Strumigenys godeffroyi
| Strumigenys godeffroyi | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Strumigenys |
| Species group: | godeffroyi |
| Species: | S. godeffroyi |
| Binomial name | |
| Strumigenys godeffroyi Mayr, 1866 | |
| Synonyms | |
| |
The widespread tramp species is native to the Oriental realm and was introduced to multiple states of India, Pakistan, Sri Lanka, Taiwan and numerous islands in the Indian and Pacific Oceans. Its native range is likely to include Cambodia, Laos and Thailand (Tang & Guenard, 2023).
| At a Glance | • Invasive |
Photo Gallery
Identification
Bolton (2000) - A member of the godeffroyi complex in the Strumigenys godeffroyi-group. Thirteen species in the godeffroyi-complex have predominantly smooth pleurae and side of propodeum. Seven of these, Strumigenys geminata, godeffroyi, Strumigenys juliae, Strumigenys lewisi, Strumigenys nytaxis, Strumigenys peraucta and Strumigenys uberyx, have the pair of erect hairs closest to the midline on the occipital margin short and stiff (best seen with the head in profile). This pair of hairs is erect or nearly so, and straight or at most only very shallowly evenly curved. In the other six (see under Strumigenys nanzanensis) the pair of hairs at this location are long and fine, sometimes subflagellate, and are either abruptly angled anteriorly in their apical halves or have their apical halves looped.
Of the seven listed above the first two, godeffroyi and nytaxis, do not have elongate standing hairs on the pronotum except for the humeral pair. These two are easily differentiated as follows.
1 In godeffroyi the dorsolateral margin of the head posterior to the flagellate apicoscrobal hair has a row of 3-4 stiffly anterolaterally projecting hairs that are more or less cylindrical; these contrast with the more strongly curved narrowly spatulate hairs that occur on the upper scrobe margin anterior to the flagellate hair. In nytaxis there are no stiffly projecting cylindrical hairs posterior to the flagellate hair.
2 In godeffroyi the dorsum and side of the pronotum is densely clothed with a conspicuous pelt of curved linear-spatulate ground-pilosity that gives the sclerite a vaguely furry appearance. In nytaxis pronotal ground-pilosity is sparse and inconspicuous dorsally, almost absent laterally.
3 In godeffroyi, with the propodeum in profile the lamella on the declivity terminates dorsally in a convex rim or crest of spongiform tissue that lies on top of the propodeal tooth or its vestige. In nytaxis the lamella on the declivity is flat-topped, the dorsal surface of the propodeal tooth or its vestige is not adorned with a convex crest of spongiform tissue.
4 In godeffroyi the dorsum of the petiole node is finely and densely reticulate-punctate, contrasting with the disc of the postpetiole which is mostly or entirely unsculptured. In nytaxis the dorsum of the petiole node is un sculptured or nearly so, very similar to the disc of the postpetiole.
The remaining five species, lewisi, juliae, geminata, peraucta and uberyx, are closely related and have a pair of erect hairs on the pronotal dorsum in addition to the humeral pair. In juliae and uberyx the dorsum and sides of the pronotum are mostly to entirely smooth and shining when clean; see comments under the latter name. This sclerite in lewisi, geminata and peraucta bears extensive reticulate-punctate sculpture either dorsally, laterally, or both. The Indian peraucta has cephalic ground-pilosity that is almost reclinate, each hair being so strongly curved basally that the main length of the hair-shaft parallels the surface or even inclines back down toward the surface. In both lewisi and geminata the cephalic ground-pilosity is decidedly elevated; the main length of each hair distinctly inclines upward, away from the surface, from base to apex. Finally, lewisi is a larger and more stockily built species than geminata, compare measurements and see comments under the latter name.
Sarnat and Economo (2012) - Fiji: Strumigenys godeffroyi is immediately recognizable among the Fijian dacetines by the plethora of long fine flagellate hairs that occur on all dorsal surfaces, including those of the gaster.
Keys including this Species
- Key to Australian Strumigenys Species
- Key to Micronesian Ants
- Key to Strumigenys of India
- Key to Strumigenys of East Asia
- Key to US Strumigenys species
Distribution
Native: India (Meghalaya, West Bengal, Sikkim), Indonesia (Borneo, Java, Lesser Sunda Islands, Sumatra), Malaysia, Myanmar, Philippines, Singapore, Vietnam (Tang & Guenard, 2023).
