Strumigenys emmae
| Strumigenys emmae | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Strumigenys |
| Species: | S. emmae |
| Binomial name | |
| Strumigenys emmae (Emery, 1890) | |
| Synonyms | |
| |
| Common Name | |
|---|---|
| Yofushi-uroko-ari | |
| Language: | Japanese |
A pantropical species, dispersed by human commerce (Brown, 1949b) and natural dispersal (Taylor in Kubota, 1976). It is found most often in highly disturbed habitats such as beach margins and agricultural areas. The geographic origin of emmae is apparently Australia, where several related species occur (Bolton 2000).
| At a Glance | • Highly invasive • Parthenogenetic |
Identification
Bolton (2000) - Within the Strumigenys emmae-group, emmae is recognised by its combination of short mandibles, broad head, short 4-merous antennae with distinctly angular leading edge on the scape, presence of orbicular hairs on the promesonotum as well as on the cephalic dorsum, absence of erect hairs on the occipital margin and lack of a strong second preapical tooth located between the apicodorsal tooth and the long spiniform preapical tooth.
Keys including this Species
- Key to Australian Strumigenys Species
- Key to Micronesian Ants
- Key to Strumigenys of India
- Key to Afrotropical Strumigenys
- Key to Neotropical and Nearctic Strumigenys
- Key to Strumigenys of East Asia
- Key to Malagasy Strumigenys
- Key to US Strumigenys species
- Key to Strumigenys of Hispaniola
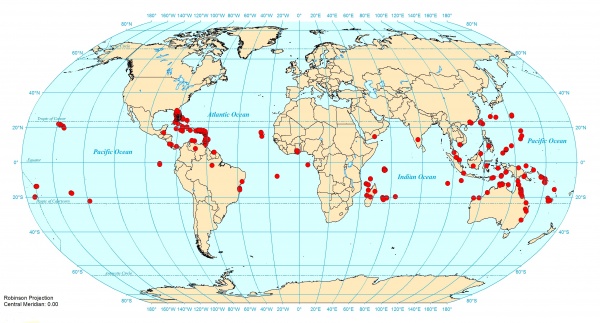
Distribution
Bolton (2000) - This very successful pantropical tramp species was described and discussed by Brown (1949a) and Bolton (1983). Previous distribution records include Hawaii, Guam, U.S.A., Puerto Rico, West Indies, Cuba, Bahamas, Surinam, Ghana, Equatorial Guinea, India, West Malaysia, Sumatra, Singapore, Philippines, Sulawesi, New Guinea, Solomon Islands, Australia, Vanuatu, Samoa (Brown, 1949a; Wilson & Taylor, 1 967; Kempf, 1972; Taylor, 1976; Bolton, 1983; Deyrup, 1997). A recent survey of Florida by Deyrup & Deyrup (1999) and Deyrup, Davis & Cover (2000) shows that emmae is widely distributed in the state.
This species has been recorded in Japan only from Ogasawara Islands (Kubota, 1976; Shindo, 1979; Japanese Ant Image Database).
Latitudinal Distribution Pattern
Latitudinal Range: -10.75° to -33.27971°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Afrotropical Region: Comoros, Equatorial Guinea, Ghana, United Republic of Tanzania, Yemen.
Australasian Region: Australia, New Caledonia.
Indo-Australian Region: Borneo, Guam, Hawaii, Indonesia, Malaysia, Marshall Islands, Micronesia (Federated States of), New Guinea, Northern Mariana Islands, Philippines, Samoa, Singapore, Solomon Islands, Tonga, Vanuatu.
Malagasy Region: Madagascar, Mayotte, Réunion, Seychelles.
Nearctic Region: United States.
Neotropical Region: Anguilla, Aruba, Barbados, Belize, British Virgin Islands, Cayman Islands, Colombia, Costa Rica, Cuba, Dominican Republic, Ecuador, Galapagos Islands, Greater Antilles, Grenada, Guadeloupe, Mexico, Netherlands Antilles, Puerto Rico, Saint Lucia, Suriname.
Oriental Region: India, Sri Lanka, Thailand, Vietnam.
