Lasius sakagamii
| Lasius sakagamii | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Formicinae |
| Tribe: | Lasiini |
| Genus: | Lasius |
| Section: | niger clade |
| Species group: | niger |
| Species: | L. sakagamii |
| Binomial name | |
| Lasius sakagamii Yamauchi & Hayashida, 1970 | |
| Common Name | |
|---|---|
| Kawara-ke-ari | |
| Language: | Japanese |
Widespread in Japan. Occurs in open areas with sandy and alluvial soils, from sea level to 500m.
| At a Glance | • Polygynous • Diploid male • Supercolonies • Limited invasive |
Identification
Japanese Ant Image Database - Total length of workers around 2.5 - 3.5 mm. Color brown. Scapes as long as the head is wide, and with more than 30 erect hairs. Anterior tibiae with many erect hairs. Petiole thick and low, anterior margin with a weak, blunt angle in lateral view.
Seifert (2020) - Lasius sakagamii represents an unmistakable combination of extremely large setae numbers, flat propodeal dome, low petiole and low clypeal pubescence distance.
Keys including this Species
Distribution
Widely spread all over Japan from sea level to 500 m. Okinawa records (Naha City) may be an introduced population resulting from human commerce.
Latitudinal Distribution Pattern
Latitudinal Range: 30.359° to 30.359°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Palaearctic Region: Japan (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
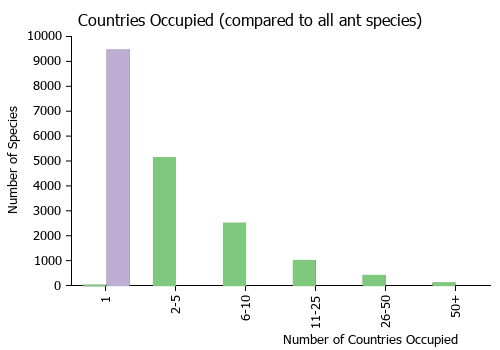
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Habitat
Yamauchi, Kinomura, Miyake (1981, part I) - Apparently stenotopic, living mainly in sandy areas with sparse vegetation. Lasius sakagamii particularly prefers flood plains.
Biology
Japanese Ant Image Database - This species nests in the soil in dry environments such as grasslands at riversides or seashores. Nests are polygynous and polydomous, and multiply by budding. The number of contained individuals is large, sometimes reaching several hundred thousand (Yamauchi, 1981; Yamauchi et al., 1981, 1982). The nuptial-flight period is relatively long for a species of subgenus Lasius with records from June to October. Collections of L. sakagamii from Okinawa (Naha City) probably represent an introduction by human commerce. Diploid males are known to occur in this species (found in 20% of 30 examined nests) (Yamauchi et al., 2001).
Seifert (2020) - Main habitat are sun-exposed deposition areas of alluvial and aeolian sands with only sparse vegetation; these are river banks, dry river beds, sand dunes or road sides. Nests are usually constructed at spots with bare sand or under stones and have numerous distinct chambers usually reaching down to 60 cm. Nuptial flights were observed from early July to late September at warm evenings with air temperatures > 22°, high air humidity and no air movements. The species is polygynous and in many nests only males fly. Nuptial flight of gynes only takes place from nests without males, otherwise they mate intranidally with subsequent dispersal flight. It may form supercolonies dominating large habitat areas.
Flight Period
| X | X | X | X | X | |||||||
| Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec |
Notes: Japan.
- Check details at Worldwide Ant Nuptial Flights Data, AntNupTracker and AntKeeping.
 Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Flight Month data or Search these data. See also a list of all data tables or learn how data is managed.
Life History Traits
- Queen number: polygynous (Rissing and Pollock, 1988; Frumhoff & Ward, 1992)
- Colony type: polydomous
- Maximum colony size: 300000 (Japanese Ant Image Database) (reported as "several hundred thousand")
- Colony founding: dependent
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- sakagamii. Lasius sakagamii Yamauchi & Hayashida, 1970: 504, figs. 3, 4, 6, 9, 10 (w.q.m.) JAPAN. See also: Yamauchi, 1979: 157; Seifert, 1992b: 42.