Latitudinal Distribution Pattern
Latitudinal Range: 5.016666667° to -20.992°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Australasian Region: Australia, New Caledonia.
Indo-Australian Region: Borneo, Fiji, Guam, Hawaii, Indonesia, Krakatau Islands, Malaysia, Marshall Islands, Micronesia (Federated States of), New Guinea, Niue, Northern Mariana Islands, Palau, Philippines, Samoa (type locality), Singapore, Solomon Islands, Tokelau, Tonga, Vanuatu, Wallis and Futuna Islands.
Malagasy Region: Mauritius, Seychelles.
Nearctic Region: United States.
Oriental Region: India, Pakistan, Sri Lanka.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
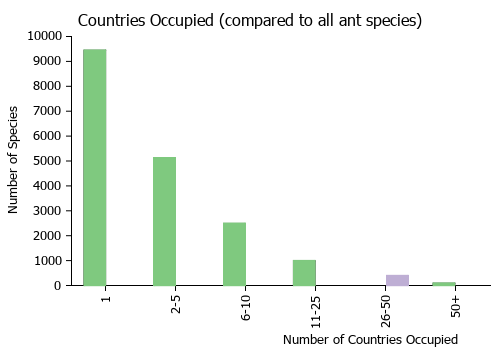
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
This ant is associated with three species of mealybugs, namely: Geococcus coffeae Green, Geococcus associatus Lit and Dysmicoccus brevipes (Cockerell). All are found on roots of Beaucarnea recurvata (Lem.) Hemi. This was also mentioned by Lit (1992) in his description of G. associatus.
Brown (1949) - This species ranges very widely, being found on many insignificant islands and coral atolls in the Pacific. It will probably be found on any really tropical shore in the central and western part of the Pacific. Since specimens have been taken in the U. S. Plant Quarantine and in other circumstances which leave no doubt that godeffroyi can and does spread through commerce as a tramp, the extent to which the species had colonized the present range through natural means, such as by floating logs and debris, may never be known. The presence of closely related species on New Zealand, Australia, and elsewhere in the Pacific indicates that the group in general is one which has been able to travel over long stretches of open sea and establish successful populations without help from man.
Castes
Worker
   
| |
| . | Owned by Museum of Comparative Zoology. |
Images from AntWeb
   
| |
| Worker. Specimen code casent0060343. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0101214. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MHNG, Geneva, Switzerland. |
   
| |
| Worker. Specimen code casent0101215. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MHNG, Geneva, Switzerland. |
Queen
Images from AntWeb
   
| |
| Queen (alate/dealate). Specimen code casent0060224. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- godeffroyi. Strumigenys godeffroyi Mayr, 1866a: 516 (w.) SAMOA. Mayr, 1887: 569 (q.); Imai, Brown, et al. 1984: 68 (k.). Senior synonym of butteli, indica: Brown, 1949d: 17; of geococci: Bolton, 2000: 791. See also: Bingham, 1903: 149.
- indica. Strumigemys godefroyi var. indica Forel, 1902c: 243 (q.) INDIA. Junior synonym of godeffroyi: Brown, 1949d: 17.
- butteli. Strumigenys godeffroyi var. butteli Forel, 1913k: 83 (w.q.) SRI LANKA. Junior synonym of godeffroyi: Brown, 1949d: 17.
- geococci. Strumigenys godeffroyi subsp. geococci Calilung, 2000: 70, fig. 3 (w.) PHILIPPINES (Luzon). Junior synonym of godeffroyi: Bolton, 2000: 791.
Type Material
- Syntype, workers, Upolu, Samoa, Naturhistorisches Museum Wien, Vienna.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
TL 2.1-2.5, HL 0.54-0.64, HW 0.40-0.45, CI 67-73, ML 0.24-0.30, MI 45-49, SL 0.33-0.40, SI 86-95, PW 0.26-0.28, AL 0.56-0.67 (20 measured).
