Azteca schimperi
| Azteca schimperi | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Dolichoderinae |
| Tribe: | Leptomyrmecini |
| Genus: | Azteca |
| Species group: | aurita |
| Species: | A. schimperi |
| Binomial name | |
| Azteca schimperi Emery, 1893 | |
| Synonyms | |
| |
This species makes large external carton nests. I have observed four nests, three in Costa Rica and one in Panama, and all have been in Cecropia trees (C. peltata, C. obtusifolia, and C. longipes). Benson (1985) also observed A. schimperi in Cecropia trees. However Forel (1908b), describing the queen and male from a collection from Bahia, Brazil, explicitly stated that the carton nest was perched on the branches of a tree that was not Cecropia. (Longino 2007)
Identification
A member of the Azteca aurita group. See the group page and keys for identification.
Longino (2007) - A relatively distinctive species with little discernible variation over its range; specimens from Argentina and Paraguay look very like specimens from Central America and Mexico. Workers possess a middle and posterior tibia without an apical spur, mandible with typical thickness at base, concave masticatory edge curved towards an enlarged apical tooth, apical tooth much larger than penultimate tooth, medial clypeal lobe strongly convex and protruding, extending far beyond the lateral clypeal lobes, head with convex sides, and strongly cordate posterior margin.
Keys including this Species
- Key to Azteca aurita group queens
- Key to Azteca aurita group workers
- Key to Costa Rica Azteca queens
- Key to Costa Rica Azteca workers
Distribution
Mexico to Argentina. Azteca schimperi has a very broad range, vying with Azteca alfari for the Azteca species with the largest range.
Latitudinal Distribution Pattern
Latitudinal Range: 19.933° to -25.266667°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Argentina, Belize, Brazil, Colombia, Costa Rica (type locality), French Guiana, Guatemala, Guyana, Mexico, Nicaragua, Panama, Paraguay, Peru, Suriname.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Longino (2007) - The three nests I have seen in Costa Rica were all at the edges of roads in highly human altered landscapes (pasture edges, coffee farms) in seasonally dry areas. The globular carton nests are very conspicuous on the highly visible branches of Cecropia trees, and in my studies of Cecropia in Costa Rica I always looked for them. Yet I have only seen these three over many years of work in Costa Rica. I have never been able to examine a nest closely, but I have been able to observe the base of the trees in which they occurred. In one case workers were foraging not only on the base of the tree but also on surrounding low vegetation. In another case, on a tree with stump sprouts at the base, workers were swarming over the sprouts but not actually entering the internodes. These observations contrast with typical Cecropia ants, which never forage off the host plant and usually enter and occupy any sprouts at the base of the tree. Thus A. schimperi seems to occupy Cecropia trees more “lightly” than the dominant obligate Cecropia ants.
A. schimperi could be a temporary social parasite of Cecropia ants, usurping an existing Azteca colony as a means of establishing its own. Such a scenario might explain the somewhat less specialized use of Cecropia by A. schimperi; it may be a specialist on Cecropia ants rather than Cecropia trees.
Guerreo et al. (2010) - Until now, Tapinoma nigerrimum (Nylander) is the only species in the Dolichoderinae subfamily for which winged or stumped workers have been reported (Scupola 2008), something that is always considered to be a characteristic of gynes. Unexpectedly, the examination of the Azteca aurita group material coming from MZSP allowed us to observe a major worker of Azteca schimperi coming from Costa Rica with all of the morphological characters of a worker except for its vestigial wings. This worker presents some characteristics that differentiate it from normal workers: the head is a little larger than the normal major workers from the same species (HLA: 1.56 vs. 1.16–1.51 mm), the median ocellus a little more developed and similar to that of the gynes of A. schimperi, and there is the presence of wing rudiments on the mesosoma. The elements that determine these kinds of morphological anomalies are generally considered to be environmental factors, as these are essential for the determination and development of the casts (Scupola 2008); these environmental factors, which have a decisive function in the formation of a hybrid phenotype, are poor nutrition and different chemicals (Heinze 1998). Nevertheless, simple genetic accidents, viruses or parasites can also provoke the development of abnormalities. This is the first report of the intercast syndrome in the genus Azteca and the second for the Dolichoderinae subfamily, after the results reported by Scupola (2008) for Tapinoma nigerrimum.
