Sericomyrmex lutzi
| Sericomyrmex lutzi | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Sericomyrmex |
| Species: | S. lutzi |
| Binomial name | |
| Sericomyrmex lutzi Wheeler, W.M., 1916 | |
Other than specimens noted as being found in forest, nothing is known about the biology of Sericomyrmex lutzi.
Identification
Jesovnik and Schultz (2017) - Large species; posterior cephalic emargination deep, gradually impressed; mandible dorsally smooth and glossy; frontal lobe triangular, relatively narrow; mesosomal tubercles low; first gastral tergite with lateral carinae weak, dorsal carinae absent.
Based on morphology, S. lutzi seems to be most closely related to Sericomyrmex mayri, but mayri has a much shallower posterior cephalic emargination; striate mandibles; narrower, anterad-directed frontal lobes; and often at least faint anteromedian dorsal gastral carinae.
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 5.3344° to -1.9075°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: French Guiana, Guyana (type locality), Suriname.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
|
Castes
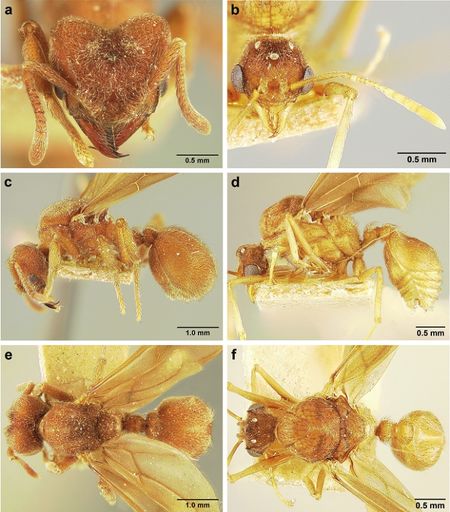
Jesovnik and Schultz 2017. Figure 29. S. lutzi worker (USNMENT00445053), SEM images. a Head, full-face view b mandibles c mesosoma and metasoma, dorsolateral view d metasoma, dorsal view. Figure 30. S. lutzi queen and male; head, lateral profile, and dorsal view. Queen (MCZ 9-11 21139) (a, c, e) Male (USNMENT01126226) (b, d, f).
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- lutzi. Sericomyrmex lutzi Wheeler, W.M. 1916c: 9 (w.q.m.) GUYANA.
- Type-material: lectotype worker (by designation of Ješovnik & Schultz, 2017a: 52), 5 paralectotype workers, 2 paralectotype queens, 1 paralectotype male.
- Type-locality: lectotype Guyana (“British Guiana”): Roraima, Kauwa Creek, 13.viii.1911, AC 3097 (A. Crampton); paralectotypes with same data.
- Type-depositories: MCZC (lectotype); AMNH, MCZC (paralectotypes).
- Status as species: Emery, 1924d: 339; Weber, 1946b: 143; Kempf, 1972a: 229; Bolton, 1995b: 382; Ješovnik & Schultz, 2017a: 52 (redescription).
- Distribution: Guyana.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Jesovnik and Schultz 2017:
Description
Worker
(lectotype): HWe 1.12–1.35 (1.21) HW 1.12–1.35 (1.21) HW1 1.08–1.27 (1.2) HW2 1.21–1.46 (1.32) HW3 0.86–0.90 (0.86) IFW1 0.70–0.88 (0.78) IFW2 0.27–0.33 (0.32) HL1 1.16–1.30 (1.16) HL2 0.96–1.12 (0.96) SL 0.80–0.93 (0.86) EL 0.14–0.19 (0.16) Om 9–11 (11) WL 1.38–1.68 (1.55) PL 0.3–0.44 (0.3) PPL 0.22–0.26 (0.26) GL 0.9–1.18 (0.99) HFL 1.20–1.40 (1.33) PW 0.73–0.94 (0.83) CI 100–104 (104) FLI 63–68 (65) SI 67–73 (71) OI 12–15 (13) CEI 14–17 (17) [N=7]
Pilosity. Pubescence dense, lighter than integument, appressed to decumbent. Hairs curved, darker in color at base, yellow to gray, appressed to suberect, mostly decumbent.
Head. Head in full-face view slightly broader than long (CI=103 ± 2), posterior corner acute, lateral margin of head convex, posterior cephalic emargination distinct, very deep (CEI=15 ± 1), gradually impressed. Vertexal impression relatively deep, frontal tumuli distinct. Mandibles with 7–9 teeth, dorsally smooth, glossy, finely transversely striate along masticatory margin. Frontal carina straight to slightly curved laterally, complete. Eyes medium-sized (OI =14 ± 1), weakly convex, 9–11 ommatidia across largest diameter. Frontal lobe triangular, relatively narrow (FLI=64 ± 2), posterior margin shorter than medial. Antennal relatively short, scape not reaching posterior cephalic corner (SI=71 ± 2).
Mesosoma. Mesosomal tubercles low and obtuse. Propodeal carinae low, reduced, sometimes with posterodorsal denticles.
Metasoma. Petiole and postpetiole each with two low, short, serrate carina dorsally, on petiole sometimes reduced to low denticles, best seen in dorsolateral view. First gastral tergite with lateral carinae weakly developed, dorsal carinae absent.