Characters of godeffroyi-complex. Cephalic dorsum with pair of erect hairs closest to midline on occipital margin short stiff and erect, straight to shallowly evenly curved, the apical half not abruptly curved anteriorly nor looped. With head in full-face view the dorsolateral margin posterior to the flagellate apicoscrobal hair has a row of 3-4 stiffly projecting hairs. These hairs contrast with the marginal hairs anterior to the flagellate hair as they are more cylindrical (i.e. not spatulate), more elevated and less strongly curved anteriorly. Pronotum with a somewhat furry appearance: ground-pilosity on pronotal dorsum dense and conspicuous, linear-spatulate, arched and somewhat elevated, in profile appearing as a pelt; side of pronotum also densely clothed with similar ground-pilosity. Dorsum of pronotum without flagellate hairs apart from the humeral pair. Pleurae and side of propodeum mostly to entirely smooth, any reticulate-punctate sculpture present is confined to periphery. Propodeal declivity with a broad and very conspicuous spongiform lamella, the propodeal teeth only weakly expressed (may be vestigial) and entirely buried in the lamella. Dorsal surface of propodeal tooth in profile surmounted by a convex crest or ridge of spongiform tissue. Disc of postpetiole un sculptured. Basigastral costulae conspicuous but not extending half the length of the tergite.
Karyotype
- See additional details at the Ant Chromosome Database.
 Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
- 2n = 44 (Indonesia) (Imai et al., 1985).
- 2n = 40 (Malaysia) (Imai et al., 1983).
References
- Bharti, H. & Akbar, S.A. 2013. Taxonomic studies on the ant genus Strumigenys Smith, 1860 (Hymenoptera, Formicidae) with report of two new species and five new records including a tramp species from India. Sociobiology 60, 387-396 (doi:10.13102/sociobiology.v60i4.387-396).
- Bingham, C. T. 1903. The fauna of British India, including Ceylon and Burma. Hymenoptera, Vol. II. Ants and Cuckoo-wasps. London: Taylor and Francis, 506 pp. (page 149, redescriptions of worker and queen)
- Bolton, B. 2000. The ant tribe Dacetini. Mem. Am. Entomol. Inst. 65: 1-1028 (page 791, figs. 432, 498 redescription of worker)
- Brown, W. L., Jr. 1949f. Revision of the ant tribe Dacetini. I. Fauna of Japan, China and Taiwan. Mushi. 20:1-25. (page 17, senior synonym of butteli and indica)
- Fisher, B. L. 1997a. Biogeography and ecology of the ant fauna of Madagascar (Hymenoptera: Formicidae). J. Nat. Hist. 31: 269-302 (page 290, catalogue)
- Imai, H. T.; Brown, W. L., Jr.; Kubota, M.; Yong, H.-S.; Tho, Y. P. 1984. Chromosome observations on tropical ants from western Malaysia. II. Annu. Rep. Natl. Inst. Genet. Jpn. 34: 66-69 (page 68, karyotype described)
- Karavaiev, V. 1935a. Neue Ameisen aus dem Indo-Australischen Gebiet, nebst Revision einiger Formen. Treubia 15: 57-118 (page 106, description of queen)
- Liu, C., Sarnat, E.M., Friedman, N.R., Hita Garcia, F., Darwell, C., Booher, D., Kubota, Y., Mikheyev, A.S., Economo, E.P. 2020. Colonize, radiate, decline: Unraveling the dynamics of island community assembly with Fijian trap‐jaw ants. Evolution 74, 1082–1097 (doi:10.1111/EVO.13983).
- Mayr, G. 1866a. Myrmecologische Beiträge. Sitzungsber. Kais. Akad. Wiss. Wien Math.-Naturwiss. Cl. Abt. I 53: 484-517 (page 516, worker described)
- Mayr, G. 1887. Südamerikanische Formiciden. Verh. K-K. Zool.-Bot. Ges. Wien 37: 511-632 (page 569, queen described)
- Musfira, S.H., Rafi, M., Gusti, M., Putri, D.H., Satria, R. 2022. New data on the genus Strumigenys (Hymenoptera: Formicidae) from Sumatra. Zoosystematica Rossica 31(1): 74–86 (doi:10.31610/zsr/2022.31.1.74).
- Rasheed, M.T., Bodlah, I., Fareen, A.G., Wachkoo, A.A., Huang, X., Akbar, S.A. 2019. A checklist of ants (Hymenoptera: Formicidae) in Pakistan. Sociobiology 66(3), 426-439 (doi:10.13102/sociobiology.v66i3.4330).
