Solenopsis carolinensis
| Solenopsis carolinensis | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Solenopsidini |
| Genus: | Solenopsis |
| Species complex: | molesta |
| Species: | S. carolinensis |
| Binomial name | |
| Solenopsis carolinensis Forel, 1901 | |
| Synonyms | |
| |
Solenopsis carolinensis has been collected in underground nests, among other ant nests, in termite nests, as well as in the trunks of rotten trees in North Carolina (Forel, 1901a). Both workers and alate males and queens were collected by berlese extraction of hardwood leaf litter in Arkansas. Solenopsis carolinensis was collected under the bark of a dead tree and in a stump in Tennessee. Solenopsis carolinensis has also been collected by black light (July), in log litter and under rocks in various localities in Texas. Brood and sexuals were in nests in June. This species was collected in a Pinus taeda, mixed hardwood forest. Solenopsis carolinensis was also collected at 8500 ft. (about 2591 m) in Woodland Park Colorado. It is also found in grasslands and shrublands. (Pacheco and Mackay 2013)
| At a Glance | • Polygynous |
Identification
A New World thief ant that is a member of the molesta species complex. (Key to New World Solenopsis Species Complexes)
Pacheco and Mackay (2013) – Worker - The workers are nearly always small and yellow, but can be brown. The lateral clypeal teeth are angulate with the extralateral teeth absent. The mesosoma is smooth and shiny. This species is abundantly hairy and hairs on the posterior tibia are usually suberect. Queen - The queen is yellow, with large eyes. The head is coarsely punctate. The ocelli are darkly pigmented. The petiolar peduncle has a well-developed flange. Male - The male is slightly bicolored with a brown head and gaster and yellowish mesosoma, petiole and postpetiole. The head is mostly smooth and shiny and the clypeus is lacking any tooth or bump. The petiolar peduncle has a small flange ventrally.
Solenopsis carolinensis is difficult to separate from Solenopsis zeteki. It is usually slightly larger to the latter, but they can be approximately equal in size. The lateral clypeal teeth of S. carolinensis are well developed and the extralateral processes are developed at least into an angle. The lateral teeth are usually absent in S. zeteki, but may be developed into small angles. The extralateral angles of S. zeteki are absent or developed into a wide, slightly expanded area. Moreover, these two species can be separated by geography as S. carolinensis is found in the USA and S. zeteki is from Central and South America.
Solenopsis carolinensis could be confused with Solenopsis texana and with Solenopsis abdita. The workers of these two species have appressed hairs present on the posterior tibiae, whereas S. carolinensis has suberect pilosity. When queens are available, S. carolinensis, is easily distinguished by the large diameter of the eye. Queens and workers can be separated from those of S. abdita by the longer scapes (scape length of two type workers 0.240 and 0.279 mm, respectively) and the relatively narrower petiole (petiolar width of two type workers 0.083 and 0.094 mm, respectively).
Keys including this Species
Distribution
United States: Arkansas, Maryland, New Jersey, North Carolina, Tennessee, Texas, Virginia.
Latitudinal Distribution Pattern
Latitudinal Range: 40.58526° to 18.2805°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Nearctic Region: United States (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
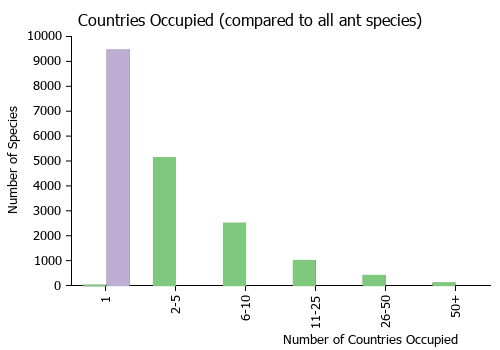
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Flight Period
| X | X | ||||||||||
| Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec |
Source: antkeeping.info.
- Check details at Worldwide Ant Nuptial Flights Data, AntNupTracker and AntKeeping.
 Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Association with Other Organisms
 Explore: Show all Associate data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Associate data or Search these data. See also a list of all data tables or learn how data is managed.
Virus
- This species is a host for the virus Aparavirus: Solenopsis invicta virus-1 (a parasite) (Quevillon, 2018) (encounter mode primary; direct transmission; transmission within nest).
