Monomorium rothsteini
| Monomorium rothsteini | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Solenopsidini |
| Genus: | Monomorium |
| Species: | M. rothsteini |
| Binomial name | |
| Monomorium rothsteini Forel, 1902 | |
| Synonyms | |
| |
Sparks et al. (2015) have split what was once thought to be a variable, widespread species into many species. Under this new taxonomic hypothesis past distribution data reported for Monomorium rothsteini and any studies involving this ant need to be reevaluated to determine what species they include.
| At a Glance | • Brachypterous Queen |
Identification
Sparks et al. (2015) - A member of the M. rothsteini species group. Small amber species with a concave clypeal margin. The dorsal surface of the propodeum lack longitudinal strigae and the longitudinal depression is very shallow or absent giving the propodeum and rounded appearance.
Keys including this Species
- Key to Australian Monomorium Species
- Key to Monomorium of the southwestern Australian Botanical Province
- Key to Monomorium rothsteini species complex
Distribution
This species is known from the coastal and inland regions of central and northern Queensland.
Latitudinal Distribution Pattern
Latitudinal Range: -12.41666698° to -37°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Australasian Region: Australia (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
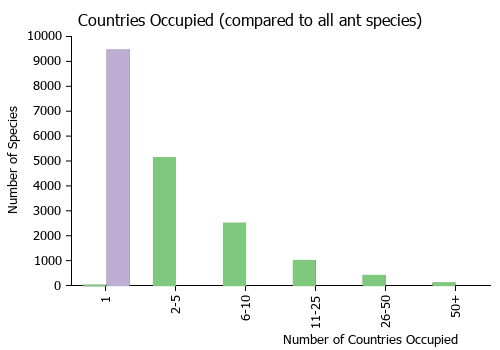
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Heterick (2001) - M. rothsteini group appears to found colonies both by claustra! colony foundation and by fission. Founding queens of claustral colonies in the study area were fully alate, whereas queens starting colonies by budding were brachypterous. Though the colour of the alate and the brachypterous queens was different they were otherwise indistinguishable. Moreover, under laboratory conditions a brachypterous queen changed to the colour of the alate after three weeks. Briese hypothesised that food stress was the determining factor in the type of colony formation.
Monomorium rothsteini is predominantly a seed harvester, taking the seeds of forbs, grasses and plants such as saltbush (Atriplex spp.). A small amount (10% <) of animal material is also eaten (Briese and Macauley 1980). The venom of M. rothsteini and another Australian Monomorium (“sp. 1066”) has recently been analysed (Andersen et al. 1991 ). The findings suggest that these species belong to a separate radiation from that of Monomorium species found elsewhere in the world, though one constituent in M. rothsteini links it with known North American Monomorium.
Genetics
COI sequences. Genbank accession numbers for this species are KC572911–KCC572920 and KC572923.
Castes
Worker
Queen
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- rothsteini. Monomorium rothsteini Forel, 1902h: 444 (w.) AUSTRALIA (Queensland).
- Type-material: lectotype worker (by designation of Sparks, et al. 2014: 517), 2 paralectotype workers.
- Type-locality: lectotype Australia: Queensland, Charters Towers (Wiederkehr); paralectotypes with same data.
- [Note: specimens from original syntype series also in MHNG.]
- Type-depository: ANIC.
- Imai, Crozier & Taylor, 1977: 348 (k.); Heterick, 2001: 409 (q.m.).
- Combination in M. (Parholcomyrmex): Santschi, 1919a: 328;
- combination in Chelaner: Ettershank, 1966: 97;
- combination in Monomorium: Taylor, 1987b: 3.
- Status as species: Emery, 1914b: 180; Wheeler, W.M. 1915g: 806; Crawley, 1915a: 134; Emery, 1922e: 171; Ettershank, 1966: 97; Taylor & Brown, 1985: 58; Taylor, 1987a: 20; Bolton, 1995b: 266; Heterick, 2001: 408 (redescription); Heterick, 2009: 160; Sparks, et al. 2014: 517 (redescription).
- Senior synonym of doddi: Heterick, 2001: 408; Sparks, et al. 2014: 517.
- Distribution: Australia.
- doddi. Monomorium (Parholcomyrmex) rothsteini st. doddi Santschi, 1919a: 328 (w.) AUSTRALIA (Queensland).
- Type-material: 7 syntype workers.
- Type-locality: Australia: Queensland, Townsville (F.P. Dodd).
- Type-depository: NHMB.
- Combination in Chelaner: Ettershank, 1966: 96;
- combination in Monomorium: Taylor, 1987b: 3.
- Subspecies of rothsteini: Ettershank, 1966: 96; Taylor & Brown, 1985: 58; Taylor, 1987a: 20; Bolton, 1995b: 261.
- Junior synonym of rothsteini: Heterick, 2001: 408.
Type Material
- Monomorium rothsteini: Lectotype (designated by Sparks, Andersen & Austin, 2015: 517), workers, Charters Towers, Queensland, Australia, Wiederkehr, ANIC32-015695, Australian National Insect Collection.