Palaearctic Region: China, Japan, Poland, United Kingdom of Great Britain and Northern Ireland.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
This species occurs in both dry and mesic woods, and in disturbed areas such as gardens (Deyrup, Davis & Cover, 2000), and is often found in relatively dry leaf litter. Dealate queens are notable common in leaf litter with small groups of workers, giving the impression that the colonies are producing new queens while the colony is small. This impression, if correct, would fit the strategy of a weedy inhabitant of frequently disturbed sites (Deyrup 1997).
Deyrup and Deyrup (1999) - Two colonies were examined. One was in a hollow acorn in leaf litter in a mesic forest at Spruce Creek Preserve, Volusia Co.; this nest had one queen, 14 workers, and brood. A second colony was found in a hollow, buried acorn of Quercus chapmanii in xeric scrub forest at the Archbold Biological Station. Highlands Co. This colony had one queen, 42 workers, and brood. When the acorn from Highlands Co. was opened, there was one dead, white mite with one leg detached, and a shriveled entombryid collembolan that was being eaten by a larva. This colony was offered sifted leaf-litter and the next day there were 7 dead entomobryids in the colony inside the acorn, including one collembolan that was being eaten by a larva. The next day two larvae were seen feeding on entomobryids. It appears that entomobryid Collembola are a preferred prey of S. emmae.
Deyrup, Davis & Cover (2000) - The species Strumigenys eggersi and Strumigenys emmae are the only dacetine ants that are commonly found in dry and mesic habitats of south and central Florida. There is little evidence that native dacetines were ever common in these areas, but if these two exotics continue their northward expansion we may be able to get some idea of their effect on native ants, since we have records of hundreds of litter samples from north Florida. These species are probably more or less specialized predators on entomobryiid Collembola. If they have not had an impact by reducing or replacing the populations of native predators of Collembola in south Florida, they must be a novel predator of the Collembola themselves in this area.
Brown (1949) - Nothing is known of the food preferences. Many of the collecting records are of single females taken under objects lying on sea beaches; workers have been taken with the sieve and Berlese funnel in both forested and agricultural areas, usually near the sea. This ant seems to show tolerance for a wider range of environmental conditions than most dacetines.
Tang et al. (2019) - In Hong Kong, this introduced species is found mostly in disturbed habitats, including managed grasslands, isolated patches of urban trees, and with a single record from a secondary forest but located slightly over 100 m from urban habitations. Common in the Mai Po Nature Reserve, a heavily disturbed landscape managed for bird populations. Known elevation range in Hong Kong is from 1 to 407 m. A single alate gyne was collected between June 27 and July 11 in a Malaise trap located within a mangrove area.
Castes
Worker
Images from AntWeb
   