Fungi
- This species is a host for the fungus Myrmicinosporidium durum (a pathogen) in United States (Pereira, 2004; Espadaler & Santamaria, 2012).
- This species is a host for the microsporidian fungus Kneallhazia carolinensae (a pathogen) (Valles et al., 2011).
Hymenoptera
- This species is a host for the diapriid wasp Auxopaedeutes sp. (a parasite) (www.diapriid.org) (potential host).
Life History Traits
- Queen number: polygynous (Frumhoff & Ward, 1992)
Castes
Worker
Images from AntWeb
   
| |
| Worker. Specimen code casent0104666. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by AMNH, New York, NY, USA. |
   
| |
| Worker. Specimen code casent0104497. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
   
| |
| Worker. Specimen code casent0010696. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by UCDC, Davis, CA, USA. |
Queen
Images from AntWeb
    
| |
| Queen (alate/dealate). Specimen code casent0104495. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Male
Images from AntWeb
   
| |
| Male (alate). Specimen code casent0104496. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- carolinensis. Solenopsis texana r. carolinensis Forel, 1901e: 345 (w.q.m.) U.S.A. (North Carolina).
- Type-material: lectotype worker (by designation of Pacheco & Mackay, 2013: 130), 5 paralectotype workers.
- Type-locality: lectotype U.S.A.: North Carolina, Faisons, 1899 (A. Forel); paralectotypes with same data.
- [Note: other original syntype workers, and queens and males, in MCZC, MHNG, not mentioned by Pacheco & Mackay, 2013: 130.]
- Type-depository: USNM.
- Combination in S. (Diplorhoptrum): Creighton, 1950a: 236.
- Subspecies of texana: Wheeler, W.M. 1904e: 301; Wheeler, W.M. 1910g: 563; Emery, 1922e: 201; Wesson, L.G. & Wesson, R.G. 1940: 92; Smith, M.R. 1951a: 814; Ettershank, 1966: 140.
- Status as species: Creighton, 1950a: 236; Smith, M.R. 1958c: 129; Smith, D.R. 1979: 1385; Thompson, 1989: 271 (in key); Thompson & Johnson, 1989: 698 (in key); Deyrup, et al. 1989: 96; Bolton, 1995b: 386; Deyrup, 2003: 47; Coovert, 2005: 67; MacGown & Forster, 2005: 69; Pacheco & Mackay, 2013: 127 (redescription); Deyrup, 2017: 102.
- Senior synonym of castanea: Pacheco & Mackay, 2013: 127.
- Senior synonym of truncorum: Pacheco & Mackay, 2013: 127.
- Distribution: U.S.A.
- castanea. Solenopsis molesta var. castanea Wheeler, W.M. 1908e: 430 (w.) U.S.A. (Colorado).
- Type-material: syntype workers (number not stated).
- Type-locality: U.S.A.: Colorado, Woodland Park (8500 ft) (W.M. Wheeler).
- Type-depository: MCZC.
- Subspecies of molesta: Wheeler, W.M. 1910g: 563; Wheeler, W.M. 1917a: 501; Emery, 1922e: 200; Essig, 1926: 859; Smith, M.R. 1951a: 814.
- Junior synonym of truncorum: Creighton, 1950a: 239; Smith, M.R. 1958c: 130; Smith, D.R. 1979: 1389; Bolton, 1995b: 386.
- Junior synonym of carolinensis: Pacheco & Mackay, 2013: 127.
- truncorum. Solenopsis texana r. truncorum Forel, 1901e: 346 (w.q.) U.S.A. (North Carolina).
- Type-material: lectotype queen (by designation of Pacheco & Mackay, 2013: 130), 1 paralectotype queen, 6 paralectotype workers.
- Type-locality: lectotype U.S.A.: North Carolina, Faisons, 28.vii.1899 (A. Forel); paralectotypes with same data.
- [Note: other original syntypes in MCZC.]
- Type-depository: MHNG.
- Subspecies of texana: Wheeler, W.M. 1904e: 301; Wheeler, W.M. 1910g: 563; Emery, 1922e: 201; Smith, M.R. 1951a: 814; Smith, M.R. 1958c: 130; Ettershank, 1966: 144; Smith, M.R. 1967: 358.
