Ponera swezeyi
| Ponera swezeyi | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Ponera |
| Species: | P. swezeyi |
| Binomial name | |
| Ponera swezeyi (Wheeler, W.M., 1933) | |
| Common Name | |
|---|---|
| Ogasawara-hari-ari | |
| Language: | Japanese |
Known from the floor of broad-leaf forest in the Ogasawara Islands, Japan (Japanese Ant Image Database).
| At a Glance | • Invasive |
Identification
Taylor (1967) - Easily recognized by the combination of small size (worker head width 0.29-0.33 mm, Sf HW 0.32-0.33 mm) with the characters of the japonica species group (5-segmented antennal club, and incised mesometanotal suture). In addition, the queen wing venation (and presumably that of the male) is peculiar. The 2nd abscissa of Rs + M arises distal to the anterior base of the medio-cubital crossvein (m-cu) (fig. 8). This character occurs in only one other Ponera species, the much larger Ponera chapmani (Philippine Islands).
P. swezeyi is the only known Ponera occurring in Hawaii. In Samoa it is easily distinguished from its congeners by its smaller size and light coloration.
Only 3 other species (Ponera petila, Ponera szaboi and Ponera szentivanyi) are known to have HW values less than 0.34 mm; they all have 4-segmented antennal clubs, and lack the dorsal mesometanotal suture.
Leong et al. (2019): Ponera swezeyi belongs to the P. japonica species group defined by Taylor (1967), which includes Ponera japonica, Ponera incerta and Ponera swezeyi. These three species have a five segmented antennal club, yellow to brown body color and are widely distributed within the Sino-Japanese, Oriental and Oceanian regions. Recently, three Oriental species, Ponera baka Xu, 2001 and Ponera xantha Xu, 2001 from Yunnan; Ponera shennong Terayama, 2009 from Taiwan were discovered, and show important similarities with P. swezeyi (yellowish brown body color, five segmented antennal club, small body size and thick petiolar node). Here, our morphological comparison suggests that P. swezeyi is similar to P. baka, P. shennong and P. xantha, instead of P. japonica and P. incerta for the following reasons:
- Head width in P. baka (HW: 0.33 mm), P. xantha (HW: 0.35 mm), P. shennong (HW: 0.28 mm) and P. swezeyi (HW: 0.32–0.33 mm) is distinctly smaller than P. incerta (HW: 0.36–0.40 mm) and P. japonica (HW: 0.41–0.50 mm).
- Petiolar node in lateral view very thick in P. baka, P. shennong, P. swezeyi and P. xantha, but moderately thick in P. incerta and P. japonica.
- Antennal scape short in P. baka, P. shennong, P. swezeyi and P. xantha (when laid backward, with a remaining distance of more than 30% of the scape length to the posterolateral corner), but relatively long in P. incerta and P. japonica (when laid backward, with a remaining distance of less than 15% of the scape length to the posterolateral corner).
- Body color yellowish in P. baka, P. shennong, P. swezeyi and P. xantha, but brown in P. incerta and P. japonica.
These morphological similarities suggest that P. swezeyi might be closer to the Oriental species than to P. japonica and P. incerta. Although the reported distribution of P. swezeyi is wide, this species may be native to the Oriental region.
Csősz et al. (2023) - In the Malagasy region, this species can only be confused with Ponera petila, but petiole characters help distinguish these two species: in P. swezeyi, the anterolateral and posterior surfaces of petiolar node meet in sharp vertical ridges, while the lateral and posterior petiolar surfaces of P. petila meet in a rounded transition. Size of workers may also provide clues for identification: head width (CWb) of P. swezeyi is smaller 306 µm [291, 323]) than that of P. petila (333 µm [323, 339]). Worldwide, P. swezeyi is the smallest species within the tenuis group. Size variance detected in a relatively large pool (n = 105) of P. swezeyi workers was found negligible (CWb: 291 µm to 323 µm) allowing for separation of P. swezeyi workers from that of other species. Only one species, Ponera szaboi overlapping with P. swezeyi (CWb: 306 µm [291, 323]), but the petiole of P. swezeyi species is considerably wider (PNI: 77–83) than that of P. szaboi (PNI: 61–65, see Taylor, 1967).
Distribution
Fiji, Hawaii and Samoa. P. swezeyi had been recorded only from Hawaii, and was thought by Wilson (1957) to have probably been introduced there by man. This opinion has been supported by discovery of the species on Samoa, where it is widespread on all 3 main islands. (Taylor 1967). More recently P. swezeyi has been collected in Fiji (Sarnat and Economo 2012). It is also found in Tanzania.
Csősz et al. (2023) - Ponera swezeyi is known to occur in the eastern coast of Madagascar, Comoros, Mauritius, Mayotte, Reunion, and Seychelles in the Malagasy region. Moreover, populations are known from Queensland (northeastern Australia), Hawaii, Christmas Island, Japan (Fiji Island), Hawaii, Japan (Ogasawara Islands), Samoa, Solomon Islands, and Tanzania (Fisher & Bolton, 2016; Leong et al., 2019).
