches, the outline of the side. With mesosoma in profile the mesonotal outline is evenly curved, without a distinct step or angle in the outline that defines conspicuous dorsal and declivitous faces. Number of setal pairs on mesosoma: pronotum 1 - 3; mesonotum 0 1 (usually 0); propodeal dorsum 0; lateral margins of propodeal declivity 1 - 2 (very rarely 3), usually with one just above the spiracle, another higher up. With the propodeum in profile its dorsum and declivity meet in a short, blunt, but very narrowly rounded curve, not a distinct sharp angle. Straight-line length of propodeal dorsum in profile is less than depth of declivity to spiracle. Gastral tergites 1 - 4 each with numerous setae, distributed everywhere on the sclerites; maximum length of setae on first gastral tergite is usually slightly less than the maximum diameter of the eye but sometimes the two are subequal. Head, mesosoma, petiole and gaster blackish brown to black; in profile the gaster is often slightly lighter than the mesosoma. Coxae, femora and tibiae uniformly blackish brown to black, same colour as the mesosoma or gaster; never with strongly contrasting lighter coxae. Tarsi of middle and hind legs white to dull yellowish, paler than the tibiae.
Karyotype
- See additional details at the Ant Chromosome Database.
 Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
- n = 9 (Spain) (Lorite et al., 2012) (as Tapinoma nigerum).
- n = 9, 2n = 18, karyotype = 16M+2A (Australia) (Crozier, 1968a; Ant Chromosome Database) (seven medium-size chromosomes metacentric and two small chromosomes one metacentric and one acrocentric.).
- 2n = 16 (Australia) (Imai et al., 1977).
- 2n = 16, karyotype = 16M (India) (Imai et al., 1984).
Determination Clarifications
The correct identities of some species referred to as albipes in relatively recent publications can be ascertained with some degree of certainty, but those of earlier studies (e.g. Starcke, 1940), and even some more recent ones (e.g. Brophy, 1994) in the absence of the relevant specimens, must remain equivocal. Material mentioned as introductions in British hothouses by Donisthorpe (1927) consists of series of albipes, Technomyrmex vitiensis and Technomyrmex pallipes. References to albipes in recent Chinese and Japanese works such as Imai, Hikara, Kondoh, et al. (2003), Zhou (2001), Wu & Wang (1995) and the intensive studies by Tsuji, Furukawa, et al. (1991), Yamauchi, Furukawa, et al. (1991), Tsuji & Yamauchi (1994) and Ogata. Murai, et al. (1996), are all Technomyrmex brunneus. Also referable to brunneus is Radchenko’s (2005) record of “albipes” from North Korea. The discussions of albipes in Bourke & Franks (I 995}, based on these Japanese publications, should also be referred to brunneus. Recent Australian references to albipes in Shattuck (1999), and Andersen's (2000) unnamed fig. 29 are probably all Technomyrmex difficilis and this is also the species which has recently colonised Florida so successfully (Deyrup, 1991; Vail, Davis, et al., 1994; DeyruJl, Davis & Cover, 2000; Warner, 2003). The species referred to as albipes by Brown (l958) in New Zealand is probably Technomyrmex jocosus. The short discussion of albipes by Shattuck (1992b) appears to be a fusion of several species; it certainly includes vitiensis as that is the only species known to occur in conservatories in Golden Gate Park, California. The vast amount of Pacific Islands material listed in Wilson & Taylor (1967) certainly includes both albipes and vitiensis, and probably also difficilis; a critical re-assessment of the entire collection would be needed to resolve the identities. Terron (1972) presented some notes on alate and ergatoid males of a species close to albipes, which may refer to Technomyrmex moerens or perhaps even pallipes.
References
- Baer, B. 2011. The copulation biology of ants (Hymenoptera: Formicidae). Myrmecological News 14: 55-68.
- Bissessur, P., Baider, C., Florens, F.B.V. 2020. Infestation by pollination-disrupting alien ants varies temporally and spatially and is worsened by alien plant invasion. Biological Invasions (doi:10.1007/s10530-020-02272-y).
- Blard, F., Dorow, W.-H.-O., Delabie, J. H. C. 2003. Les Fourmis de l’île de la Réunion (Hymenoptera, Formicidae). Bulletin de La Société Entomologique de France, 108(2), 127–137 (doi:10.3406/bsef.2003.16939).
- Bolton, B. 2007b. Taxonomy of the dolichoderine ant genus Technomyrmex Mayr (Hymenoptera: Formicidae) based on the worker caste. Contributions of the American Entomological Institute. 35(1): 1-149.
- Borowiec, L. 2014. Catalogue of ants of Europe, the Mediterranean Basin and adjacent regions (Hymenoptera: Formicidae). Genus (Wroclaw) 25(1-2): 1-340.
- Cantone S. 2017. Winged Ants, The Male, Dichotomous key to genera of winged male ants in the World, Behavioral ecology of mating flight (self-published).
- Cantone S. 2018. Winged Ants, The queen. Dichotomous key to genera of winged female ants in the World. The Wings of Ants: morphological and systematic relationships (self-published).
- Collingwood, C.A., Agosti, D., Sharaf, M.R., van Harten, A. 2011. Order Hymenoptera, family Formicidae. Arthropod fauna of the UAE 4: 405-474.
- Crozier, R. H. 1969a [1968]. Cytotaxonomic studies on some Australian dolichoderine ants (Hymenoptera: Formicidae). Caryologia 21: 241-259 (page 245, karyotype described)
- Davis, T. 2009. The ants of South Carolina (thesis, Clemson University).
- Dekoninck, W., Wauters, N., Delsinne, T. 2019. Capitulo 35. Hormigas invasoras en Colombia. Hormigas de Colombia.
- Dias, R.K.S., Kosgamage, K.R.K.A. 2013. Occurrence and species diversity of ground-dwelling worker ants (Family: Formicidae) in selected lands in the dry zone of Sri Lanka. Journal of Science of the University of Kelaniya Sri Lanka 7: 55-72 (doi:10.4038/josuk.v7i0.6233).
- Dias, R.K.S., Perera, A.P.S. 2016. Species richness of arboreal ant assemblages (Hymenoptera, Formicidae) and frequency of Oecophylla smaragdina (Fabricius) occurrence in a wet zone cashew (Anacardium occidentale L.) field in Sri Lanka. Journal of Science of the University of Kelaniya Sri Lanka 11, 1-10 (doi:10.4038/josuk.v11i1.7996).