- Sarnat, E. M. and Economo, E. P. 2012. The ants of Fiji. University of California Publications in Entomology. 132:1-384.
- Sarnat, E.M., Hita-Garcia, F., Dudley, K., Liu, C., Fischer, G., Economo, E.P. 2019. Ready species one: Exploring the use of augmented reality to enhance systematic biology with a revision of Fijian Strumigenys (Hymenoptera: Formicidae). Insect Systematics and Diversity 3(6): 6; 1–43 (doi:10.1093/isd/ixz005).
- Tang, K. L., Guénard, B. 2023. Further additions to the knowledge of Strumigenys (Formicidae: Myrmicinae) within South East Asia, with the descriptions of 20 new species. European Journal of Taxonomy 907, 1–144 (doi:10.5852/ejt.2023.907.2327).
- Wang, W.Y., Soh, E.J.Y., Yong, G.W.J., Wong, M.K.L., Benoit Guénard, Economo, E.P., Yamane, S. 2022. Remarkable diversity in a little red dot: a comprehensive checklist of known ant species in Singapore (Hymenoptera: Formicidae) with notes on ecology and taxonomy. Asian Myrmecology 15: e015006 (doi:10.20362/am.015006).
- Wheeler, W. M. 1922k. Ants of the American Museum Congo expedition. A contribution to the myrmecology of Africa. IX. A synonymic list of the ants of the Malagasy region. Bull. Am. Mus. Nat. Hist. 4 45: 1005-1055 (page 1034, catalogue)
- Yoshimura, M.; Onoyama, K. 2003. A new record of a dacetine ant, Strumigenys godeffroyi Mayr, 1866 (Hymenoptera: Formicidae) from Japan. Edaphologia 71: 9-10 (see also)
References based on Global Ant Biodiversity Informatics
- Abe T., S. Yamane, and K. Onoyama. Ants collected on the Krakatau Islands 100 years after the great eruptions. Biogeography 14: 65-75.
- Asfiya W., R. Ubaidillah, and Sk. Yamane. 2008. Ants (Hymenoptera: Formicidae) of the Krakataus, and Sebesi and Sebuku islands. Treubia 36: 1-9.
- Bolton, B. 2000. The Ant Tribe Dacetini. Memoirs of the American Entomological Institute 65
- Brown E.S. 1959. Immature nutfall of coconuts in the Solomon Islands. II. Changes in ant populations, and their relation to vegetation. Bulletin of Entomological Research 50: 523-558.
- Brown W. L., Jr. 1949. Revision of the ant tribe Dacetini. I. Fauna of Japan, China and Taiwan. Mushi 20: 1-25.
- CSIRO Collection
- Calilung, M.V.J. 2000. A new genus, two new species and a new subspecies of Philippine ants. Philipp. Entomol. 14: 65-73
- Chapman, J. W., and Capco, S. R. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monogr. Inst. Sci. Technol. Manila 1: 1-327
- Collingwood, C. A. and Van Harten, Antonius. 2001. The Ants (Hym., Formicidae)of Niue, Souh West Pacific. Entomologist's Monthly Magazine. 137:139-143.
- Dad J. M., S. A. Akbar, H. Bharti, and A. A. Wachkoo. 2019. Community structure and ant species diversity across select sites ofWestern Ghats, India. Acta Ecologica Sinica 39: 219–228.
- Dias R. K. S. 2002. Current knowledge on ants of Sri Lanka. ANeT Newsletter 4: 17- 21.
- Dias R. K. S. 2006. Current taxonomic status of ants (Hymenoptera: Formicidae) in Sri Lanka. The Fauna of Sri Lanka: 43-52. Bambaradeniya, C.N.B. (Editor), 2006. Fauna of Sri Lanka: Status of Taxonomy, Research and Conservation. The World Conservation Union, Colombo, Sri Lanka & Government of Sri Lanka. viii + 308pp.
- Dias R. K. S., K. R. K. A. Kosgamage, and H. A. W. S. Peiris. 2012. The Taxonomy and Conservation Status of Ants (Order: Hymenoptera, Family: Formicidae) in Sri Lanka. In: The National Red List 2012 of Sri Lanka; Conservation Status of the Fauna and Flora. Weerakoon, D.K. & S. Wijesundara Eds., Ministry of Environment, Colombo, Sri Lanka. p11-19.