- Monomorium rothsteini: Paralectotype (designated by Sparks, Andersen & Austin, 2015: 517), 2 workers, Charters Towers, Queensland, Australia, Wiederkehr, Australian National Insect Collection.
- Monomorium (Paraholcomyrmex) rothsteini doddi Santschi, 1919: Syntype, 5 workers, Townsville, Queensland, Australia, F.P. Dodd, Naturhistorisches Museum, Basel.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Sparks et al. (2015) - (n=9). HW 0.70–0.96, HL 0.74–0.96, EL 0.16–0.18, PMH 0.29–0.37, PH 0.25–0.33, PNH 0.18–0.23, LHW 0.45–0.59, EW 0.10–0.12, PML 0.47–0.64, ML 0.80–1.05, PL 0.37–0.43, PNWdv 0.20–0.27.
Small species with a medium sized, rectangular head which narrows from immediately behind eyes to vertex; posterior cephalic margin shallowly depressed medially. Anterodorsal margin of the clypeus concave with frontolateral carinae forming rounded ridges that extend just beyond anteroventral clypeal margin, anterolateral angles are bluntly rounded; anteroventral margin without a small median projection, margin appears straight. Sculpture on frons variable: strigate, extending well above antennal lobes or restricted to antennal lobes and area in between or rarely limited to a few strigae only present on antennal lobes; coarse lateral cephalic strigae not reaching eye margin or rarely with one or two strigae reaching eye margin. Eyes medium, EW 0.20–0.23 x LHW, 10 ommatidia in longest vertical axis, 8 in longest horizontal axis.
Mesonotum with coarse strigae extending from metanotal groove onto posterior region only, rugulose between strigae, remainder smooth and shining; posterior mesonotum generally flat and contiguous with surrounding surface of mesonotum or rarely raised with a faint horseshoe shaped margin. Mesopleuron alveolate with strigulae spanning area between metanotal groove and promesonotal suture on dorsal half. Propodeum in lateral view with dorsolateral angles clearly forming an obtuse angle; laterally alveolate with 3–4 strigae over the metapleural gland bulla; dorsally with anterodorsal transverse carina present, a few weak transverse strigae present on some specimens, longitudinal carinae absent, longitudinal depression very shallow or absent, propodeum appears flat or slightly rounded dorsally; posterolateral corners rounded above metapleural lobes and dorsolateral angles rounded. Petiole node of medium breadth: between 2 and 2.5 x eye width when viewed from above; shape in posterior view oval, in lateral view anterior and posterior faces converging, apex narrowly rounded from higher anterior face to lower posterior face; posterior surface of petiole node finely reticulate laterally and basally but not extending more than half way to dorsal surface or may appear to be without sculpture, smooth and shining, postpetiole faintly reticulate on posterior surface.T1 completely smooth and without sculpture.
Head and mesosoma dark amber orange to orange brown, legs brown, petiole and postpetiole dorsally infuscated, metasomal tergites dark brown.
Queen
Heterick (2001) - HML 4.10-6.00; HL 1.24-1.51; HW 1.38-1.72; Cel 105-121; SL 0.79-1.10; SI 56-64; PW 1.17-1.59 (16 measured).
As for the queen of Monomorium sordidum, but with the following apomorphies.
Head. Vertex planar, or convex; frons longitudinally striate with erect and suberect setae. (Viewed from front) compound eyes set in posterior half of head capsule, or set in anterior half of head capsule; (viewed laterally) compound eyes set posterior of the midline of head capsule, or set at midline of head capsule.
Alitrunk. Mesoscutal pilosity consisting of decumbent and subdecumbent setulae anteriad and erect and suberect setae posteriad; dorsal appearance of mesoscutum smooth and shining with a few scattered piliferous pits, or striolate and microreticulate. Propodeal sculpture present as uniform rugosity, with well defined costulae on declivitous face of propodeum. Metapleural lobes absent or indistinct. Propodeal spiracle lateral and nearer metanotal groove than declivitous face of propodeum.
Petiole and postpetiole. Propodeal sculpture present; petiolar node rugose. Ratio of greatest node breadth (viewed from front) to greatest node width (viewed in profile) near 4:3. Height ratio of petiole to postpetiole near 1: 1; height-length ratio of postpetiole near 2:1. Sculpture present in form of microreticulation; ventral process absent or vestigial.
General characters. Colour variable: brown, orange or red, alone or in combination, head, appendages and gaster often lighter than alitrunk. Brachypterous alates seen and examined. Ergatoid or worker-female intercastes not seen.
Male
Heterick (2001) - HML 3.86-4.62; HL 0.97-1.10; HW 1.17-1.41; Cel 121-128; SL 0.26-0.34; SI 24-29; PW 1.38-1.66 (7 measured).
As for the male of Monomorium sordidum, but with the following apomorphies.
Alitrunk. Axillae separated by distance of about half greatest width of scutellum.
Petiole and postpetiole. Sculpture present in form of microreticulation, or present; petiolar node rugose. Heightlength ratio of postpetiole near 2: 1.
General characters. Colour brown to black.