| |
| Worker. Specimen code casent0060342. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Syntype of Strumigenys emmae. Worker. Specimen code casent0102082. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MSNG, Genoa, Italy. |
   
| |
| Worker. Specimen code casent0104133. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
   
| |
| Worker. Specimen code casent0133445. Photographer Erin Prado, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0135353. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0178454. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by BNM, Koror, Palau. |
   
| |
| Specimen code lacm. . | |
  
| |
| Worker. Specimen code casent0173254. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CDRS, Galapagos, Ecuador. |
  
| |
| Worker. Specimen code casent0133446. Photographer Erin Prado, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
Queen
Images from AntWeb
   
| |
| Queen (alate/dealate). Specimen code casent0060340. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Worker. Specimen code casent0104516. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
   
| |
| Queen (alate/dealate). Specimen code casent0135362. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
   
| |
| Queen (alate/dealate). Specimen code casent0173253. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by CDRS, Galapagos, Ecuador. |
Male
Images from AntWeb
     
| |
| Male (alate). Specimen code casent0133418. Photographer Erin Prado, uploaded by California Academy of Sciences. | Owned by CAS, San Francisco, CA, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- emmae. Epitritus emmae Emery, 1890b: 70, pl. 8, fig. 6 (w.) ANTILLES. Wheeler, W.M. 1908a: 149 (q.). Combination in Quadristruma: Brown, 1949b: 48; in Strumigenys: Bolton, 1999: 1674. Senior synonym of clypeatus, malesiana, wheeleri: Brown, 1949b: 48. See also: Bolton, 1983: 400; Bolton, 2000: 950.
- clypeatus. Epitritus clypeatus Szabó, 1909: 27, fig. 1 (w.) NEW GUINEA. Junior synonym of emmae: Brown, 1949b: 48.
- malesiana. Epitritus clypeatus var. malesiana Forel, 1913k: 83 (w.q.) INDONESIA (Sumatra). Junior synonym of emmae: Brown, 1949b: 48.
- wheeleri. Epitritus wheeleri Donisthorpe, 1916a: 121 (w.) HAWAII. Junior synonym of emmae: Brown, 1949b: 48.
Type Material
- Epitritus emmae Emery, 1890: Holotype, worker, St. Thomas Island, Virgin Islands, museum unknown (prob. MCSN (Genoa)).
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Bolton (2000) - TL 1.5 - 1.9, HL 0.40 - 0.46, HW 0.33 - 0.39, CI 80 - 88, ML 0.11 - 0.16, MI 26 - 35, SL 0.18 - 0.24, SI 56 - 62, PW 0.21 - 0.25 , AL 0.40 - 0.48 (33 measured). Exposed length of fully closed mandible less than width of anterior clypeal margin. Preapical dentition of mandible somewhat variable: inner margin between the spiniform preapical tooth and the apicodorsal tooth may be unarmed, may have a low blunt tubercle or welt, or may have a tiny denticle (intermediates between these are known). Intercalary and preapical dentition may be obscured by the principal teeth when the mandibles are fully closed. Antenna with 4 segments; second funicular segment not obviously much longer than broad. Cephalic dorsum with orbicular hairs; upper scrobe margin fringed with similar hairs and with a clavate apicoscrobal hair. Occipital margin of head without short erect hairs. Leading edge of scape flattened and expanded into an obtuse angle or broad but shallowly convex lobe in the median third of its length. Pronotal humeral hair stiff, stout. Ground-pilosity of pronotal dorsum orbicular but the hairs tending to be smaller than on head. Mesonotum usually with a single pair of short erect hairs but these sometimes absent. Promesonotal dorsum reticulate-punctate, without longitudinal striae. Disc of postpetiole usually with weak sculpture at least in part, only rarely entirely smooth; sides of disc in dorsal view with projecting spongiform tissue. First gastral tergite with short suberect to erect stubbly hairs that are simple to feeble expanded apically. Basigastral costulae at least equal in length to postpetiole disc.
Type Material
Bolton (2000):
Holotype worker, ST THOMAS I. (Antilles) (Museo Civico di Storia Naturale, Genoa).
Epitritus clypeatus Szabó, 1909: 1, figs. la, c. Syntype workers, NEW GUINEA: Berlinhafen (L. Biró); and SINGAPORE (L. Biró) (Hungarian Natural History Museum) [not seen].