- Dias, R.K.S., Rajapaksa, R.P.K.C. 2017. Geographic records of subfamilies, genera and species of ants (Hymenoptera: Formicidae) in the four climatic zones of Sri Lanka: A review. Journal of Science of the University of Kelaniya Sri Lanka 11, 23-45. (doi:10.4038/josuk.v11i2.7999).
- Donisthorpe, H. 1932b. On the identity of some ants from Ceylon described by F. Walker. Ann. Mag. Nat. Hist. 10(9): 574-576 (page 575, Junior synonym of detorguens)
- Emery, C. 1888d. Über den sogenannten Kaumagen einiger Ameisen. Z. Wiss. Zool. 46: 378-412 (page 392, Combination in Technomyrmex)
- Emery, C. 1893h. Voyage de M. E. Simon à l'île de Ceylan (janvier-février 1892). Formicides. Ann. Soc. Entomol. Fr. 62: 239-258 (page 249, Senior synonym of albitarse)
- Forel, A. 1891c. Les Formicides. [part]. In: Grandidier, A. Histoire physique, naturelle, et politique de Madagascar. Volume XX. Histoire naturelle des Hyménoptères. Deuxième partie (28e fascicule). Paris: Hachette et Cie, v + 237 pp. (page 98, queen described)
- Forel, A. 1908b. Fourmis de Ceylan et d'Égypte récoltées par le Prof. E. Bugnion. Lasius carniolicus. Fourmis de Kerguelen. Pseudandrie? Strongylognathus testaceus. Bull. Soc. Vaudoise Sci. Nat. 44: 1-22 (page 21, ergatoid male described)
- Hasin, S., Tasen, W. 2020. Ant community composition in urban areas of Bangkok, Thailand. Agriculture and Natural Resources 54: 507-514 (doi:10.34044/j.anres.2020.54.5.07).
- Hazarika, H.N., Khanikor, B. 2021. Integration of morphological and molecular taxonomic characters for identification of Odontoponera denticulata (Hymenoptera: Formicidae:Ponerinae) with the description of the antennal sensilla. Zoologischer Anzeiger 293: 89-100 (doi:10.1016/j.jcz.2021.05.009).
- Heterick, B.E. 2022. A guide to the ants of Western Australia. Part II: Distribution and biology. Records of the Western Australian Museum, supplement 86: 247-510 (doi:10.18195/issn.0313-122x.86.2022.247-510).
- Hoey-Chamberlain, R., Rust, M.K. 2014. Food and bait preferences of Liometopum occidentale (Hymenoptera: Formicidae). Journal of Entomological Science 49(1): 30-43.
- Hoey-Chamberlain, R.V. 2012. Food preference, survivorship, and intraspecific interactions of Velvety Tree Ants. M.S. thesis, University of California, Riverside.
- Hosoishi, S., Rahman, M. M., Heng, S. 2022. Exotic ants (Hymenoptera: Formicidae) of Cambodia. Far Eastern Entomologist 460, 15–24 (doi:10.25221/fee.460.3).
- Imai, H.T., Kihara, A., Kondoh, M., Kubota, M., Kuribayashi, S., Ogata, K., Onoyama, K., Taylor, R.W., Terayama, M., Yoshimura, M., Ugawa, Y. 2003. Ants of Japan. 224 pp, Gakken, Japan.
- Iwata, K., Eguchi, K., Yamane, S. 2005. A case study on urban ant fauna of southern Kyusyu, Japan, with notes on a new monitoring protocol (Insecta, Hymenoptera, Formicidae). Journal of Asia-Pacific Entomology 8, 263-272.
- Jaitrong, W., Yamane, S. 2024. A revision of the ants Technomyrmex Mayr, 1872 (Hymenoptera: Formicidae: Dolichoderinae) in Thailand and Laos. Natural History Bulletin of the Siam Society 66 (1): 29–118.
- Karavaiev, V. 1926d. Ameisen aus dem Indo-Australischen Gebiet. Treubia 8: 413-445 (page 441, male described)
- Khachonpisitsak, S., Yamane, S., Sriwichai, P., Jaitrong, W. 2020. An updated checklist of the ants of Thailand (Hymenoptera, Formicidae). ZooKeys 998, 1–182 (doi:10.3897/zookeys.998.54902).
- Kreider, J.J., Chen, T.W., Hartke, T.R., Buchori, D., Hidayat, P., Nazarreta, R., Scheu, S., Drescher, J. 2021. Rainforest conversion to monocultures favors generalist ants with large colonies. Ecosphere 12 (doi:10.1002/ecs2.3717).
- Lee, C.-C., Weng, Y.-M., Lai, L.-C., Suarez, A.V., Wu, W.-J., Lin, C.-C., Yang, C.-C.S. 2020. Analysis of recent interception records reveals frequent transport of arboreal ants and potential predictors for ant invasion in Taiwan. Insects 11, 356 (doi:10.3390/INSECTS11060356).
- Liu, C., Fischer, G., Hita Garcia, F., Yamane, S., Liu, Q., Peng, Y.Q., Economo, E.P., Guénard, B., Pierce, N.E. 2020. Ants of the Hengduan Mountains: a new altitudinal survey and updated checklist for Yunnan Province highlight an understudied insect biodiversity hotspot. ZooKeys 978, 1–171 (doi:10.3897/zookeys.978.55767).
- Mamet, R. 1954. The ants (Hymenoptera Formicidae) of the Mascarene Islands. Mauritius Inst. Bull. 3: 249-259 (see also)
- Mayr, G. 1863a. Formicidarum index synonymicus. Verh. K-K. Zool.-Bot. Ges. Wien 13: 385-460 (page 455, Combination in Tapinoma)
- Mayr, G. 1872. Formicidae Borneenses collectae a J. Doria et O. Beccari in territorio Sarawak annis 1865-1867. Ann. Mus. Civ. Stor. Nat. 2: 133-155 (page 147, Senior synonym of nigrum)
- Mayr, G. 1876. Die australischen Formiciden. J. Mus. Godeffroy 12: 56-115 (page 83, Senior synonym of nigrum)
- Meurville, M.-P., LeBoeuf, A.C. 2021. Trophallaxis: the functions and evolution of social fluid exchange in ant colonies (Hymenoptera: Formicidae). Myrmecological News 31: 1-30 (doi:10.25849/MYRMECOL.NEWS_031:001).