Description
Worker
Seifert (2020) - Medium-sized (CS 887 µm). Head moderately long (CL/CW900 1.071). Scape long (SL/CS900 1.034). Postocular distance small and eye medium-sized (PoOc/ CL900 0.225, EYE/CS900 0.244). Torulo-clypeal distance large (dClAn900 5.21). Number of mandibular dents large (MaDe 8.52). Terminal segment of maxillary palp rather long (MP6/CS900 0.194). Pubescence on clypeus dense (sqPDCL900 3.78) and on frons rather long (PLF900 33.0). Gular and pronotal setae rather long (GuHL/CS900 0.110, PnHL/CS900 0.157). Whole body covered by extremely numerous standing setae (nOcc900 23.4, nGen900 16.1, nGu900 18.6, nSc900 32.0, nHT900 29.8, nSt900 12.6). Mesosoma with very flat propodeal dome and convex to angulate-convex posterior propodeal slope that is transversally carinulate. Petiole scale in lateral view thick, low and with a blunt apex; in anterior view rather narrow, with convex to nearly straight subparallel sides. Coloration: mesosoma medium brown with a yellowish-reddish tinge, head a little and gaster distinctly darker; sometimes whole body concolorous pale or dark brown.
Also see table 11 in Seifert 2020 for additional morphometric measurements, ratios, and functions. Abbreviations given above for measurements, ratios and functions are defined here: Seifert 2020 Lasius characters.
Type Material
Seifert (2020) - One paratype worker from the holotype nest series labelled ”Sapporo Hokkaido 30-VII-1966 K. Yamauchi“; depository Staatliches Museum für Naturkunde Görlitz.
Karyotype
- See additional details at the Ant Chromosome Database.
 Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
- n = 15, 2n = 30, karyotype = 2M+28A (Japan) (Yamauchi et al., 2001).
The diploid chromosome number in female Lasius sakagamii is 30, while the haploid number in normal males is 15. Six of the 30 studied colonies (20%), however, contained many diploid males. Although the body size of the diploid males was, on average, larger than that of the haploid males, both types of males showed normal external and internal morphologies. Furthermore, there was no reduction in chromosome numbers in either diploid or haploid males during spermatogenesis. The spermatid nuclei of diploid males, however, were significantly larger than those of haploid males, and the amount of DNA in the diploid males, as measured by flow cytometry, was twice as much as that in the haploid males, clearly indicating that diploid males produce diploid sperm (Yamauchi et al., 2001).
References
- Armitage, S., Boomsma, J., Bear, B. 2010. Diploid male production in a leaf-cutting ant. Ecological Entomology 35, 175–182 (doi:10.1111/j.1365-2311.2009.01167.x).
- Elias, J., Mazzi, D., Dorn, S. 2009. No need to discriminate? Reproductive diploid males in a parasitoid with complementary sex determination. PLoS ONE 4, e6024. (doi:10.1371/journal.pone.0006024).
- Harpur, B.A., Sobhani, M., Zayed, A. 2013. A review of the consequences of complementary sex determination and diploid male production on mating failures in the Hymenoptera. Entomologia Experimentalis et Applicata 146, 156–164. (doi:10.1111/J.1570-7458.2012.01306.X).
- Imai, H.T., Kihara, A., Kondoh, M., Kubota, M., Kuribayashi, S., Ogata, K., Onoyama, K., Taylor, R.W., Terayama, M., Yoshimura, M., Ugawa, Y. 2003. Ants of Japan. 224 pp, Gakken, Japan.
- Jacobs, S. 2020. Population genetic and behavioral aspects of male mating monopolies in Cardiocondyla venustula (Ph.D. thesis).
