Anochetus paripungens
| Anochetus paripungens | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Anochetus |
| Species: | A. paripungens |
| Binomial name | |
| Anochetus paripungens Brown, 1978 | |
Anochetus paripungens is restricted to the Top End of the Northern Territory where it is found primarily in rainforest sites (with only a few records from savannah woodlands). Most specimens are encountered as foragers in leaf litter. Nests are in soil generally under objects, in rotten wood and in termite nests. The type series of this species was found nesting in a rotten log which it shared with an Anochetus graeffei colony.
Identification
Propodeal angles produced as spines which are angled slightly posteriorly, the posterior face of the propodeum forming angle at the base of the spines; sculpture on front of head extending well beyond eyes and ending approximately half-way between eyes and posterior margin; pronotum shining with scattered shallow, oval shaped punctures; hind tibiae with semi-erect hairs restricted to the inner surfaces. This species is most similar to Anochetus wiesiae but differs in having a more lightly coloured body, shallow punctures on the dorsum of the pronotum, more posteriorly directed propodeal spines, a narrower petiolar node with less angular sides, and the lack of erect hairs on the outer surfaces of the hind tibiae. It is also only known from the Top End of the Northern Territory while A. wiesiae occurs on coastal Queensland.
Keys including this Species
Distribution
Northern Australia.
Latitudinal Distribution Pattern
Latitudinal Range: -11.175972° to -13.48333333°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Australasian Region: Australia (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
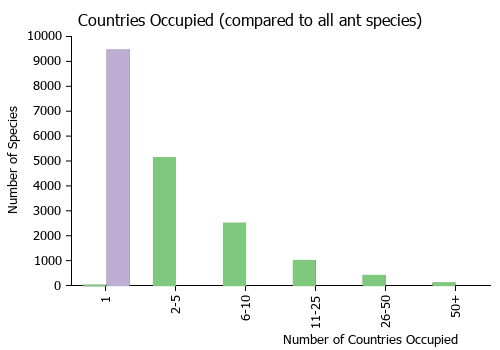
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Gibson et al. (2018) found the trap-jaws of Anochetus targionii and Anochetus paripungens have mandible strikes that overall closely outperform but closely resemble those found in Odontomachus ruginodis, reaching a mean maximum rotational velocity and acceleration of around 3.7 x 104 rad s-1 and 8.5 x 108 rad s-2, respectively. This performance is consistent with predictions based on body size scaling relationships described for Odontomachus.
|
Castes
Queen and male unknown.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- paripungens. Anochetus paripungens Brown, 1978c: 596, fig. 35 (w.) AUSTRALIA (Northern Territory).
- Type-material: holotype worker, 12 paratype workers.
- Type-locality: holotype Australia: Northern Territory, Howard Springs, Darwin area, 7-8.vii.1951, rotten log, shady gallery forest (W.L. Brown & W. Bateman); paratypes with same data.
- Type-depositories: ANIC (holotype); ANIC, MCZC (paratypes).
- Status as species: Taylor & Brown, 1985: 20; Taylor, 1987a: 7; Bolton, 1995b: 65; Shattuck & Slipinska, 2012: 15 (redescription).
- Distribution: Australia.
Type Material
Holotype one of 13 workers selected from a small nest series taken in a rotten log in shady gallery forest at Howard Springs, Darwin area, Northern Territory, Australia, 7-8 July 1951, by W. L. Brown, Jr. and W. Bateman. In the same log we found a colony of Anochetus graeffei. The type locality, on the Howard River, was at the Darwin water supply source in 1951. I returned to this place in 1972, but found that it had been transformed into a public park and campground, so that the habitat was largely destroyed.
- Holotype, worker, Howard Springs, Darwin area, Northern Territory, Australia, Museum of Comparative Zoology.
- Paratype, 11 workers, Howard Springs, Darwin area, Northern Territory, Australia, Museum of Comparative Zoology.
- Paratype, 2 workers, Howard Springs, Darwin area, Northern Territory, Australia, ANIC32-016009, Australian National Insect Collection.
Description
Worker
Worker, holotype: TL 5.2, HL 1.28, HW 1.18, ML 0.70, scape L 1.05, eye L 0.22, WL 1.64 mm; CI 92, MI 55.
Worker paratypes: TL 5.0-5.2, HL 1.24-1.31, HW 1.13-1.20, ML 0.70-0.72, scape L 1.01-1.07, eye L 0.21-0.22, WL 1.55-1.64 mm; CI 91-92, MI 55-56.
Head with mandibles and antennae much as in Anochetus rectangularis, but the eyes a little smaller, more nearly circular, strongly convex. Antennal scapes just barely surpassing posterior margin of occipital lobe in full-face view. Frontal striation more extensive than in rectangularis, reaching a bit more than halfway from the eyes to the nuchal carina in the middle. Mandibles with straight, cultrate dorsomedial margin and obscurely crenulate ventromedial margin; preapical angle present but weak; preapical excision small; intercalary apical tooth conical, arising from near base of ventral apical tooth. Upper inner temporal areas, just beyond oblique field of frontal striation, with numerous small punctures; head otherwise generally smooth and shining.
Antennae slender; funicular segments II, III and IV together about twice as long as I; IV about twice as long as broad, and slightly longer than II. Scapes surpassing posterior border of «occipital» lobe by only about their apical thickness when the head is seen in perfect full-face view.