- Mukherjee, A., Roy, U.S. 2021. Role of physicochemical parameters on diversity and abundance of ground–dwelling, diurnal ant species of Durgapur Government College Campus, West Bengal, India. Munis Entomology and Zoology Journal 16(1): 366-380.
- Nazarreta, R., Hartke, T.R., Hidayat, P., Scheu, S., Buchori, D., Drescher, J. 2020. Rainforest conversion to smallholder plantations of rubber or oil palm leads to species loss and community shifts in canopy ants (Hymenoptera: Formicidae). Myrmecological news 30, 175-186 (doi:10.25849/MYRMECOL.NEWS_030:175).
- Pech, P., Bezděk, A. 2016. Ergatomorph wingless males in Technomyrmex vitiensis Mann, 1921 (Hymenoptera: Formicidae). Journal of Hymenoptera Research 53, 25–34 (doi:10.3897/jhr.53.8904).
- Perrichot, V., Boudinot, B.E., Engel, M.S., Xu, C., Bojarski, B., Szwedo, J. 2022. Ants (Hymenoptera: Formicidae) from Miocene Ethiopian amber: filling gaps in the geological record of African terrestrial biota, Zoological Journal of the Linnean Society, 2022, zlac053 (doi:10.1093/zoolinnean/zlac053).
- Radchenko, A.G., Fisher, B.L., Esteves, F.A., Martynova, E.V., Bazhenova, T.N., Lasarenko, S.N. 2023. Ant type specimens (Hymenoptera, Formicidae) in the collection of Volodymyr Opanasovych Karawajew. Communication 1. Dorylinae, Poneromorpha and Pseudomyrmecinae. Zootaxa, 5244(1), 1–32 (doi:10.11646/zootaxa.5244.1.1).
- Rosas-Mejía, M., Guénard, B., Aguilar-Méndez, M. J., Ghilardi, A., Vásquez-Bolaños, M., Economo, E. P., Janda, M. 2021. Alien ants (Hymenoptera: Formicidae) in Mexico: the first database of records. Biological Invasions 23(6), 1669–1680 (doi:10.1007/s10530-020-02423-1).
- Sharaf, M. R.; Collingwood, C. A. and Aldawood, S. A. 2011. Technomyrmex montaseri sp. n., a new ant species of the T. gibbosus-group from Oman (Hymenoptera, Formicidae) with a key to the Technomyrmex species of the Arabian Peninsula. ZooKeys. 108:11-19.
- Sharaf, M.R., Al Dhafer, H.M., Aldawood, A.S. 2018. Review of the ant genus Technomyrmex Mayr, 1872 in the Arabian Peninsula (Hymenoptera, Formicidae). ZooKeys 780: 35–59 (DOI10.3897/zookeys.780.26272).
- Shattuck, S. O. 1994. Taxonomic catalog of the ant subfamilies Aneuretinae and Dolichoderinae (Hymenoptera: Formicidae). Univ. Calif. Publ. Entomol. 112:i-xix, 1-241. (page 157, see also)
- Siddiqui, J. A., Li, J., Zou, X., Bodlah, I., Huang, X. 2019. Meta-analysis of the global diversity and spatial patterns of aphid-ant mutualistic relationships. Applied Ecology and Environmental Research 17: 5471-5524 (doi:10.15666/aeer/1703_54715524).
- Siddiqui, J.A., Bamisile, B.S., Khan, M.M., Islam, W., Hafeez, M., Bodlah, I., Xu, Y. 2021. Impact of invasive ant species on native fauna across similar habitats under global environmental changes. Environmental Science and Pollution Research 28(39), 54362–54382 (doi:10.1007/s11356-021-15961-5).
- Smith, F. 1861b. Catalogue of hymenopterous insects collected by Mr. A. R. Wallace in the islands of Ceram, Celebes, Ternate, and Gilolo. [part]. J. Proc. Linn. Soc. Lond. Zool. 6: 36-48 (page 38, worker described)
- Terayama, M., Sunamura, E., Fujimaki, R., Ono, T., Eguchi, K. 2021. A Surprisingly Non-attractiveness of Commercial Poison Baits to Newly Established Population of White-Footed Ant, Technomyrmex brunneus (Hymenoptera: Formicidae), in a Remote Island of Japan. Sociobiology 68, 5898 (doi:10.13102/sociobiology.v68i1.5898).
- Trible, W., Kronauer, D.J.C. 2017. Caste development and evolution in ants: it's all about size. Journal of Experimental Biology 220, 53–62 (doi:10.1242/jeb.145292).
- Tseng, S.-P. 2020. Evolutionary history of a global invasive ant, Paratrechina longicornis (Dissertation_全文 ). Ph.D. thesis, Kyoto University.
- Tseng, S.-P., Hsu, P.-W., Lee, C.-C., Wetterer, J.K., Hugel, S., Wu, L.-H., Lee, C.-Y., Yoshimura, T., Yang, C.-C.S. 2020. Evidence for common horizontal transmission of Wolbachia among ants and ant crickets: Kleptoparasitism added to the list. Microorganisms 8, 805. (doi:10.3390/MICROORGANISMS8060805).
- Tsuji, K.; Furukawa, T.; Kinomura, K.; Takamine, H.; Yamauchi, K. 1991. The caste system of the dolichoderine ant Technomyrmex albipes (Hymenoptera: Formicidae): morphological description of queens, workers and reproductively active intercastes. Insectes Sociaux 38: 413-422. (= T. brunneus)
- Wang, W.Y., Soh, E.J.Y., Yong, G.W.J., Wong, M.K.L., Benoit Guénard, Economo, E.P., Yamane, S. 2022. Remarkable diversity in a little red dot: a comprehensive checklist of known ant species in Singapore (Hymenoptera: Formicidae) with notes on ecology and taxonomy. Asian Myrmecology 15: e015006 (doi:10.20362/am.015006).
- Wheeler, G. C.; Wheeler, J. 1951. The ant larvae of the subfamily Dolichoderinae. Proc. Entomol. Soc. Wash. 53: 169-210 (page 205, larva described)
- Wilson, E. O.; Taylor, R. W. 1967b. The ants of Polynesia (Hymenoptera: Formicidae). Pac. Insects Monogr. 14: 1-109 (page 82, Senior synonym of rufescens and vitiensis, Revived from synonymy)
- Wu, J. & Wang, C. 1992. Formicidae (pp. 1301-1320). In Peng, J. et al. Iconography of Forest Insects in Hunan, China. Forest Bureau of Hunan Province: 1473 pp. Hunan Scientific and Technical Publishing House.