Queen
HWe 1.4 HW 1.4 HW1 1.32 HW2 1.52 HW3 IFW1 0.9 IFW2 0.34 HL1 1.36 HL2 1.16 SL 0.92 EL 0.25 Om 18 EW 0.1 WL 2.08 PL 0.44 PPL 0.28 GL 1.72 HFL 1.56 PW 1.18 1 FWg 6.82 HWg 4.6 CI 103 FLI 64 SI 66 OI 18 [N=1]
Head. Mandibles with nine teeth, dorsally glossy and smooth, finely transversely striate only along masticatory margin. Preocular carina fading posterior to eye. Eye large, convex, 18 ommatidia across largest diameter. Frontal lobe as in worker, antennal scape not reaching posterior cephalic corner.
Mesosoma. Scutum in dorsal view with notauli and median mesoscutal line reduced. Parapsidal lines faint, slightly curved. Scutellum in dorsal view narrowing posteriorly, posterior notch shallow. Propodeum with two low, reduced denticles.
Metasoma. First tergite of gaster with lateral carinae strongly developed, dorsal carinae absent, anteromedian groove distinct in dorsal view.
Male
Measurements in mm: HWe 0.9 HW 0.74 IFW1 0.27 IFW2 017 HL1 0.7 SL 0.74 EL 0.29 Om 30 EW 0.13 WL 1.74 PL 0.4 PPL 0.22 GL 1.4 HFL 1.67 PW 0.80 IOD 0.58 FWg 5.21 HWg 3.55 CI 128 FLI 30 SI 83 OI 32 [N=1]
Head in full-face view longer than broad (CI=128). Eyes large (OI=32), 30 ommatidia across largest diameter. Preocular carina extending posterior to median ocellus, medially curved before fading. In dorsal view scutum with notauli well developed, mesoscutal line distinct, with dark-brown reticulation, best seen in frontodorsal view. Groove between axillae with one transverse, short costa medially. Propodeum devoid of protuberances or carinae except for spiracular tubercles. Petiole with weak lateral carinae; postpetiole simple, smooth, without denticles or carinae.
MISC S. lutzi was originally described from a handful of specimens collected on Mt. Roraima, a mountain plateau (tepui) on the border of Guyana, Venezuela, and Brazil. The only other specimens of this species were collected on the slopes of the eastern-most tepui in Guyana, Mt. Ayanganna. It is very likely that this species has a restricted distribution and is endemic to the tepuis of the Guiana Highlands, an area known for its endemic flora and fauna (Brown 1975, Steyermark 1987, Berry et al. 1995, LaPolla et al. 2007).
Type Material
Lectotype worker (here designated): GUYANA, Roraima, Kauwa creek [5.223, -60.73], 13 Aug 1911, A. Crampton, AC3097 (Museum of Comparative Zoology: 1w, MCZ 12-14 21139, topmost worker on the pin). Paralectotypes: same data as lectotype (MCZ: 2w, MCZ 12-14 21139, lower two specimens on the pin) (MCZ: 2w, 1q, MCZ 9-11 21139) (American Museum of Natural History: 1w, 1q, 1m, USNMENT01126226).
References
- Franco, W., Ladino, N., Delabie, J.H.C., Dejean, A., Orivel, J., Fichaux, M., Groc, S., Leponce, M., Feitosa, R.M. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674, 509–543 (doi:10.11646/zootaxa.4674.5.2).
- Ješovnik, A. and Schultz, T.R. 2017. Revision of the fungus-farming ant genus Sericomyrmex Mayr (Hymenoptera, Formicidae, Myrmicinae). ZooKeys. 670:1–109. doi:10.3897/zookeys.670.11839
- Jesovnik, A., Sosa-Calvo, J., Lloyd, M.W., Branstetter, M.G., Andez. F.F., Schultz, T.R. 2017. Phylogenomic species delimitation and host-symbiont coevolution in the fungus-farming ant genus Sericomyrmex Mayr (Hymenoptera: Formicidae): ultraconserved elements (UCEs) resolve a recent radiation. Systematic Entomology 42, 523–542 (doi:10.1111/syen.12228).
- Wheeler, W. M. 1916c. Ants collected in British Guiana by the expedition of the American Museum of Natural History during 1911. Bull. Am. Mus. Nat. Hist. 35: 1-14 (page 9, worker, queen, male described)
References based on Global Ant Biodiversity Informatics
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Franco W., N. Ladino, J. H. C. Delabie, A. Dejean, J. Orivel, M. Fichaux, S. Groc, M. Leponce, and R. M. Feitosa. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674(5): 509-543.
- Jesovnik A., J. Chaul, and T. Schultz. 2018. Natural history and nest architecture of the fungus-farming ant genus Sericomyrmex (Hymenoptera: Formicidae). Myrmecological News 26: 65-80.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Klingenberg, C. and C.R.F. Brandao. 2005. The type specimens of fungus growing ants, Attini (Hymenoptera, Formicidae, Myrmicinae) deposited in the Museu de Zoologia da Universidade de Sao Paulo, Brazil. Papeis Avulsos de Zoologia 45(4):41-50
- Weber N. A. 1946. The biology of the fungus-growing ants. Part IX. The British Guiana species. Revista de Entomologia (Rio de Janeiro) 17: 114-172.