- Status as species: Creighton, 1950a: 239; Hunt & Snelling, 1975: 22; Smith, D.R. 1979: 1389; Mackay, Lowrie, et al. 1988: 101 (in key); Thompson & Johnson, 1989: 697 (in key); Bolton, 1995b: 391; Mackay & Mackay, 2002: 233; Deyrup, 2003: 47; Ward, 2005: 67.
- Junior synonym of carolinensis: Pacheco & Mackay, 2013: 127.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Pacheco and Mackay (2013) - Measurements (n=6). TL 1.32-1.48 (1.42); HL 0.402-0.420 (0.412); HW 0.324-0.360 (0.337); EL 0.036-0.042 (0.041); ED 0.030-0.036 (0.031); SL 0.270-0.300 (0.279); FSL 0.102-0.120 (0.108); CI 78.6-85.7 (81.8); SI 64.3-71.4 (67.7); PL 0.060-0.072 (0.065); PW 0.090-0.102 (0.094); PI 58.8-75.0 (69.4); PPL 0.066-0.084 (0.076); PPW 0.120-0.126 (0.122); PPI 55.0-70.0 (62.3); WL 0.300-0.330 (0.305); PSL 0.024-0.030 (0.029); PSW 0.024.
Small, concolorous yellow; head subquadrate, longer than wide, posterior border straight; lateral clypeal teeth angulate, extralateral teeth absent; clypeal carinae well defined; scape reaches 3/4 length of head; minor funicular segments 3-8 short; eyes small, 3-5 ommatidia; mesosoma smooth and shiny; metapleuron with faint horizontal striae; posterior propodeal margin rounded; petiole wider than postpetiole viewed laterally; petiolar node rounded, subtriangular, with tooth ventrally; postpetiolar node rounded, globose, sculpture of petiole and postpetiole smooth and shiny.
Abundantly hairy; erect and suberect hairs of various lengths on all body surfaces; hairs on posterior tibia usually at least suberect.
Queen
Pacheco and Mackay (2013) - Measurements (n=2). TL 4.08-4.20 (4.14); HL 0.648; HW 0.600; EL 0.240; ED 0.180-0.192 (0.186); MOL 0.078-0.090 (0.084); MOD 0.084-0.096 (0.090); SL 0.450-0.462 (0.456); FSL 0.222-0.240 (0.231); CI 92.6; SI 69.4-71.3 (70.4); PSL 0.066; PSW 0.048-0.060 (0.054); PL 0.132; PW 0.246-0.270 (0.258); PI 48.9-53.7 (51.3); PPL 0.198; PPW 0.282-0.288 (0.285); PPI 68.8-70.2 (69.5); WL 0.900.
Moderately large; concolorous yellow; head sub quadrate, longer than wide, coarsely punctate, posterior margin straight; lateral clypeal teeth angulate, extralateral teeth absent; clypeal carinae poorly defined; frontal lobes vertically striated; eye black, large; ocelli darkly pigmented, medial ocellus large; pronotum coarsely punctate, smooth and shiny between punctures, mesopleuron without sculpturing, lower metapleuron with horizontal striae; petiole wider than postpetiole viewed laterally; petiolar peduncle with well-developed flange ventrally. Abundantly hairy; erect and suberect hairs of various lengths covering all body surfaces; most hairs on petiole and postpetiole curve posteriorly.
Male
Pacheco and Mackay (2013) - Measurements (n=4). TL 2.88-3.00 (2.91); HL 0.432-0.450 (0.441); HW 0.402-0.480 (0.446); EL 0.282-0.300 (0.291); ED 0.240; MOL 0.084-0.090 (0.087); MOD 0.132-0.150 (0.141); SL 0.144-0.168 (0.155); FSL 0.900-0.960 (0.945); CI 93.1-107 (101); SI 33.3-37.3 (35.0); PSL 0.072; PSW 0.060-0.066 (0.064); PL 0.138-0.144 (0.139); PW 0.198-0.216 (0.209); PI 63.9-69.7 (66.9); PPL 0.144-0.162 (0.153); PPW 0.228; PPI 63.2-71.1 (67.1); WL 0.720-0.840 (0.780).