Trunk, petiole and gaster shown in fig. 35; notable are the small but sharp teeth on the propodeal angles and the strong, divergent teeth extending the free corners of the petiolar node, as shown in fig. 35. Pronotum with 5 sharp costulae running around anterior slope, otherwise smooth and shining, with numerous small, separated punctures on disc. Mesonotal disc more than twice as broad as long, nearly smooth, shining. Propodeum coarsely, transversely striate (or costulate) over dorsal surface, its sides smooth and shining, as are petiolar node and gaster. Mandibles and femora smooth, shining, sparsely punctulate; scapes and tibiae more densely, but very finely punctulate, moderately shining; tarsi and funiculi densely punctulate and opaque or nearly so.
Pubescente sparse on dorsal surfaces of body and undersides of head and fore coxae, especially sparse on gaster, consisting of fine, inconspicuous, appressed and decumbent hairs. Erect hairs fine, tapered, sparsely arranged along median third of head, anterior underside of head, posterior vertex, and frontal carinae; about 14 on pronotum and mesonotum (10-18 in paratypes); more and longer hairs on upper and lower surfaces of gaster (few on sides of gaster); a few scattered erect hairs on scapes, anterior sides of fore coxae and flexor surfaces of femora, as well as inner ventral margins and apices of mandibles.
Color castaneous (dull brownish-orange); middle of vertex, mesopleura and gaster darker reddish-brown; posterior corners of head, mandibles, petiole and legs ferruginous yellow.
Description from Shattuck & Slipinska (2012):
Worker description. Sculpturing on front of head extending approximately half-way between the level of the eyes and posterior margin. Scapes not reaching posterolateral corners ('lobes') of head; with abundant, appressed pubescence and a limited number of erect hairs. Eyes relatively small (EL < 0.22mm). Pronotum shining and with scattered, weak, oval shaped punctures. Mesonotum smooth and shining. Anterior section of pronotum with transverse wrinkles and ridges. Propodeum with angles produced as spines. Dorsum of propodeum rounded laterally, sculptured with coarse transverse striations and with inclined erect hairs. Metapleuron mostly smooth and shining. Dorsolateral corners of petiolar node with long spines. Hind tibiae with semierect hairs restricted to the inner surfaces. Colour brown, head, antennae and mandibles yellow-brown, legs yellow.
Measurements. Worker (n = 5): CI 90–93; EI 16–18; EL 0.19–0.21; HL 1.24–1.31; HW 1.12–1.21; HFL 1.11–1.14; ML 1.40–1.50; MandL 0.68–0.72; MTL 0.79–0.84; PronI 54–57; PronW 0.63–0.67; SL 1.04–1.11; SI 91–93.
References
- Brown, W. L., Jr. 1978c. Contributions toward a reclassification of the Formicidae. Part VI. Ponerinae, tribe Ponerini, subtribe Odontomachiti. Section B. Genus Anochetus and bibliography. Stud. Entomol. 20: 549-638 (page 596, fig. 35 worker described)
- Gibson, J. C., F. J. Larabee, A. Touchard, J. Orivel, and A. V. Suarez. 2018. Mandible strike kinematics of the trap-jaw ant genus Anochetus Mayr (Hymenoptera: Formicidae). Journal of Zoology. 306:119-128. doi:10.1111/jzo.12580
- Hazarika, H.N., Khanikor, B. 2021. Integration of morphological and molecular taxonomic characters for identification of Odontoponera denticulata (Hymenoptera: Formicidae:Ponerinae) with the description of the antennal sensilla. Zoologischer Anzeiger 293: 89-100 (doi:10.1016/j.jcz.2021.05.009).
- Shattuck, S.O. & Slipinska, E. 2012. Revision of the Australian species of the ant genus Anochetus (Hymenoptera Formicidae). Zootaxa 3426, 1–28.
References based on Global Ant Biodiversity Informatics
- Andersen A. N., J. C. Z. Woinarski, and B. Hoffman. 2004. Biogeography of the ant fauna of the Tiwi Islands, in northern Australia's moonsoonal tropics. Australian Journal of Zoology 52: 97-110.
- Andersen A. N., L. T. van Ingen, and R. I. Campos. 2007. Contrasting rainforest and savanna ant faunas in monsoonal northern Australia: a rainforest patch in a tropical savanna landscape. Australian Journal of Zoology 55: 363-369.
- Andersen A. N., M. Houadria, M. Berman, and M. van der Geest. Rainforest ants of the Tiwi Islands: a remarkable centr of endemism in Australia's monsoonal tropics. Insectes Sociaux 59: 433-441.
- Andersen A. N., R. R. Ribbons, M. Pettit, and C. L. Parr. 2014. Burning for biodiversity: highly resilient ant communities respond only to strongly contrasting fire regimes in Australias seasonal tropics. Journal of Applied Ecology 51: 14061413.
- Andersen, Alan N., John C.Z. Woinarski and Ben D. Hoffman. 2004. Biogeography of the ant fauna of the Tiwi Islands, in northern Australia's monsoonal tropics. Australian Journal of Zoology 52: 97-110.
- Brown Jr., W.L. 1978. Contributions toward a reclassification of the Formicidae. Part VI. Ponerinae, Tribe Ponerini, Subtribe Odontomachiti, Section B. Genus Anochetus and Bibliography. Studia Entomologia 20(1-4): 549-XXX
- Brown W.L. Jr. 1978. Contributions toward a reclassification of the Formicidae. Part VI. Ponerinae, tribe Ponerini, subtribe Odontomachiti. Section B. Genus Anochetus and bibliography. Studia Ent. 20(1-4): 549-638.
- Shattuck S. O., and E. Slipinska. 2012. Revision of the Australian species of the ant genus Anochetus (Hymeoptera: Formicidae). Zootaxa 3426: 1-28.