- Lee, C.-C., Weng, Y.-M., Lai, L.-C., Suarez, A.V., Wu, W.-J., Lin, C.-C., Yang, C.-C.S. 2020. Analysis of recent interception records reveals frequent transport of arboreal ants and potential predictors for ant invasion in Taiwan. Insects 11, 356 (doi:10.3390/INSECTS11060356).
- Lorite, P., Palomeque, T. 2010. Karyotype evolution in ants (Hymenoptera: Formicidae), with a review of the known ant chromosome numbers. Myrmecological News 13: 89-102.
- Schär, S., Talavera, G., Rana, J.D., Espadaler, X., Cover, S.P., Shattuck, S.O., Vila, R. 2022. Integrative taxonomy reveals cryptic diversity in North American Lasius ants, and an overlooked introduced species. Scientific Reports 12: 5970: 1-12 (doi:10.1038/s41598-022-10047-9).
- Schultner, E., Pulliainen, U. 2020. Brood recognition and discrimination in ants. Insectes Sociaux 67, 11–34 (doi:10.1007/s00040-019-00747-3).
- Seifert, B. 1992b. A taxonomic revision of the Palaearctic members of the ant subgenus Lasius s.str. (Hymenoptera: Formicidae). Abh. Ber. Naturkundemus. Görlitz 66(5): 1-67 (page 42, see also)
- Seifert, B. 2020. A taxonomic revision of the Palaearctic members of the subgenus Lasius s.str. (Hymenoptera, Formicidae). Soil Organisms 92(1): 15-86 (doi:10.25674/so92iss1pp15).
- Sunamura, E., Tamura, S., Urano, T., Shoda-Kagaya, E. 2020. Predation of invasive red-necked longhorn beetle Aromia bungii (Coleoptera: Cerambycidae) eggs and hatchlings by native ants in Japan. Applied Entomology and Zoology 55, 291–298 (doi:10.1007/S13355-020-00681-Y).
- Whiting, P.W. 1960. Polyploidy in Mormoniella. Genetics 45: 949–970. Yamauchi, K., K. Kinomura and S. Miyake, 1981. Sociobiological studies of the polygynic ant Lasius sakagamii. I-General features of its polydomous system. Insectes Sociaux 28: 279–296.
- Yamauchi, K. 1979 [1978]. Taxonomical and ecological studies on the ant genus Lasius in Japan (Hymenoptera: Formicidae). I. Taxonomy. Sci. Rep. Fac. Educ. Gifu Univ. (Nat. Sci.) 6: 147-181 (page 157, see also)
- Yamauchi, K., Yoshida, T., Ogawa, T., Itoh, S., Ogawa, Y., Jimbo, S., Imai, H.T. 2001. Spermatogenesis of diploid males in the formicine ant, Lasius sakagamii. Insectes Sociaux 48 28–32.
- Yamauchi, K.; Hayashida, K. 1970. Taxonomic studies on the genus Lasius in Hokkaido, with ethological and ecological notes (Formicidae, Hymenoptera). II. The subgenus Lasius. J. Fac. Sci. Hokkaido Univ. Ser. VI. Zool. 17: 501-519 (page 504, figs. 3, 4, 6, 9, 10 worker, queen, male described)
References based on Global Ant Biodiversity Informatics
- Choi B.M., K. Ogata, and M. Terayama. 1993. Comparative studies of ant faunas of Korea and Japan. 1. Faunal comparison among islands of Southern Korean and northern Kyushu, Japan. Bull. Biogeogr. Soc. Japan 48(1): 37-49.
- Hosoishi S., M. Yoshimura, Y. Kuboki, and K. Ogata. 2007. Ants from Yakushima Island , Kagoshima Prefecture. Ari 30: 47-54.
- Kondoh M., and Y. Kitazawa. 1984. Ant communities of the campus of UOEH and in an adjacent natural forest. Journal of UOEH 6(3): 221-234.
- Masuko K., H. Yamaoka, T. Kannari, and S. Usuba. 1985. Ants of Mt. Kiyosumi (4). Kiyosumi 11: 9-12.