Bicolored, brown head and gaster, yellow mesosoma, petiole and postpetiole, antennae yellow; head wider than long, smooth, shiny; clypeus convex, lacking tooth or bump; frontal lobes widely separated, 0.096 mm in greatest width; mesosoma smooth, shiny; petiole wider than postpetiole viewed laterally; petiolar peduncle with small flange ventrally; postpetiole wider than petiole viewed dorsally, lacking tooth or flange ventrally.
Abundantly hairy, with erect and suberect hairs of various lengths covering all body surfaces; most hairs on petiole and postpetiole curve posteriorly.
Type Material
Pacheco and Mackay (2013) - North Carolina, Faisons, (lectotype worker and 5 paralectotype workers [here designated], cotype #201786 National Museum of Natural History). Solenopsis texanus truncorum, (lectotype queen Musee d'Histoire Naturelle Genève, 1 paralectotype queen, 6 paralectotype workers [here designated] MHNG). Solenopsis texana truncorum, North Carolina, Faisons du tronc 28.VII.
Worker Morphology
 Explore: Show all Worker Morphology data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Worker Morphology data or Search these data. See also a list of all data tables or learn how data is managed.
- Caste: monomorphic
References
- Atchison, R. A., Lucky, A. 2022. Diversity and resilience of seed-removing ant species in Longleaf Sandhill to frequent fire. Diversity 14, 1012 (doi:10.3390/d14121012).
- Baty, J.W., Bulgarella, M., Dobelmann, J., Felden, A., Lester, P.J. 2020. Viruses and their effects in ants (Hymenoptera: Formicidae). Myrmecological News 30: 213-228 (doi:10.25849/MYRMECOL.NEWS_030:213).
- Creighton, W. S. 1950a. The ants of North America. Bulletin of the Museum of Comparative Zoology 104: 1-585 (page 236, Combination in S. (Diplorhoptrum), raised to species)
- Davis, T. 2009. The ants of South Carolina (thesis, Clemson University).
- Espadaler, X., Santamaria, S. 2012. Ecto- and Endoparasitic Fungi on Ants from the Holarctic Region. Psyche Article ID 168478, 10 pages (doi:10.1155/2012/168478).
- Forel, A. 1901j. Variétés myrmécologiques. Ann. Soc. Entomol. Belg. 45: 334-382 (page 345, worker, queen, male described)
- Gochnour, B.M., Suiter, D.R., Booher, D. 2019. Ant (Hymenoptera: Formicidae) fauna of the Marine Port of Savannah, Garden City, Georgia (USA). Journal of Entomological Science 54, 417-429 (doi:10.18474/jes18-132).
- Hill, J.G. 2015. Ants (Hymenoptera: Formicidae) of the Big Thicket Region of Texas. Midsouth Entomologist 8: 24-34.
- Ipser, R.M., Brinkman, M.A., Gardner, W.A., Peeler, H.B. 2004. A survey of ground-dwelling ants (Hymenoptera: Formicidae) in Georgia. Florida Entomologist 87: 253-260.
- Ivanov, K. 2019. The ants of Ohio (Hymenoptera, Formicidae): an updated checklist. Journal of Hymenoptera Research 70: 65–87 (doi:10.3897@jhr.70.35207).
- MacGown, J.A., Booher, D., Richter, H., Wetterer, J.K., Hill, J.G. 2021. An updated list of ants of Alabama (Hymenoptera: Formicidae) with new state records. Transactions of the American Entomological Society 147: 961-981 (doi:10.3157/061.147.0409).
- Pacheco, J.A. & Mackay, W.P. 2013. The systematics and biology of the New World thief ants of the genus Solenopsis (Hymenoptera: Formicidae). Edwin Mellen Press, Lewiston, New York. 501 pp.
- Pereira, R.M. 2004. Occurrence of Myrmicinosporidium durum in red imported fire ant, Solenopsis invicta, and other new host ants in eastern United States. Journal of Invertebrate Pathology 86: 38-44 (doi:10.1016/j.jip.2004.03.005).
- Sharaf, M.R., Gotzek, D., Guénard, B., Fisher, B.L., Aldawood, A.S., Al Dhafer, H.M., Mohamed, A.A. 2020. Molecular phylogenetic analysis and morphological reassessments of thief ants identify a new potential case of biological invasions. Scientific Reports 10, 12040 (doi:10.1038/s41598-020-69029-4).