- Emery C. 1893. Voyage de M. E. Simon à l'île de Ceylan (janvier-février 1892). Formicides. Annales de la Société Entomologique de France 62: 239-258.
- Emery C. Formiche raccolte da Elio Modigliani in Sumatra, Engano e Mentawei. Annali del Museo Civico di Storia Naturale 40: 661-722.
- Emery, C. 1914. Les fourmis de la Nouvelle-Calédonie et des îles Loyalty. Nova Caledonia. A. Zoologie 1:393-437.
- Emery, C. "Formiche raccolte da Elio Modigliani in Sumatra, Engano e Mentawei." Annali del Museo Civico di Storia Naturale Giacomo Doria (Genova) (2) 20, no. 40 (1900): 661-722.
- Fayle T. M., D. P. Edwards, E. C. Turner, A. J. Dumbrell, P. Eggleton, and W. A. Foster. 2012. Public goods, public services and by-product mutualism in an antfern symbiosis. Oikos 121(8): 1279-1286.
- Field Museum Collection, Chicago, Illinois (C. Moreau)
- Forel A. 1913k. Wissenschaftliche Ergebnisse einer Forschungsreise nach Ostindien ausgeführt im Auftrage der Kgl. Preuss. Akademie der Wissenschaften zu Berlin von H. v. Buttel-Reepen. II. Ameisen aus Sumatra, Java, Malacca und Ceylon. Gesammelt von Herrn Prof. Dr. v. Buttel-Reepen in den Jahren 1911-1912. Zoologische Jahrbücher. Abteilung für Systematik, Geographie und Biologie der Tiere 36:1-148.
- Framenau V.W., and M.L. Thomas. 2008. Ants of Christmas Island (Indian Ocean); identification and distribution. Records of the Western Australian Museum 25: 45-85.
- Greenslade P.J.M. and Greenslade Penelope. 1977. Some Effects of Vegetation Cover and Disturbance on a Tropical Ant Fauna. Insectes Sociaux 24(2): 163-182
- Greenslade P.J.M. and P. Greenslade. 1977. Some effects of vegetation cover and disturbance on a tropical ant fauna. Insectes Sociaux 24(2): 163-182.
- Guénard B., and R. R. Dunn. 2012. A checklist of the ants of China. Zootaxa 3558: 1-77.
- Helms J. A., S. M. Helms, N. I. Fawzi, Tarjudin, F. Xaverius. 2017. Ant community of an Acacia mangium forest in Indonesian Borneo. Serangga 22(1): 147-159.
- Hua Li-zhong. 2006. List of Chinese insects Vol. IV. Pages 262-273. Sun Yat-sen university Press, Guangzhou. 539 pages.
- Imai H. T., M. Kubota, W. L. Brown, Jr., M. Ihara, M. Tohari, and R. I. Pranata. 1985. Chromosome observations on tropical ants from Indonesia. Annu. Rep. Natl. Inst. Genet. Jpn. 35: 46-48.
- Janda M., G. D. Alpert, M. L. Borowiec, E. P. Economo, P. Klimes, E. Sarnat, and S. O. Shattuck. 2011. Cheklist of ants described and recorded from New Guinea and associated islands. Available on http://www.newguineants.org/. Accessed on 24th Feb. 2011.
- Jennings J. T., L. Krogmann, and C. Burwell. 2013. Review of the hymenopteran fauna of New Caledonia with a checklist of species. Zootaxa 3736(1): 1-53.
- Karavaiev V. 1935. Neue Ameisen aus dem Indo-Australischen Gebiet, nebst Revision einiger Formen. Treubia 15: 57-118.
- Li Z.h. 2006. List of Chinese Insects. Volume 4. Sun Yat-sen University Press
- Lin C.C., and W.J. Wu. 1996. Revision of the ant genus Strumigenys Fr. Smith (Hymenoptera: Formicidae) of Taiwan. Chinese Journal of Entomology 16:137-152.
- Mann W. M. 1919. The ants of the British Solomon Islands. Bulletin of the Museum of Comparative Zoology 63:273-391.