Keys including this Species
Latitudinal Distribution Pattern
Latitudinal Range: -13.583° to -17.3°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Afrotropical Region: Comoros, Mozambique, United Republic of Tanzania.
Australasian Region: Australia.
Indo-Australian Region: Christmas Island, Hawaii, Samoa, Singapore, Solomon Islands, Tokelau.
Malagasy Region: Madagascar, Mauritius, Mayotte, Réunion, Seychelles.
Palaearctic Region: Japan.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Taylor (1967) - The only Samoan nest records are those from Le Mafa, Upolu. Several colonies were found at this site, about 1 .5 meters above ground level, in an almost vertical clay road cutting, in an area of rather disturbed rain forest. P. swezeyi appears to differ in nesting preferences from the other small Samoan species (Ponera incerta and Ponera tenuis), colonies of which were almost always taken in rotting logs, or under moss on logs or trees. Occasional swezeyi workers were taken in moss berlesates but no nests were found in such sites, and the only sexuals taken were from soil/leaf-mold samples. This apparent preference for soil nesting may have been important in the distributional history of swezeyi, which would thus readily be carried by Polynesian and European man in soil attached to root crops such as taro or kumara, sugar cane or pineapple plants, etc. Recall that most Hawaiian records are from soil in pineapple plantations.
One wonders whether swezeyi is a Samoan autochthon secondarily introduced into Hawaii, or whether it has reached these island groups from a third source area, presumably in E. Melanesia. The new records from primary rain forest at high elevations on Upolu and Savaii could indicate that it is a Samoan autochthon. However, the Samoan fauna contains numerous introduced species, including some not usually considered tramps, though clearly carried to the islands by man. Most of the latter are, like P. swezeyi, cryptobiotic or soil-nesting forms, which could easily have been transported in soil attached to plant roots.
Sarnot and Economo (2012) - Several collections of P. swezeyi were made on Koro from litter sifting, and it was collected in a wet log from Taveuni. An alate queen was captured from a malaise trap in Sigatoka (Viti Levu).
Castes
Worker
Queen
Images from AntWeb
   
| |
| Cotype of Ponera swezeyi. Queen (alate/dealate). Specimen code casent0173002. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MCZ, Cambridge, MA, USA. |
Phylogeny
| Ponera |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Relationships among selected species of Ponera based on Branstetter & Longino (2019). The focus of this study was the species Ponera exotica and Ponera pennsylvanica.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- swezeyi. Pseudocryptopone swezeyi Wheeler, W.M. 1933g: 16, fig. 6 (w.q.) HAWAII. Combination in Ponera: Wilson, 1957b: 370. See also: Taylor, 1967a: 85.
Type Material
- Syntypes Hawaii, USA: CASENT0249111, Is. Oahu, R. H. Van Zwaluwenburg, (1w, BPBM, CASENT0249111) [examined by Csősz et al., 2023]; MCZ-ENT00593416, Is. Oahu, R. H. Van Zwaluwenburg, (3w, MCZC, MCZ-ENT00593416) [examined by Csősz et al., 2023].
Description
Worker
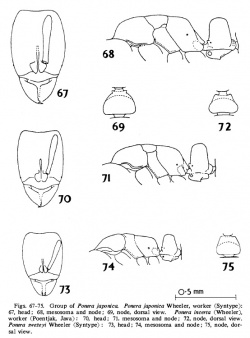
Wilson (1957) -HW 0.29-0.30 mm, HL 0.38-0.41 mm, SL 0.25-0.2 6 mm, CI 72-78, 81 84-85, PW 0.21-0.24 mm, petiole height (single measurement) 0.24 mm, dorsal petiole width 0.18 mm. Mandible with three distinct apical teeth occupying slightly less than half the masticatory border; the remainder of the border bearing an indeterminate number of minute denticles. Antennal club distinctly 5-jointed. Petiolar node seen from directly above forming slightly more than a half-circle. Subpetiolar process well developed, its apex right-angular.
Sculpturing about as in P. caledonica except that on the alitrunk only the declivitous faces of the propodeum are smooth and shining, the remainder of the. alitruncal surfaces being lightly shagreened and only feebly shining.
Body concolorous light brownish yellow; appendages clear yellow.
Taylor (1967) - Wilson's (1957) measurements and indices require modification because of additional material: HW 0.29-0.33 mm; HL 0.38-0.44 mm; SL 0.25-0.28 mm; CI 72-78 mm; SI 84-85; PW 0.21-0.24 mm; PH 0.24-0.26 mm; DPW 0.18-0.19 mm. There is no apparently significant structural variation between the Hawaiian and Samoan material.
Wheeler (1933) stated that eyes were not present in swezeyi workers; an opinion accepted by Wilson (1957). Examination of Samoan specimens and re-examination of the types, indicates that eyes are developed in this species. They consist of minute single facets, lying about 0.84-0.86 X the distance from the lateral occipital border to the midpoint of the anterior genal border. They are distinctly pigmented in fresh specimens, but fade on drying. Palpal formula: Maxillary 2: Labial 2: (2 Upolu specimens dissected). Mesometanotal suture finely but distinctly incised on mesosomal dorsum.