- Thompson, C. R.; Johnson, C. 1989. Rediscovered species and revised key to the Florida thief ants (Hymenoptera: Formicidae). Fla. Entomol. 72: 697-698 (page 698, see also)
- Valles, S.M., Becnel, J.J. & Pereira, R.M. 2011. Kneallhazia carolinensae sp. nov., a microsporidian pathogen of the thief ant, Solenopsis carolinensis. Journal of Invertebrate Pathology, 108(1), 59–62 (DOI 10.1016/j.jip.2011.06.012).
References based on Global Ant Biodiversity Informatics
- Annotated Ant Species List Ordway-Swisher Biological Station. Downloaded at http://ordway-swisher.ufl.edu/species/os-hymenoptera.htm on 5th Oct 2010.
- Colby, D. and D. Prowell. 2006. Ants (Hymenoptera: Formicidae) in Wet Longleaf Pine Savannas in Louisiana. Florida Entomologist 89(2):266-269
- Coovert, G.A. 2005. The Ants of Ohio (Hymenoptera: Formicidae) Ohio Biological Survey Bulletin New Series Volume 15(2):1-196
- Dash S. T. and L. M. Hooper-Bui. 2008. Species diversity of ants (Hymenoptera: Formicidae) in Louisiana. Conservation Biology and Biodiversity. 101: 1056-1066
- Dattilo W. et al. 2019. MEXICO ANTS: incidence and abundance along the Nearctic-Neotropical interface. Ecology https://doi.org/10.1002/ecy.2944
- Deyrup M. 1998. Smithistruma memorialis (Hymenoptera: Formicidae), a new species of ant from the Kentucky Cumberland Plateau. Entomological News 109: 81-87.
- Deyrup M., C. Johnson, G. C. Wheeler, J. Wheeler. 1989. A preliminary list of the ants of Florida. Florida Entomologist 72: 91-101
- Deyrup, M. 2003. An updated list of Florida ants (Hymenoptera: Formicidae). Florida Entomologist 86(1):43-48.
- Deyrup, M. and J. Trager. 1986. Ants of the Archbold Biological Station, Highlands County, Florida (Hymenoptera: Formicidae). Florida Entomologist 69(1):206-228
- Forel A. 1901. Variétés myrmécologiques. Annales de la Société Entomologique de Belgique 45: 334-382.
- Forster J.A. 2005. The Ants (hymenoptera: Formicidae) of Alabama. Master of Science, Auburn University. 242 pages.
- Gregg, R.T. 1963. The Ants of Colorado.
- Ipser R. M. 2004. Native and exotic ants (Hymenoptera: Formicidae) of Georgia: Ecological Relationships with implications for development of biologically-based management strategies. Doctor of Philosophy thesis, University of Georgia. 165 pages.
- Ivanov, K. 2019. The ants of Ohio (Hymenoptera, Formicidae): an updated checklist. Journal of Hymenoptera Research 70: 65–87.
- Ivanov K., L. Hightower, S. T. Dash, and J. B. Keiper. 2019. 150 years in the making: first comprehensive list of the ants (Hymenoptera: Formicidae) of Virginia, USA. Zootaxa 4554 (2): 532–560.
- Johnson C. 1986. A north Florida ant fauna (Hymenoptera: Formicidae). Insecta Mundi 1: 243-246
- Johnson R. Personnal Database. Accessed on February 5th 2014 at http://www.asu.edu/clas/sirgtools/resources.htm
- Lubertazzi D. and Tschinkel WR. 2003. Ant community change across a ground vegetation gradient in north Floridas longleaf pine flatwoods. 17pp. Journal of Insect Science. 3:21
- Lynch J. F. 1988. An annotated checklist and key to the species of ants (Hymenoptera: Formicidae) of the Chesapeake Bay region. The Maryland Naturalist 31: 61-106
- MacGown J. A. 2015. Report on the ants collected on Spring Island, Beaufort County, South Carolina. A report submitted to Spring Island Nature Preserve, May 2015. Mississippi Entomological Museum Report #2015-01. 8 pp
- MacGown J. A., J. G. Hill, R. L. Brown, T. L. Schiefer, J. G. Lewis. 2012. Ant diversity at Noxubee National Wildlife Refuge in Oktibbeha, Noxubee, and Winston Counties, Mississippi. Mississippi Agricultural and Forestry Experiment Station Bulletin 1197: 1-30
- MacGown J. A., J. G. Hill, and M. Deyrup. 2009. Ants (Hymenoptera: Formicidae) of the Little Ohoopee River Dunes, Emanuel County, Georgia. J. Entomol. Sci. 44(3): 193-197.