- Wu, J. & Wang, C. 1992. Formicidae (pp. 1301-1320). In Peng, J. et al. Iconography of Forest Insects in Hunan, China. Forest Bureau of Hunan Province: 1473 pp. Hunan Scientific and Technical Publishing House.
- Yamane, S., Leong, C.-M., Lin, C.C. 2018. Taiwanese species of the ant genus Technomyrmex (Formicidae: Dolichoderinae). Zootaxa 4410: 35–56 (DOI 10.11646/zootaxa.4410.1.2).
- Yamane, S., Tanaka, H.O., Hasimoto, Y., Ohashi, M., Meleng, P., Itioka, T. 2021. A list of ants from Lambir Hills National Park and its vicinity, with their biological information: Part II. Subfamilies Leptanillinae, Proceratiinae, Amblyoponinae, Ponerinae, Dorylinae, Dolichoderinae, Ectatomminae and Formicinae. Contributions from the Biological Laboratory, Kyoto University 31, 87–157.
References based on Global Ant Biodiversity Informatics
- Abe T., S. Yamane, and K. Onoyama. Ants collected on the Krakatau Islands 100 years after the great eruptions. Biogeography 14: 65-75.
- Alcantara M. J., S. Modi, T. C. Ling, J. Monkai, H. Xu, S. Huang, and A. Nakamura. 2019. Differences in geographic distribution of ant species (Hymenoptera: Formicidae) between forests and rubber plantations: a case study in Xishuangbanna, China, and a global meta-analysis. Myrmecological News 29: 135-145.
- Amarasinghe H. E. 2010. Species composition and nesting habits of ants in a hill-country home garden in Sri Lanka. Asian Myrmecology 3: 9-20.
- André E. 1892. Voyage de M. Chaper à Bornéo. Catalogue des fourmis et description des espèces nouvelles. Mém. Soc. Zool. Fr. 5: 46-55.
- Asfiya W., R. Ubaidillah, and Sk. Yamane. 2008. Ants (Hymenoptera: Formicidae) of the Krakataus, and Sebesi and Sebuku islands. Treubia 36: 1-9.
- Ashmead W. H. 1904. A list of the Hymenoptera of the Philippine Islands, with descriptions of new species. J. N. Y. Entomol. Soc. 12:1-22.
- Azuma, S. and M. Kinjo. 1987. Family Formicidae, In Checklist of the insects of Okinawa. The Biological Society of Okinawa, Nishihara. Pages 310-312.
- Baroni Urbani C. 1977. Katalog der Typen von Formicidae (Hymenoptera) der Sammlung des Naturhistorischen Museums Basel (2. Teil). Mitt. Entomol. Ges. Basel (n.s.) 27: 61-102.
- Bharti H., Y. P. Sharma, M. Bharti, and M. Pfeiffer. 2013. Ant species richness, endemicity and functional groups, along an elevational gradient in the Himalayas. Asian Myrmecology 5: 79-101.
- Bolton B. 2007. Taxonomy of the dolichoderine ant genus Technomyrmex Mayr (Hymenoptera: Formicidae) based on the worker caste. Contributions of the American Entomological Institute 35(1): 1-150.
- Bolton, B. "Taxonomy of the dolichoderine ant genus Technomyrmex Mayr (Hymenoptera: Formicidae) based on the worker caste." Contributions of the American Entomological Institute 35, no. 1 (2007): 1-149.
- CSIRO Collection
- Chapman, J. W., and Capco, S. R. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monogr. Inst. Sci. Technol. Manila 1: 1-327
- Chen P., Y. Su, S. S. Rao, Y. F. Long, and C. H. Du. 2012. Study on the ant diversity in different Illicium verum stands. Journal of West China Forestry Science 41(1): 60-68.
- Chen Y. Q., Q. Li, Y. L. Chen, Z. X. Lu, X. Y. Zhou. 2011. Ant diversity and bio-indicators in land management of lac insect agroecosystem in Southwestern China. Biodivers. Conserv. 20: 3017-3038.
- Chen Y., C.-W. Luo, H. W Li, Z. H. Xu, Y. J. Liu, and S. J. Zhao. 2011. The investigation of soil ant resources on the West slope of Mt Ailao. Hubei Agricultural Sciences 50(7): 1356-1359.
- Cheng D., Z. Chen, and S. Zhou. 2015. An analysis on the ant fauna of Jinzhongshan Nature Reserve in Gunagxi, China. Journal of Guangxi Normal University: Natural Science Edition 33(3): 129.137.
- Dad J. M., S. A. Akbar, H. Bharti, and A. A. Wachkoo. 2019. Community structure and ant species diversity across select sites ofWestern Ghats, India. Acta Ecologica Sinica 39: 219–228.
- Davidson D. W., S. C. Cook, R. R. Snelling and T. H. Chua. 2003. Explaining the Abundance of Ants in Lowland Tropical Rainforest Canopies. Science 300: 969-972.
- Dhote J. 2012. Ant (Hymenoptera: Formicidae) records from Shri Shivaji science college campus, Amravati, India. Applied Research and Development Institute Journal 3(12): 114-117.
- Dias R. K. S. 2006. Current taxonomic status of ants (Hymenoptera: Formicidae) in Sri Lanka. The Fauna of Sri Lanka: 43-52. Bambaradeniya, C.N.B. (Editor), 2006. Fauna of Sri Lanka: Status of Taxonomy, Research and Conservation. The World Conservation Union, Colombo, Sri Lanka & Government of Sri Lanka. viii + 308pp.
- Dias R. K. S. 2013. Diversity and importance of soil-dweeling ants. Proceedings of the National Symposium on Soil Biodiversity, chapt 4, pp 19-22.
- Dias R. K. S., H. P. G. R. C. Ruchirani, K. R. K. A. Kosgamage, and H. A. W. S. Peiris. 2013. Frequency of nest occurrence and nest density of Aneuretus simoni Emery (Sri Lankan Relict Ant) and other ant fauna in an abandoned rubber plantation (Kirikanda Forest) in southwest Sri Lanka. Asian Myrmecology 5: 59-67.
- Dias R. K. S., K. R. K. A. Kosgamage, and H. A. W. S. Peiris. 2012. The Taxonomy and Conservation Status of Ants (Order: Hymenoptera, Family: Formicidae) in Sri Lanka. In: The National Red List 2012 of Sri Lanka; Conservation Status of the Fauna and Flora. Weerakoon, D.K. & S. Wijesundara Eds., Ministry of Environment, Colombo, Sri Lanka. p11-19.