- Mann William. 1916. The Ants of the British Solomon Islands. Bulletin of the Museum of Comparative Zoology at Harvard College 63(7): 273-391
- Mann, W.M. 1919. The ants of the British Solomon Islands. Bulletin of the Museum of Comparative Zoology of Harvard College 63: 273-391
- Mathew R., and R. N. Tiwari. 2000. Insecta: Hymenoptera: Formicidae. Pp. 251-409 in: Director; Zoological Survey of India (ed.) 2000. Fauna of of Meghalaya. Part 7. [State Fauna Series 4.] Insecta 2000. Calcutta: Zoological Survey of India, 621 pp.
- Neville P.J., D.J. O'Dowd, and A.L. Yen. 2008. Issues and implications for research on disturbed oceanic islands illustrated through an ant survey of the Cocos (Keeling Islands). Journal of Insect Conservation 12: 313-323.
- Neville, P. J., O'Dowd, D. J., and Yen, A. L. 2008. Issues and implications for research on disturbed oceanic islands illustrated through an ant survey of the Cocos (Keeling) Islands. J Insect Conserv. 12:313-323.
- Neville, P.J., D.J. O'Dowd and A.L. Yen. 2008. Issues and implications for research on disturbed oceanic islands illustrated through an ant survey of the Cocos (Keeling) Islands. Insect Conserv. 12. 313-323.
- Pfeiffer M.; Mezger, D.; Hosoishi, S.; Bakhtiar, E. Y.; Kohout, R. J. 2011. The Formicidae of Borneo (Insecta: Hymenoptera): a preliminary species list. Asian Myrmecology 4:9-58
- Rasheed M. T., I. Bodlah, A. G. Fareen, A. A. Wachkoo, X. Huang, and S. A. Akbar. 2019. A checklist of ants (Hymenoptera: Formicidae) in Pakistan. Sociobiology 66(3): 426-439.
- Taylor R. W. 1976. The ants of Rennell and Bellona Islands. Natural History of Rennell Island, British Solomon Islands 7: 73-90.
- Taylor R. W. 1987. A checklist of the ants of Australia, New Caledonia and New Zealand (Hymenoptera: Formicidae). CSIRO (Commonwealth Scientific and Industrial Research Organization) Division of Entomology Report 41: 1-92.
- Trainor C.R., and A.N. Andersen. 2010. The ant fauna of Timor and neighbouring islands: potential bridges between the disjunct faunas of South East Asia and Australia. Australian Journal of Zoology 58: 133-144.
- Wetterer, James K. 2006. Ants (Hymenoptera: Formicidae) of Niue, Polynesia. Pacif Science. 60:(3)413-416.
- Wheeler W. M. 1937. Additions to the ant-fauna of Krakatau and Verlaten Island. Treubia 16: 21-24.
- Wheeler W.M. 1935. Check list of the ants of Oceania. Occasional Papers of the Bernice Pauahi Bishop Museum 11(11):1-56.
- Wheeler, W. M. 1927. The ants of Lord Howe Island and Norfolk Island. Proc. Am. Acad. Arts Sci. 62: 121-153
- Wheeler, William Morton. 1927. The Ants of Lord Howe Island and Norfolk Island. Proceedings of the American Academy of Arts and Sciences 62(4): 121-153
- Wheeler, William Morton.1935.Checklist of the Ants of Oceania.Occasional Papers 11(11): 3-56
- Yamane S. 2013. A Review of the ant fauna of the Krakatau Islands, Indonesia. Bull. Kitakyushu Mus. Nat. Hist. Hum. Hist. Ser: A, 11: 1-66
- Yamane Sk. 2005. Krakatau in 1982, and the commencement of myrmecological research. The nature and Insects (Konchu to shizen) 40: 27-33.
- Zryanin V. A. 2011. An eco-faunistic review of ants (Hymenoptera: Formicidae). In: Structure and functions of soil communities of a monsoon tropical forest (Cat Tien National Park, southern Vietnam) / A.V. Tiunov (Editor). – M.: KMK Scientific Press. 2011. 277 р.101-124.
- Zryanin V. A., and M. V. Mokrousov. 2015. Contribution to the ant fauna of Lombok Island. Proceedings of the 10th ANeT International Conference, 23-26 October 2015, University of Kelaniya, Sri Lanka. 34