- MacGown J. A., J. G. Hill, and R. L. Brown. 2010. Native and exotic ant in Mississippi state parks. Proceedings: Imported Fire Ant Conference, Charleston, South Carolina, March 24-26, 2008: 74-80.
- MacGown J. A., and R. Whitehouse. 2015. A preliminary report of the ants of West Ship Island. A report submitted to the Gulf Islands National Seashore. Mississippi Entomological Museum Report #2015-02. 9 pp.
- MacGown, J.A and J.A. Forster. 2005. A preliminary list of the ants (Hymenoptera: Formicidae) of Alabama, U.S.A. Entomological News 116(2):61-74
- MacGown, J.A. and JV.G. Hill. Ants of the Great Smoky Mountains National Park (Tennessee and North Carolina).
- MacGown, J.A. and T. Lockley. Ants of Horn Island, Jackson County, Mississippi
- MacGown. J. 2011. Ants collected during the 25th Annual Cross Expedition at Tims Ford State Park, Franklin County, Tennessee
- Mackay W. P. and Mackay, E. E. 2002. The ants of New Mexico (Hymenoptera: Formicidae). Lewiston, New York: Edwin Mellen Press, 400 pp.
- Mann H. R., E. Rowe, J. Selfridge, and D. L. Price. 2018. Leaf litter and arboreal ants (Hymenoptera: Formicidae) in a Mid-Atlantic Forest. Northeastern Naturalist 25(2): 341-354.
- McDonald D. L., D. R. Hoffpauir, and J. L. Cook. 2016. Survey yields seven new Texas county records and documents further spread of Red Imported Fire Ant, Solenopsis invicta Buren. Southwestern Entomologist, 41(4): 913-920.
- Moreau C. S., M. A. Deyrup, and L. R. David Jr. 2014. Ants of the Florida Keys: Species Accounts, Biogeography, and Conservation (Hymenoptera: Formicidae). J. Insect Sci. 14(295): DOI: 10.1093/jisesa/ieu157
- O'Neill J.C. and Dowling A.P.G. 2011. A Survey of the Ants (hymenoptera: Formicidae) of Arkansas and the Ozark Mountains. An Undergraduate Honors, University of Arkansas. 18pages.
- Oyama L., J. R. King, and D. G. Jenkins. 2018. Diversity and distribution of Solenopsis (Hymenoptera: Formicidae) thief ants belowground. Myrmecological News 27: 47-57.
- Pacheco J. A., and W. P. Mackay. 2013. The systematics and biology of the New World thief ants of the genus Solenopsis (Hymenoptera: Formicidae). Lewiston, New York: Edwin Mellen Press, 501 pp.
- Parys K. A., M. L. Gimmel, and S. J. Johnson. 2013. Checklist of Insects Associated with Salvinia minima Baker in Louisiana, USA. Check List 9(6): 14881495.
- Rowles, A.D. and J. Silverman. 2009. Carbohydrate supply limits invasion of natural communities by Argentine ants. Oecologia 161(1):161-171
- Thompson C. R., and C. Johnson. 1989. Rediscovered species and revised key to the Florida thief ants (Hymenoptera: Formicidae). Florida Entomologist 72: 697-698.
- Wheeler W. M. 1904. The ants of North Carolina. Bulletin of the American Museum of Natural History. 20: 299-306.
- Pages using DynamicPageList3 parser function
- Polygynous
- North temperate
- North subtropical
- Tropical
- FlightMonth
- Virus Associate
- Host of Aparavirus: Solenopsis invicta virus-1
- Fungus Associate
- Host of Myrmicinosporidium durum
- Microsporidian fungus Associate
- Host of Kneallhazia carolinensae
- Diapriid wasp Associate
- Host of Auxopaedeutes sp.
- Species
- Extant species
- Formicidae
- Myrmicinae
- Solenopsidini
- Solenopsis
- Solenopsis carolinensis
- Myrmicinae species
- Solenopsidini species
- Solenopsis species
- Ssr