- Dias R. K. S., and K. R. K. A. Kosgamage. Systematics and community composition of foraging workers ants (Family: Formicidae) collected from three habitats in a dry zone region of Sri Lanka. Proceedings of the Annual Research Symposium 2008. Faculty of Graduate Studies, University of Kelaniya.
- Dias R. K. S., and K. R. K. Anuradha Kosgamage. 2012. Occurrence and species diversity of ground-dwelling worker ants (Family: Formicidae) in selected lands in the dry zone of Sri Lanka. J. Sci. Univ. Kelaniya 7: 55-72.
- Dias R. K. S., and R. P. K. C. Rajapaksa. 2016. Geographic records of subfamilies, genera and species of ants (Hymenoptera: Formicidae) in the four climatic zones of Sri Lanka: a review. J. Sci. Univ. Kelaniya 11(2): 23-45.
- Dias R. K. S., and W. S. Udayakantha. 2016. Discovery of the Sri Lankan Relict Ant, Aneuretus simoni Emery (Formicidae, Aneuretinae) and the nest density of the species in a selected region of Meethirigala Forest Reserve, Sri Lanka. Asian Myrmecology 8: 1-8. DOI: 10.20362/am.008005
- Dias R. K. S.; Perera K. A. M. 2011. Worker ant community observed by repeated sampling and information on endemic Aneuretus simoni Emery in the Gilimale Forest Reserve in Sri Lanka. Asian Myrmecology 4: 69-78.
- Dias, R.K.S. 2006. Overview of ant research in Sri Lanka: 2000-2004. ANeT Newsletter 8:7-10
- Emery C. 1887. Catalogo delle formiche esistenti nelle collezioni del Museo Civico di Genova. Parte terza. Formiche della regione Indo-Malese e dell'Australia. [part]. Ann. Mus. Civ. Stor. Nat. 24(4): 209-258.
- Emery C. 1893. Voyage de M. E. Simon à l'île de Ceylan (janvier-février 1892). Formicides. Annales de la Société Entomologique de France 62: 239-258.
- Emery C. 1901. Ameisen gesammelt in Ceylon von Dr. W. Horn 1899. Deutsche Entomologische Zeitschrift 1901: 113-122.
- Emery C. 1901. Formiciden von Celebes. Zoologische Jahrbücher. Abteilung für Systematik, Geographie und Biologie der Tiere 14:565-580.
- Emery C. Formiche raccolte da Elio Modigliani in Sumatra, Engano e Mentawei. Annali del Museo Civico di Storia Naturale 40: 661-722.
- Emery, C. "Catalogo delle formiche esistenti nelle collezioni del Museo Civico di Genova. Parte terza. Formiche della regione Indo-Malese e dell'Australia." Annali del Museo Civico di Storia Naturale Giacomo Doria (Genova) (2) 4, no. 24 (1887): 209-258.
- Emery, C. "Formiche raccolte da Elio Modigliani in Sumatra, Engano e Mentawei." Annali del Museo Civico di Storia Naturale Giacomo Doria (Genova) (2) 20, no. 40 (1900): 661-722.
- Fellowes J. R. 1999. Exotic ants in Asia: is the mainland at risk? The case of Hong Kong. Aliens 9: 5-6.
- Fellowes J. R., and D. Dudgeon. 2003. Common ants of lowland forests in Hong Kong, Tropical China. Proceedings of the 2nd ANeT Workshop and Seminar, p. 19-43.
- Fontanilla A. M., A. Nakamura, Z. Xu, M. Cao, R. L. Kitching, Y. Tang, and C. J. Burwell. 2019. Taxonomic and functional ant diversity along tropical, subtropical, and subalpine elevational transects in southwest China. Insects 10, 128; doi:10.3390/insects10050128
- Forel A. 1895. Les Formicides de l'Empire des Indes et de Ceylan. Part V. J. Bombay Nat. Hist. Soc. 9: 453-472.
- Forel A. 1904. Note sur les fourmis du Musée Zoologique de l'Académie Impériale des Sciences à St. Pétersbourg. Ezheg. Zool. Muz. 8: 368-388.
- Forel A. 1905. Ameisen aus Java. Gesammelt von Prof. Karl Kraepelin 1904. Mitt. Naturhist. Mus. Hambg. 22: 1-26.
- Forel A. 1909. Études myrmécologiques en 1909. Fourmis de Barbarie et de Ceylan. Nidification des Polyrhachis. Bull. Soc. Vaudoise Sci. Nat. 45: 369-407.
- Forel A. 1911. Ameisen aus Ceylon, gesammelt von Prof. K. Escherich (einige von Prof. E. Bugnion). Pp. 215-228 in: Escherich, K. Termitenleben auf Ceylon. Jena: Gustav Fischer, xxxii + 262 pp.
- Forel A. 1911. Ameisen aus Java beobachtet und gesammelt von Herrn Edward Jacobson. II. Theil. Notes Leyden Mus. 33: 193-218.
- Forel A. 1913. H. Sauter's Formosa-Ausbeute: Formicidae II. Arch. Naturgesch. (A)79(6): 183-202
- Forel A. 1913. Quelques fourmis des Indes, du Japon et d'Afrique. Rev. Suisse Zool. 21: 659-673
- Forel A. 1913k. Wissenschaftliche Ergebnisse einer Forschungsreise nach Ostindien ausgeführt im Auftrage der Kgl. Preuss. Akademie der Wissenschaften zu Berlin von H. v. Buttel-Reepen. II. Ameisen aus Sumatra, Java, Malacca und Ceylon. Gesammelt von Herrn Prof. Dr. v. Buttel-Reepen in den Jahren 1911-1912. Zoologische Jahrbücher. Abteilung für Systematik, Geographie und Biologie der Tiere 36:1-148.
- Forel, A. 1908. Fourmis de Ceylan et d'Égypte récoltées par le Prof. E. Bugnion. Lasius carniolicus. Fourmis de Kerguelen. Pseudandrie? Strongylognathus testaceus. Bull. Soc. Vaudoise Sci. Nat. 44: 1-22
- Gaume L., M. Zacharias, V. Gosbois, and R. M. Borges. 2005. The fitness consequences of bearing domatia and having the right ant partner: experiments with protective and non-protective ants in a semi-myrmecophyte. Oecologia 145: 76-86.