Karyotype
- 2n = 22 (Australia) (Imai et al., 1977) (as Chelaner rothsteini).
References
- Ettershank, G. 1966. A generic revision of the world Myrmicinae related to Solenopsis and Pheidologeton (Hymenoptera: Formicidae). Aust. J. Zool. 14: 73-171 (page 97, Combination in Chelaner)
- Forel, A. 1902j. Fourmis nouvelles d'Australie. Rev. Suisse Zool. 10: 405-548 (page 444, worker described)
- Heterick, B. E. 2001. Revision of the Australian ants of the genus Monomorium (Hymenoptera: Formicidae). Invertebrate Taxonomy. 15:353-459. (page 409, queen, male described; page 408, Senior synonym of bogischi, doddi, humilior, leda and subapterum)
- Heterick, B.E. 2021. A guide to the ants of Western Australia. Part I: Systematics. Records of the Western Australian Museum, Supplement 86, 1-245 (doi:10.18195/issn.0313-122x.86.2021.001-245).
- Heterick, B.E. 2022. A guide to the ants of Western Australia. Part II: Distribution and biology. Records of the Western Australian Museum, supplement 86: 247-510 (doi:10.18195/issn.0313-122x.86.2022.247-510).
- Hoffmann, B., Eldridge, J., Marston, C. 2023. The first eradication of an exotic ant species from the entirety of Australia: Pheidole fervens. Management of Biological Invasions, 14(4), 619–624 (doi:10.3391/mbi.2023.14.4.03).
- Imai, H. T.; Crozier, R. H.; Taylor, R. W. 1977. Karyotype evolution in Australian ants. Chromosoma (Berl.) 59: 341-393 (page 348, karyotype described)
- Meurville, M.-P., LeBoeuf, A.C. 2021. Trophallaxis: the functions and evolution of social fluid exchange in ant colonies (Hymenoptera: Formicidae). Myrmecological News 31: 1-30 (doi:10.25849/MYRMECOL.NEWS_031:001).
- Minkey, D.M., Spafford, H. 2008. Weed seed removal by ants in the crop growing areas of Western Australia. Proceedings of the 16th Australian Weeds Conference, pp. 474-476.
- Palfi, Z., Robinson, W., Spooner, P.G. 2020. Cheaters and removalists: the influence of soil disturbance on ant–seed interactions in roadside vegetation. Insectes Sociaux (doi:10.1007/S00040-020-00778-1).
- Santschi, F. 1919a. Cinq notes myrmécologiques. Bull. Soc. Vaudoise Sci. Nat. 52: 325-350 (page 328, Combination in M. (Parholcomyrmex))
- Sozanski, K., Prado, L.P., Mularo, A.J., Sadowski, V.A., Jones, T.H., Adams, R.M.M. 2020. Venom function of a new species of Megalomyrmex Forel, 1885 (Hymenoptera: Formicidae). Toxins 12, 679 (doi:10.3390/toxins12110679).
- Sparks, K. 2015. Australian Monomorium: Systematics and species delimitation with a focus of the M. rothsteini complex. Ph.D. thesis, University of Adelaide.
- Sparks, K.S., Andersen, A.N. and Austin, A.D. 2015. Systematics of the Monomorium rothsteini Forel species complex (Hymenoptera: Formicidae), a problematic ant group in Australia. Zootaxa. 3893:489–529. doi:10.11646/zootaxa.3893.4.2
- Taylor, R. W. 1987b. A checklist of the ants of Australia, New Caledonia and New Zealand (Hymenoptera: Formicidae). First supplement, 10 July, 1987. CSIRO Div. Entomol. Rep. 41(Suppl. .1: 1-5 (page 3, Combination in Monomorium)
References based on Global Ant Biodiversity Informatics
- Crawley W. C. 1915. Ants from north and central Australia, collected by G. F. Hill. - Part I. Annals and Magazine of Natural History (8)15: 130-136.
- Debuse V. J., J. King, and A. P. N. Hous. 2007. Effect of fragmentation, habitat loss and within-patch habitat characteristics on ant assemblages in semi-arid woodlands of eastern Australia. Lanscape Ecology 22: 731-745.
- Emery C. 1914. Formiche d'Australia e di Samoa raccolte dal Prof. Silvestri nel 1913. Bollettino del Laboratorio di Zoologia Generale e Agraria della Reale Scuola Superiore d'Agricoltura. Portici 8: 179-186.
- Heterick B. E. 2009. A guide to the ants of south-western Australia. Records of the Western Australian Museum Supplement 76: 1-206.
- Imai H. T., R. H. Crozier, and R. W. Taylor. 1977. Karyotype evolution in Australian ants. Chromosoma 59: 341-393.
- Taylor R. W. 1987. A checklist of the ants of Australia, New Caledonia and New Zealand (Hymenoptera: Formicidae). CSIRO (Commonwealth Scientific and Industrial Research Organization) Division of Entomology Report 41: 1-92.
- Taylor R. W., and D. R. Brown. 1985. Formicoidea. Zoological Catalogue of Australia 2: 1-149.