- Mizutani A. 1979. A myrmecofaunal survey at Hiyama Experiment Forest, Hokkaido University. Research Bulletin of the College Experiment Forests, Hokkaido University 36:509-516.
- Mizutani A. 1979. A myrmecofaunal survey at Hiyama Experiment Forest, Hokkaido University. Research Bulletins of the College Experiment Forests Hokkaido University 36(2): 509-516.
- Negoro H. 1994. Ants from Toyama Prefecture, Hokuriku. Bulletin of the Toyama Science Museum 17: 35-47.
- Ogata K. 1981. The ant fauna of the Goto islands, Natural history of the Goto? Islands, Japan : Iki Tsushima to no taihi (Danjo Gunto? Ko?rai Sone o fukumu Japan: 347-351.
- Shimana Y., and S. Yamane. 2009. Geogrpahical distribution of Technomyrmex brunneus Forel (Hymenoptera, Formicidae) in the western part of the mainland of Kagoshima, South Kyushu, Japan. Ari 32: 9-19.
- Talavera G., X. Espadaler, and R. Vila. Discovered just before extinction? The first endemic ant from the Balearic Islands (Lasius balearicus sp. nov.) is endangered by climate change. Journal of Biogeography doi:10.1111/jbi.12438
- Terayama M. 1977. Checklist of the known ants of Saitama Prefecture. Insects and nature 12(4): 26-27
- Terayama M. 1983. Kagoshima-ken-hondo no ari. Kanagawa-chucho (Journal of the Kanagawa Entomologists Association): 13-24.
- Terayama M. 1992. Structure of ant communities in East Asia. A. Regional differences and species richness. Bulletin of the Bio-geographical Society of Japan 47: 1-31.
- Terayama M., K. Ogata, and B.M. Choi. 1994. Distribution records of ants in 47 prefectures of Japan. Ari (report of the Myrmecologists Society of Japan) 18: 5-17.
- Terayama M., S. Kubota, and K. Eguchi. 2014. Encyclopedia of Japanese ants. Asakura Shoten: Tokyo, 278 pp.
- Terayama M., and S. Kubota. 1994. Ants from Aogashima Island. Ari 17: 11.
- Terayama M., and S. Kubota. 2002. Ants of Tokyo, Japan. ARI 26: 1-32.
- Teruyama. M. 1988. Ant fauna of Saitama Prefecture, Japan. ARI Reports of the Myrmecologists Society (Japan) 16: 4-13
- Yamane S., S. Ikudome, and M. Terayama. 1999. Identification guide to the Aculeata of the Nansei Islands, Japan. Sapporo: Hokkaido University Press, xii + 831 pp. pp, 138-317.
- Yamane S., Y. Harada, and K. Eguchi. 2013. Classification and ecology of ants. Natural history of ants in Southern Kyushu. 200 pages
- Yamane S.; Ikudome, S.; Terayama, M. 1999. Identification guide to the Aculeata of the Nansei Islands, Japan. Sapporo: Hokkaido University Press, xii + 831 pp. pp138-317.
- Yamauchi K. 1979. Taxonomical and ecological studies on the ant genus Lasius in Japan (Hymenoptera: Formicidae). I. Taxonomy. Sci. Rep. Fac. Educ. Gifu Univ. (Nat. Sci.) 6: 147-181.
- Yamauchi K., and K. Hayashida. 1970. Taxonomic studies on the genus Lasius in Hokkaido, with ethological and ecological notes (Formicidae, Hymenoptera). II. The subgenus Lasius. J. Fac. Sci. Hokkaido Univ. Ser. VI. Zool. 17: 501-519.
- Yoshimura M., T. Hirata, A. Nakajima, and K. Onoyama. 2003. Ants found in scats and pellets taken from the nests of the Japanese Wryneck Jynx torquilla japonica. Ornithol. Sci. 2: 127-131.