De Oliveira et al. (2015), studying ant occupancy of Cecropia trees in southwest Bahia, Brazil, found a colony of Azteca schimperi opportunistically nesting in a Cecropia pachystachya tree.
Vanoye-Eligio et al. (2020) provide the following notes based on collections using Multilure-type traps from northeastern Mexico: A total of 4 workers and a queen were collected. Nests of this species are difficult to detect because of its arboreal habit, as documented by Longino (2007), who reported 3 nests of A. schimperi on Cecropia (Urticaceae) after several years of sampling in Costa Rica. It forms large nests constructed from gnawed plant material and is reported elsewhere nesting in trees of the genus Cecropia (Benson 1985). Visual searches near the location of the traps indicated that their foraging territories were not limited to the host tree, but extended into the surrounding undergrowth. Additional searching detected A. schimperi nesting in cavities of broken or rotten branches of mango trees (Mangifera indica L.; Anacardiaceae), which are common components of the tropical forest in the region.
Castes
Male
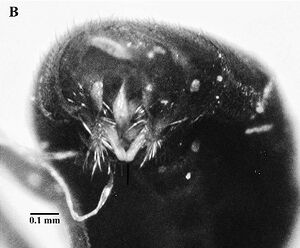
| |
| . | |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- schimperi. Azteca schimperi Emery, 1893b: 140, pl. 2, figs. 43-46 (w.) COSTA RICA.
- Type-material: syntype workers (number not stated).
- [Note: Shattuck, 1994: 25, cites 8w syntypes (2 MCZC, 6 MHNG).]
- Type-locality: Costa Rica: Alajuela (A. Alfaro).
- Type-depositories: MCZC, MHNG, MSNG.
- Forel, 1908c: 387 (q.m.).
- Status as species: Forel, 1899c: 110; Wheeler, W.M. 1907a: 276; Forel, 1908c: 387; Emery, 1913a: 34; Crawley, 1916b: 375; Wheeler, W.M. & Bequaert, 1929: 33; Kempf, 1972a: 34; Shattuck, 1994: 25; Bolton, 1995b: 79; Wild, 2007b: 24; Longino, 2007: 58 (redescription); Guerrero, et al. 2010: 53 (in key); Branstetter & Sáenz, 2012: 253; Guerrero, 2019: 706.
- Senior synonym of clariceps: Longino, 2007: 58.
- Senior synonym of fiebrigi: Longino, 2007: 58.
- Senior synonym of pallida: Longino, 2007: 59.
- Distribution: Argentina, Belize, Brazil, Colombia, Costa Rica, French Guiana, Guatemala, Guyana, Mexico, Nicaragua, Panama, Paraguay, Suriname.
- clariceps. Azteca lanuginosa var. clariceps Santschi, 1933e: 121 (w.) ARGENTINA (Formosa).
- Type-material: syntype workers (number not stated).
- [Note: Baroni Urbani, 1977e: 80, and Shattuck, 1994: 20, cite 11w syntypes NHMB.]
- Type-locality: Argentina: Formosa; Pilcomayo, Tacaaglé, no. 1954 (J. Zurflüh).
- Type-depository: NHMB.
- Subspecies of lanuginosa: Kempf, 1972a: 33; Brandão, 1991: 329; Shattuck, 1994: 20; Bolton, 1995b: 78.
- Junior synonym of schimperi: Longino, 2007: 58.
- fiebrigi Forel, 1909a: 261 (w.q.m.) PARAGUAY.
- Type-material: syntype workers, syntype queens, syntype males (numbers not stated).
- [Notes (i): Baroni Urbani, 1977e: 80, cites 2w syntypes NHMB; (ii) Shattuck, 1994: 17, cites 13w, 3q, 1m syntypes (2w MCZC, 9w, 3q, 1m MHNG, 2w NHMB).]
- Type-locality: Paraguay: San Bernardino (K.A.G. Fiebrig).
- Type-depositories: MCZC, MHNG, NHMB.
- Status as species: Emery, 1913a: 33; Santschi, 1916e: 391; Kempf, 1972a: 31; Shattuck, 1994: 17; Bolton, 1995b: 78.
- Junior synonym of schimperi: Longino, 2007: 58.
- pallida. Azteca muelleri var. pallida Stitz, 1937: 135 (w.) MEXICO (Veracruz).
- Type-material: 6 syntype workers.
- Type-locality: Mexico: Misantla (Gugelmann).