Taylor (1967) - [Notes based on 2 Hawaiian specimens (including a syntype), and 4 Samoan ones, 3 from Upolu, 1 from Tutuila]: HL 0.42-0.43 mm; HW 0.32-0.33 mm; SL 0.26-0.28 mm; CI 76-78; SI 81-85; PW 0.27-0.28 mm; PNL 0.13-0.14 mm; PH 0.25-0.28 mm; DPW 0.19-0.20 mm; PNI 71-74; maximum diameter of compound eye 0.09 mm; ocular index 27-28; palpal formula: Maxillary 2: Labial 2 (1 Samoan specimen dissected). Agreeing with the generic plan for this caste, and differing from workers in the usual characters. Wing venation peculiar and diagnostic (see identification section above).
References
- Csősz, S., Loss, A. C., Fisher, B. L. 2023. Exploring the diversity of the Malagasy Ponera (Hymenoptera: Formicidae) fauna via integrative taxonomy. Organisms Diversity & Evolution (doi:10.1007/s13127-023-00610-1).
- Leong, C.-M., Guenard, B., Shiao, S.-F., Lin, C.-C. 2019. Taxonomic revision of the genus Ponera Latreille, 1804 (Hymenoptera: Formicidae) of Taiwan and Japan, with a key to East Asian species. Zootaxa 4594 (1): 1–86 (DOI 10.11646/zootaxa.4594.1.1).
- Sarnat, E. M.; Economo, E. P. 2012. The ants of Fiji. University of California Publications in Entomology 132:1-384. PDF
- Taylor, R. W. 1967a. A monographic revision of the ant genus Ponera Latreille (Hymenoptera: Formicidae). Pac. Insects Monogr. 13: 1-112 (page 85, see also)
- Terayama, M. 1996. Taxonomic studies on the Japanese Formicidae, part 2. Seven genera of Ponerinae, Cerapachyinae and Myrmicinae. Nature and Human Activities. 1:9-32.
- Wang, W.Y., Soh, E.J.Y., Yong, G.W.J., Wong, M.K.L., Benoit Guénard, Economo, E.P., Yamane, S. 2022. Remarkable diversity in a little red dot: a comprehensive checklist of known ant species in Singapore (Hymenoptera: Formicidae) with notes on ecology and taxonomy. Asian Myrmecology 15: e015006 (doi:10.20362/am.015006).
- Wheeler, W. M. 1933g. Three obscure genera of ponerine ants. American Museum Novitates 672: 1-23 (page 16, fig. 6 worker, queen described)
- Wilson, E. O. 1957b. The tenuis and selenophora groups of the ant genus Ponera (Hymenoptera: Formicidae). Bulletin of the Museum of Comparative Zoology 116: 355-386 (page 370, Combination in Ponera)
References based on Global Ant Biodiversity Informatics
- Abbott, K.L., M. Sarty and P.J. Lester. 2006. The ants of Tokelau. New Zealand Journal of Zoology. 33:157-164.
- Dlussky G.M. 1994. Zoogeography of southwestern Oceania. Zhivotnoe naselenie ostrovov Iugo-Zapadnoi Okeanii ekologo-geograficheskie issledovanii 48-93.
- Kami K.S., and S. E. Miller. 1998. Samoan insects and related arthropods: checklist and bibliography. Bishop Museum Technical Report 13, pp 121.
- Kami KS & Miller SE. 1998. Samoan insects and related arthropods: checklist and bibliography. Bishop Museum Technical Report No. 13.
- Peck R. W., P. C. Banko, F. Pendleton, M. A. Schmaedick, and K. Ernsberger. 2014. Arthropods of Rose Atoll with special reference to ants and Pulvinaria urbicola scales (Hemiptera: Coccidae) on Pisonia grandis trees. Technical Report HCSU-057, 22 pages.
- Sarnat Eli M. 2009. The Ants [Hymenoptera: Formicdiae] of Fiji: Systematics, Biogeography and Conservation of an Island Arc Fauna. 80-252
- Taylor R. W. 1967. A monographic revision of the ant genus Ponera Latreille (Hymenoptera: Formicidae). Pacific Insects Monograph 13: 1-112.
- Wetterer, James K. and Vargo, Donald Vargo L. 2003. Ants (Hymenoptera: Formicidae) of Samoa. Pacific Science. 57(4):409-419.
- Wilson E. O.; Taylor, R. W. 1967. The ants of Polynesia (Hymenoptera: Formicidae). Pacific Insects Monograph 14:1-109.
- Wilson EO, Hunt GL. 1967. Ant fauna of Futuna and Wallis Islands, stepping stones to Polynesia. Pacific Insects 9.4: 563-584.
- Wilson EO, Taylor RW. 1967. The ants of Polynesia. Pacific Insects Monograph 14:1-109.
- Wilson, Edward O. and George L. Hunt. 1967. Ant Fauna of Futuna and Wallis Islands, Stepping Stones To Polynesia. Pacific Insects. 9(4):563-584.
- Wilson, Edward O. and Hunt, George L. Jr. 1967. Ant Fauna of Futuna and Wallis Islands, Stepping Stones to Polynesia. Pacific Insects. 9(4):563-584