- Ghosh S. N., S. Sheela, B. G. Kundu, S. Roychowdhury, and R. N. Tiwari. 2006. Insecta: Hymenoptera: Formicidae. Pp. 369-398 in: Alfred, J. R. B. (ed.) 2006. Fauna of Arunachal Pradesh. (Part -2). [State Fauna Series 13.]. New Delhi: Zoological Survey of India, iv + 518 pp.
- Ghosh S. N., S. Sheela, and B. G. Kundu. 2005. Ants (Hymenoptera: Formicidae) of Rabindra Sarovar, Kolkata. Records of the Zoological Survey of India. Occasional Paper 234: 1-40.
- Ghosh S. N., and S. Sheela. 2008. On a collection of Formicidae (Hymenoptera: Vespoidea) from Buxa Tiger Reserve, West Bengal, India, with new records of one rare genus and a rare species. Asian Myrmecology 2: 99-102.
- Gumawardene, N.R., J.D. Majer and J.P. Edirisinghe. 2008. Diversity and richness of ant species in a lowland wet forest reserve in Sri Lanka. Asian Myrmecology 2:71-83
- Gunawardene N. R., J. D. Majer, and J. P. Edirisinghe. 2008. Diversity and richness of ant species in a lowland wet forest reserve in Sri Lanka. Asian Myrmecology 2: 71-83.
- Gunawardene N. R., J. D. Majer, and J. P. Edirisinghe. 2012. Correlates of ant 5Hymenoptera: Formicidae) and tree species diversity in Sri Lanka. Myrmecological News 17: 81-90.
- Hashimoto Y., Y. Morimoto, E. S. Widodo, and M. Mohamed. 2006. Vertical distribution pattern of ants in a Bornean tropical rainforest (Hymenoptera: Formicidae). Sociobiology 47(3): 697- 710.
- Herwina H., N. Nasir, Jumjunidang, and Yaherwandi. 2013. The composition of ant species on banana plants with Banana Bunchy-top Virus (BBTV) symptoms in West Sumatra, Indonesia. Asian Myrmecology 5: 151-161.
- Herwina H., R. Satria, Yaherwandi, and Y. Sakamaki. 2018. Subterranean ant species diversity (Hymenoptera: Formicidae) in educational and biological research forest of universitas andalas, Indonesia. Journal of Entomology and Zoology Studies 6(1): 1720-1724.
- Hu C.-H. 2006. Indigenized conservation and biodiversity maintenance on Orchid Island. PhD Thesis, graduate school of the University of Minnesota. 150 pages.
- Hua Li-zhong. 2006. List of Chinese insects Vol. IV. Pages 262-273. Sun Yat-sen university Press, Guangzhou. 539 pages.
- Huong N. T. T., P. V. Sang, and B. T. Viet. 2015. A preliminary study on diversity of ants (Hymenoptera: Formicidae) at Hon Ba Nature Reserve. Environmental Scientific Conference 7: 614-620.
- Imai H. T., C. Baroni Urbani, M. Kubota, G. P. Sharma, M. H. Narasimhanna, B. C. Das, A. K. Sharma, A. Sharma, G. B. Deodikar, V. G. Vaidya, and M. R. Rajasekarasetty. 1984. Karyological survey of Indian ants. Japanese Journal of Genetics 59: 1-32.
- Jaitrong W., B. Guenard, E. P. Economo, N. Buddhakala, and S. Yamane. 2016. A checklist of known ant species of Laos (Hymenoptera: Formicidae). Asian Myrmecology 8: 1-32. DOI: 10.20362/am.008019
- Jaitrong W.; Nabhitabhata, J. 2005. A list of known ant species of Thailand. The Thailand Natural History Museum Journal 1(1): 9-54.
- Kadoorie Farm and Botanic Garden, 2002. Report of Rapid Biodiversity Assessments at Dinghushan Biosphere Reserve, Western Guangdong, 1998 and 2000. South China Forest Biodiversity Survey Report Series (Online Simplified Version): No. 7. KFBG, Hong Kong SAR, ii + 24pp.
- Karavaiev V. 1926. Ameisen aus dem Indo-Australischen Gebiet. Treubia 8: 413-445.
- Karmaly K. A.; S. Sumesh, T. P. Rabeesh, and L. Kishore. 2010. A checklist of ants of Thirunelli in Wayanad, Kerala. J. of the Bombay Natural History Society 107(1): 64-67.
- Kosgamage K. R. K. A., and R. K. S. Dias 2008. Systematics and community composition of Foraging worker ants ((Order: Hymenoptera, Family: Formicidae) collected from three habitats on a dry zone region of Sri Lanka.Proceedings of Postgraduate Symposium of Kelaniya University. 115pp.
- Lee T. L., and Y. S. Wei. 2005. Study for the temporal and spatial variation of the ant assemblages as the biological indicator in national parks. Journal of Animal and Veterinary Advances 4(4): 491-496.
- Li Shu-ping, Liu Fur-lin, Kang Jie, and Wang Yu-ling. 2005. Hymenoptera Formicidae Insect Name Record in Henan Province. Henan nong ye ke xue 5: 33-36.
- Li Z.h. 2006. List of Chinese Insects. Volume 4. Sun Yat-sen University Press
- Liu K. L., M. H. Peng, Y. C. Hung, and K. B. Neoh. 2019. Effects of park size, peri-urban forest spillover, and environmental filtering on diversity, structure, and morphology of ant assemblages in urban park. Urban Ecosystems https://doi.org/10.1007/s11252-019-00851-z)
- Lu Z., K. Li, N. Zhang, and Y. Chen. 2017. Diversity and indicator species of leaf-litter ants in Eucalyptus grandis plantations and secondary natural forests. Forest Research 29(4): 576-580
- Lu Z., and Y. Chen. 2016. Effects of habitat on ant functional groups: a case study of Luchun County, Yunnan Province, China. Chinese Journal of Eco-Agriculture 24(5): 801-810.
- Mohanraj P., M. Ali, and K. Veerakumari. 2010. Formicidae of the Andaman and Nicobar Islands (Indian Ocean: Bay of Bengal). Journal of Insect Science 10: Article 172
- Mohanraj, P., M. Ali and K. Veenakumari. 2010. Formicidae of the Andaman and Nicobar Islands (Indian Ocean: Bay Of Bengal). Journal of Insect Science 10:172.
- Narendra A., H. Gibb, and T. M. Ali. 2011. Structure of ant assemblages in Western Ghats, India: role of habitat, disturbance and introduced species. Insect Conservation and diversity 4(2): 132-141.