- Type-depository: MNHU.
- Subspecies of muelleri: Kempf, 1972a: 33; Shattuck, 1994: 23; Bolton, 1995b: 79.
- Junior synonym of schimperi: Longino, 2007: 59.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Longino (2007) - (n=4): HLA 1.42 (1.16–1.51), HW 1.41 (1.18–1.45), SL 0.94 (0.85–1.01), CI 99 (96–102), SI 68 (64–73).
Palpal formula 4,3; middle and hind tibia lacking apical spur; mandible of typical thickness at base, with microareolate sculpture, dull, becoming smooth and shiny, somewhat flattened toward masticatory margin; masticatory margin concave, curving toward enlarged apical tooth, apical tooth much larger than penultimate tooth; medial clypeal lobe strongly convex and protruding, extending well beyond lateral clypeal lobes; head with convex sides, strongly cordate posterior margin; in lateral profile promesonotum forming single convexity, evenly convex to middle of mesonotum, posterior mesonotum drops steeply to depressed basal face of propodeum; petiolar node somewhat pillow-shaped, blunt and weakly bilobed apically, posteroventral lobe shallow, weakly convex from front to back, ending posteriorly in a somewhat abrupt shelf, rising steeply to tergosternal suture, leaving distinct posterior rim on sternite, laterally very strongly compressed, forming sharp median carina (this character less developed on South American material); scape, entire head capsule, mesosoma, legs, petiolar node, and gastral dorsum with appressed pubescence but completely lacking erect setae; color clear yellow orange to red brown, face shining.
Queen
Longino (2007) - (n=3 queens from Colombia, Brazil, and Paraguay): HLA 1.44 (1.40–1.47), HW 1.05 (1.04–1.06), SL 0.97 (0.94–1.01), CI 72 (71–76), SI 69 (65–69).
Palpal formula 4,3; middle and hind tibia lacking apical spur; apical tooth of mandible about twice as long as penultimate tooth, dorsal surface shiny, smooth, with sparse piligerous puncta; medial clypeal lobe strongly convex and protruding, extending well beyond lateral clypeal lobes; head rectangular, sides parallel and flat, posterolateral margins subangular but rounded, posterior margin a moderately developed V-shaped impression; petiolar node triangular; posteroventral petiolar lobe developed, moderately convex from front to back, laterally compressed and tectiform; scape, sides and posterior margin of head with dense vestiture of short, erect setae; femora with similar setae but erect setae nearly absent from outer margins of middle and hind tibia; pronotum and mesoscutum with these short setae very sparse, more abundant on scutellum and propodeum; petiolar node and ventral margin of petiole with abundant short setae; gaster devoid of erect setae; entire body red brown, with smooth, highly polished and reflective surface.
Type Material
Longino (2007) - Syntype workers: Costa Rica, Alajuela [10°01'N, 84°13'W, 1100m] (Alfaro) Museo Civico di Storia Naturale, Genoa, Museum of Comparative Zoology, Musee d'Histoire Naturelle Genève (examined).
References
- de Oliveira, G. V., M. M. Correa, I. M. A. Goes, A. F. P. Machado, R. J. de Sa-Neto, and J. H. C. Delabie. 2015. Interactions between Cecropia (Urticaceae) and ants (Hymenoptera: Formicidae) along a longitudinal east-west transect in the Brazilian Northeast. Annales De La Societe Entomologique De France. 51:153-160. doi:10.1080/00379271.2015.1061231
- Emery, C. 1893l. Studio monografico sul genere Azteca Forel. Mem. R. Accad. Sci. Ist. Bologna (5)3:119-152 (page 140, pl. 2, figs. 43-46 soldier, worker described)
- Forel, A. 1908h. Ameisen aus Sao Paulo (Brasilien), Paraguay etc. gesammelt von Prof. Herm. v. Ihering, Dr. Lutz, Dr. Fiebrig, etc. Verh. K-K. Zool.-Bot. Ges. Wien 58: 340-418 (page 387, queen, male described)
- Franco, W., Ladino, N., Delabie, J.H.C., Dejean, A., Orivel, J., Fichaux, M., Groc, S., Leponce, M., Feitosa, R.M. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674, 509–543 (doi:10.11646/zootaxa.4674.5.2).
- Longino, J.T. 2007. A taxonomic review of the genus Azteca in Costa Rica and a global revision of the aurita group. Zootaxa. 1491:1-63.
- Vanoye-Eligio, V., Berrones-Morales, M., Rosas-Mejía, M. 2020. Myrmecological fauna (Hymenoptera: Formicidae) collected with Multilure-type traps in a Neotropical transitional region from northeastern Mexico. Florida Entomologist 103: 127-129.
References based on Global Ant Biodiversity Informatics
- Baroni Urbani C. 1977. Katalog der Typen von Formicidae (Hymenoptera) der Sammlung des Naturhistorischen Museums Basel (2. Teil). Mitt. Entomol. Ges. Basel (n.s.) 27: 61-102.
- Branstetter M. G. and L. Sáenz. 2012. Las hormigas (Hymenoptera: Formicidae) de Guatemala. Pp. 221-268 in: Cano E. B. and J. C. Schuster. (eds.) 2012. Biodiversidad de Guatemala. Volumen 2. Guatemala: Universidad del Valle de Guatemala, iv + 328 pp
- Crawley W. C. 1916. Ants from British Guiana. Ann. Mag. Nat. Hist. 8(17): 366-378.
- Emery C. 1893. Studio monografico sul genere Azteca Forel. Memorie della Reale Accademia delle Scienze dell'Istituto di Bologna (5)3: 119-152
- Emery C. 1896. Studi sulle formiche della fauna neotropica. XVII-XXV. Bullettino della Società Entomologica Italiana 28: 33-107.
- Emery C. 1913. Hymenoptera. Fam. Formicidae. Subfam. Dolichoderinae. Genera Insectorum 137: 1-50.
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Forel A. 1909. Ameisen aus Guatemala usw., Paraguay und Argentinien (Hym.). Deutsche Entomologische Zeitschrift 1909: 239-269.
- Franco W., N. Ladino, J. H. C. Delabie, A. Dejean, J. Orivel, M. Fichaux, S. Groc, M. Leponce, and R. M. Feitosa. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674(5): 509-543.
- Guerrero R. J., J. H. C. Delabie, and A. Dejean. 2010. Taxonomic contribution to the aurita group of the ant genus Azteca (Formicidae: Dolichoderinae). Journal of Hymenoptera Research 19(1): 51-65.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Longino J. T. 2007. A taxonomic review of the genus Azteca (Hymenoptera: Formicidae) in Costa Rica and a global revision of the aurita group. Zootaxa 1491: 1-63
- Maes, J.-M. and W.P. MacKay. 1993. Catalogo de las hormigas (Hymenoptera: Formicidae) de Nicaragua. Revista Nicaraguense de Entomologia 23.
- Pires de Prado L., R. M. Feitosa, S. Pinzon Triana, J. A. Munoz Gutierrez, G. X. Rousseau, R. Alves Silva, G. M. Siqueira, C. L. Caldas dos Santos, F. Veras Silva, T. Sanches Ranzani da Silva, A. Casadei-Ferreira, R. Rosa da Silva, and J. Andrade-Silva. 2019. An overview of the ant fauna (Hymenoptera: Formicidae) of the state of Maranhao, Brazil. Pap. Avulsos Zool. 59: e20195938.
- Salinas P. J. 2010. Catalogue of the ants of the Táchira State, Venezuela, with notes on their biodiversity, biogeography and ecology (Hymenoptera: Formicidae: Amblyioponinae, Ponerinae, Proceratiinae, Myrmicinae, Ecitoninae, Formicinae, Pseudomyrmecinae, Dolichoderinae). Boletín de la SEA 47: 315-328.
- Santschi F. 1916. Formicides sudaméricains nouveaux ou peu connus. Physis (Buenos Aires). 2: 365-399.
- Vásquez-Bolaños M. 2011. Lista de especies de hormigas (Hymenoptera: Formicidae) para México. Dugesiana 18: 95-133
- Wild, A. L. "A catalogue of the ants of Paraguay (Hymenoptera: Formicidae)." Zootaxa 1622 (2007): 1-55.
- de Oliveira G. V., M. M. Correa, I. M. A. Goes, A. F. P. Machado, R. J. de Sa-Neto, and J. H. C. Delabie. 2015. Intera ctions between Cecropia (Urticaceae) and ants (Hymenoptera: Formicidae) along a longitudinal east-west transect in the Brazilian Northeast. Annales de la Société entomologique de France http://dx.doi.org/10.1080/00379271.2015.1061231