- Narendra A., and T. V. Ramachandra. 2008. Remote detection and distinction of ants using nest-site specific LISS-derived normalised difference vegetation index. Asian Myrmecology 2: 51-62.
- Norval G., W. F. Hsiao, C. C. Lin, and S. C. Huang. 2011. Ambushing the supply line: a report on Anolis sagrei predation on ants in Chiayi County, Taiwan. Russian Journal of Herpetology 18(1): 39-46.
- Pajni H. R., and R. K. Suri. 1978. First report on the Formicid fauna (Hymenoptera) of Chandigarh. Res. Bull. (Science) Punjab University 29: 5-12.
- Parui A. K., S. Chatterjee, and P. Basu. 2015. Habitat characteristics shaping ant species assemblages in a mixed deciduous forest in Eastern India. Journal of Tropical Ecology: 1-14. doi:10.1017/S0266467415000036
- Pfeiffer M.; Mezger, D.; Hosoishi, S.; Bakhtiar, E. Y.; Kohout, R. J. 2011. The Formicidae of Borneo (Insecta: Hymenoptera): a preliminary species list. Asian Myrmecology 4:9-58
- Pfeiffer, M., H. Cheng Tuck, and T. Chong Lay. 2008. Exploring arboreal ant community composition and co-ccurrence patterns in plantations of oil palm Elaeis guineensis in Borneo and Peninsular Malaysia. Ecography 31(1): 21-32.
- Rajan P. D., M. Zacharias, and T. M. Mustak Ali. 2006. Insecta: Hymenoptera: Formicidae. Fauna of Biligiri Rangaswamy Temple Wildlife Sanctuary (Karnataka). Conservation Area Series, Zool. Surv. India.i-iv,27: 153-188.
- Ranganathan Y., and R. M. Borges. 2009. Predatory and trophobiont-tending ants respond differently to fig and fig wasp volatiles. Animal Behaviour 77(6): 1539-1545.
- Rickson F.R., and M.M. Rickson. 1998. The cashew nut, Anacardium occidentale (Anacardiaceae), and its perennial association with ants: extrafloral nectary location and the potential for ant defense. American Journal of Botany 85(6): 835-849.
- Rizali A., D. J. Lohman, D. Buchori, L. Budi Prasetyo, H. Triwidodo, M. M. Bos, S. Yamane, and C. H. Schulze. 2009. Ant communities on small tropical islands: effects of island size and isolation are obscured by habitat disturbance and tramp ant species. Journal of Biogeography 37(2): 229-236.
- Rizali A., M. M. Bos, D. Buchori, Sk. Yamane, and C. H. Schulze. 2008. Ants in tropical urban habitats: the myrmecofauna in a densely populated area of Bogor, West Java, Indonesia. Hayati Journal of Biosciences 15(2): 77-84.
- Rizali A., M.M. Bos, D. Buchori, Sk. Yamane, C. Hans, and J. Schulze. 2008. Ants in tropical urban habitats: the myrmecofauna in a densely populated area of Bogor, West Java, Indonesia. Hayati Journal of Biosciences 77-84.
- Rizali A., Y. Clough, D. Buchori, M. L . A. Hosang, M. M. Bos, and T. Tscharntke. 2012. Long-term change of ant community structure in cacao agroforestry landscapes in Indonesia. Insect Conservation and Diversity doi: 10.1111/j.1752-4598.2012.00219.x
- Santschi F. 1937. Fourmis du Japon et de Formose. Bulletin et Annales de la Société Entomologique de Belgique. 77: 361-388.
- Shattuck S. O. 1994. Taxonomic catalog of the ant subfamilies Aneuretinae and Dolichoderinae (Hymenoptera: Formicidae). University of California Publications in Entomology 112: i-xix, 1-241.
- Smith F. 1861. Catalogue of hymenopterous insects collected by Mr. A. R. Wallace in the islands of Ceram, Celebes, Ternate, and Gilolo. [part]. Journal and Proceedings of the Linnean Society of London. Zoology 6: 36-48.
- Song Y., Z. Xu, C. Li, N. Zhang, L. Zhang, H. Jiang, and F. Mo. 2013. An Analysis on the Ant Fauna of the Nangun river Nature Reserve in Yunnan, China. Forest Research 26(6): 773-780.
- Tanaka H. O., S. Yamane, and T. Itioka. 2012. Effects of a fern-dwelling ant species, Crematogaster difformis, on the ant assemblages of emergent trees in a Bornean tropical rainforest. Ann. Entomol. Soc. Am. 105(4): 592-598.
- Tang J., Li S., Huang E., Zhang B. and Chen Y. 1995. Hymenoptera: Formicidae (1). Economic Insect Fauna of China 47: 1-133.
- Tie Ru, and Xu Sheng-quan. 2005. Ant species list in central part of Mt Qinling. Sichuan Journal of Zoology 24(1): 46.
- Tiwari R. N., B. G. Kundu, S. Roy Chowdhury, and S. N. Ghosh. 2003. Insecta: Hymenoptera: Formicidae. Fauna of Sikkim. Part 4. State Fauna Series. 9.Zool.Surv.India. i-iii, 1-512. Chapter pagination: 467-506.
- Trainor C.R. and A.N. Andersen. 2010. The ant fauna of Timor and neighbouring islands: potential bridges between the disjunct faunas of South East Asia and Australia. Australian Journal of Zoology 58: 133-144.
- Trainor C.R., and A.N. Andersen. 2010. The ant fauna of Timor and neighbouring islands: potential bridges between the disjunct faunas of South East Asia and Australia. Australian Journal of Zoology 58: 133-144.
- Varghese T. 2004. Taxonomic studies on ant genera of the Indian Institute of Science campus with notes on their nesting habits. Pp. 485-502 in : Rajmohana, K.; Sudheer, K.; Girish Kumar, P.; Santhosh, S. (eds.) 2004. Perspectives on biosystematics and biodiversity. Prof. T.C. Narendran commemoration volume. Kerala: Systematic Entomology Research Scholars Association, xxii + 666 pp.
- Viehmeyer H. 1912. Ameisen aus Deutsch Neuguinea gesammelt von Dr. O. Schlaginhaufen. Nebst einem Verzeichnisse der papuanischen Arten. Abhandlungen und Berichte des Königlichen Zoologischen und Anthropologische-Ethnographischen Museums zu Dresden 14: 1-26.
- Viehmeyer H. 1914. Ameisen aus Perak, Bali und Ceram (Hym.) (Freiburger Molukken-Expedition), gesammelt von E. Streesemann. Entomologische Mitteilungen. Berlin-Dahlem 3: 112-116.
- Wang W. R., S. Q. Zhang, and Z. H. Xu. 2012. A faunistic and taxonomic study of ants (Hymenoptera: Formicidae) in Shenzhen Municipality. Journal of Southwest Forestry University 32(1): 64-73.
- Wang W., S. Zhang, and Z Xu. 2012. Distribution patters of ant species in Shenzhen City. Journal of Southwest Forestry University 32(3): 70-74.
- Wang W., S. Zhang, and Z. Xu. 2012. Distribution Patterns of Ant Species in Shenzhen City. Journal of Southwest Forestry University 32(3): 69-74.
- Wang Wei. 2007. Fauna of Formicidae ants from three nature reserves in southwest Hubei. Chinese Bulletin of Entomology 44(2): 267-270.
- Way M. J., and B. Bolton. 1997. Competition between ants for coconut palm nesting sites. Journal of Natural History 31: 439-455.
- Wheeler W. M. 1909. Ants of Formosa and the Philippines. Bulletin of the American Museum of Natural History 26: 333-345.
- Wheeler W. M. 1913. Zoological results of the Abor Expedition, 1911-1912, XVII. Hymenoptera, II: Ants (Formicidae). Records of the Indian Museum 8: 233-237.
- Wheeler W. M. 1919. The ants of Borneo. Bulletin of the Museum of Comparative Zoology 63:43-147.
- Wheeler W. M. 1922. Ants of the American Museum Congo expedition. A contribution to the myrmecology of Africa. VIII. A synonymic list of the ants of the Ethiopian region. Bulletin of the American Museum of Natural History 45: 711-1004
- Wheeler W. M. 1923. Chinese ants collected by Professor S. F. Light and Professor A. P. Jacot. American Museum Novitates 69: 1-6.
- Wheeler W. M. 1927. Ants collected by Professor F. Silvestri in Indochina. Bollettino del Laboratorio di Zoologia Generale e Agraria della Reale Scuola Superiore d'Agricoltura. Portici 20: 83-106.
- Wheeler W. M. 1927. Chinese ants collected by Professor S. F. Light and Professor N. Gist Gee. American Museum Novitates 255: 1-12.
- Wheeler W. M. 1928. Ants collected by Professor F. Silvestri in China. Bollettino del Laboratorio di Zoologia Generale e Agraria della Reale Scuola Superiore d'Agricoltura. Portici 22: 3-38.
- Wheeler W. M. 1929. Ants collected by Professor F. Silvestri in Formosa, the Malay Peninsula and the Philippines. Bollettino del Laboratorio di Zoologia Generale e Agraria della Reale Scuola Superiore d'Agricoltura. Portici 24: 27-64.
- Wheeler W. M. 1930. A list of the known Chinese ants. Peking Natural History Bulletin 5: 53-81.
- Wheeler W. M. 1930. Formosan ants collected by Dr. R. Takahashi. Proceedings of the New England Zoological Club 11: 93-106.
- Wheeler W. M. 1937. Additions to the ant-fauna of Krakatau and Verlaten Island. Treubia 16: 21-24.
- Wu B., Y. Lu, G. Liang, and L. Zeng. 2010. Influence of the red inported fire ant, Solenopsis invicta Buren (Hymenoptera: Formicidae) on the diversity of ant communities in a newly infested longan orchard and grass areas nearby. Acta Ecologica Sinica 30(8): 2075-2083.
- Wu Z. W., X. D. Bi, X. He, Z. X. Lu, L. J. Wei, and Y. Q Chen. 2015. Impact of continuous fire disturbance on ground-dwelling ant communities in arid-hot valleys of Panzhihua, Sichuan. Journal of Yunnan University 37(3): 467-474.
- Xu Z. H., B. L. Yang, and G. Hu. 1999. Formicidae ant communities in fragments of montane rain forest in Xishuangbanna, China. Zoological Research 20(4): 288-293.
- Xu Z., Zeng G., Liu T.-Y. and He Y.-F. 1999. [A study on communities of Formicidae ants in different subtypes of vegetation in Xishuangbanna District of China.] Zoological Research 20: 118-125
- Yamane S. 2013. A Review of the ant fauna of the Krakatau Islands, Indonesia. Bull. Kitakyushu Mus. Nat. Hist. Hum. Hist. Ser: A, 11: 1-66
- Yamane S.; Bui T. V.; Ogata K.; Okido H.; Eguchi K. 2002. Ant fauna of Cuc Phuong National Park, North Vietnam (Hymenoptera: Formicidae). Bulletin of the Institute of Tropical Agriculture Kyushu University 25: 51-62.
- Yamane Sk. 2005. Krakatau in 1982, and the commencement of myrmecological research. The nature and Insects (Konchu to shizen) 40: 27-33.
- Yasumatsu, K. 1940. Matériaux pour servir à la faune myrmécologique des îles de Yaeyama. Mushi 13: 67-70
- Zhang N. N., Y. Q. Chen, Z. X. Lu, W. Zhang, and K. L. Li. 2013. Species diversity, community structure difference and indicator species of leaf-litter ants in rubber plantations and secondary natural forests in Yunnan, southwestern China. Acta Entomologica Sinica 56(11): 1314-1323.
- Zhou S.-Y. 2001. Ants of Guangxi. Guangxi Normal University Press, Guilin, China, Guilin, China. 255 pp.
- Zryanin V. A. 2011. An eco-faunistic review of ants (Hymenoptera: Formicidae). In: Structure and functions of soil communities of a monsoon tropical forest (Cat Tien National Park, southern Vietnam) / A.V. Tiunov (Editor). – M.: KMK Scientific Press. 2011. 277 р.101-124.
- Pages using DynamicPageList3 parser function
- Highly invasive
- Supercolonies
- Photo Gallery
- North subtropical
- Tropical
- Aphid Associate
- Host of Aphis clerodendri
- Host of Aphis gossypii
- Host of Hysteroneura setariae
- Host of Pentalonia nigronervosa
- Trematode Associate
- Host of Eurytrema pancreaticum
- Syrphid fly Associate
- Host of Bardistopus papuanum
- FlightMonth
- Karyotype
- Species
- Extant species
- Formicidae
- Dolichoderinae
- Technomyrmex
- Technomyrmex albipes
- Dolichoderinae species
- Technomyrmex species
- Ssr