Epitritus clypeatus var. malesiana Forel, 1913e: 83. Syntype workers and queen, INDONESIA: Sumatra (Buttel-Reepen) (Musee d'Histoire Naturelle Genève) [not seen].
Epitritus wheeleri Donisthorpe, 1916: 121. Holotype worker, HAWAII: Oahu, Honolulu (R.C.L. Perkins) [not in The Natural History Museum, holotype lost].
Etymology
Named after Emma Forel, the wife of ant taxonomist Auguste Forel.
References
- Albuquerque, E., Prado, L., Andrade-Silva, J., Siqueira, E., Sampaio, K., Alves, D., Brandão, C., Andrade, P., Feitosa, R., Koch, E., Delabie, J., Fernandes, I., Baccaro, F., Souza, J., Almeida, R., Silva, R. 2021. Ants of the State of Pará, Brazil: a historical and comprehensive dataset of a key biodiversity hotspot in the Amazon Basin. Zootaxa 5001, 1–83 (doi:10.11646/zootaxa.5001.1.1).
- Bharti, H. & Akbar, S.A. 2013. Taxonomic studies on the ant genus Strumigenys Smith, 1860 (Hymenoptera, Formicidae) with report of two new species and five new records including a tramp species from India. Sociobiology 60, 387-396 (doi:10.13102/sociobiology.v60i4.387-396).
- Blard, F., Dorow, W.-H.-O., Delabie, J. H. C. 2003. Les Fourmis de l’île de la Réunion (Hymenoptera, Formicidae). Bulletin de La Société Entomologique de France, 108(2), 127–137 (doi:10.3406/bsef.2003.16939).
- Bolton, B. 1983. The Afrotropical dacetine ants (Formicidae). Bulletin of the British Museum (Natural History). Entomology. 46:267-416. (page 400, see also)
- Bolton, B. 2000. The ant tribe Dacetini. Mem. Am. Entomol. Inst. 65: 1-1028 (page 950, see also)
- Borowiec, L. 2014. Catalogue of ants of Europe, the Mediterranean Basin and adjacent regions (Hymenoptera: Formicidae). Genus (Wroclaw) 25(1-2): 1-340.
- Brassard, F., Leong, C.-M., Chan, H.-H., Guénard, B. 2021. High diversity in urban areas: How comprehensive sampling reveals high ant species richness within one of the most urbanized regions of the world. Diversity 13, 358 (doi:10.3390/d13080358).
- Brown, W. L., Jr. 1949c. Revision of the ant tribe Dacetini: III. Epitritus Emery and Quadristruma new genus (Hymenoptera: Formicidae). Trans. Am. Entomol. Soc. 75:43-51. (page 48, Combination in Quadristruma, and senior synonym of clypeatus, malesiana and wheeleri)
- Dekoninck, W., Wauters, N., Delsinne, T. 2019. Capitulo 35. Hormigas invasoras en Colombia. Hormigas de Colombia.
- Deyrup, M. 1997. Dacetine ants of the Bahamas (Hymenoptera: Formicidae). Bahamas J. Sci. 5:2-6.
- Deyrup, M. and S. Deyrup. 1999. Notes on introduced ant Quadristruma emmae (Hymenoptera: Formicidae) in Florida. Entomol. News. 110:13-21.
- Deyrup, M., Davis, L. & Cover, S. 2000. Exotic ants in Florida. Transactions of the American Entomological Society 126, 293-325.
- Deyrup, M.A., Carlin, N., Trager, J., Umphrey, G. 1988. A review of the ants of the Florida Keys. Florida Entomologist 71: 163-176.
- Emery, C. 1890c. Studii sulle formiche della fauna neotropica. Bullettino della Società Entomologica Italiana 22:38-80. (page 70, pl. 8, fig. 6 worker described)
- Fisher, B. L. 1997a. Biogeography and ecology of the ant fauna of Madagascar (Hymenoptera: Formicidae). J. Nat. Hist. 31: 269-302 (see also)
- Fontenla, J.L., Brito, Y.M. 2011. Hormigas invasoras y vagabundas de Cuba. Fitosanidad 15(4), 253-259.
- Guerrero, R.J., Fernandez, F., Escarraga, M.E., Perez-Pedraza, L.F., Serna, F., Mackay, M.P., Sandoval, V., Vergara, V., Suarez, D., Garcia, E.I., Sanchez, A., Meneses, A.D., Tocora, M.C., Sosa-Calvo, J. 2018. New records of myrmicine ants (Hymenoptera: Formicidae) for Colombia. Revista Colombiana de Entomología 44: 238-259 (DOI 10.25100/socolen.v44i2.7115).
- Hamer, M.T., Telfer, M.G., Turner, C.R. 2023. Rediscovered after 102 years: Strumigenys rogeri (Formicidae; Myrmicinae) is rediscoverd in Britain from the Eden Project East Cornwall (VC 22). British Journal of Entomology, Natural History 36, 225-231.
- Hamer, M.T., Turner, C.R. 2024. Strumigenys emmae (Emery, 1890) (Myrmicinae) new to Britain, with an updated key to the known Strumigenys of the West Palaearctic. Zootaxa 5415(4), 570–576 (doi:10.11646/zootaxa.5415.4.6).
- Herrera, H.W., Baert, L., Dekoninck, W., Causton, C.E., Sevilla, C.R., Pozo, P., Hendrickx, F. 2020. Distribution and habitat preferences of Galápagos ants (Hymenoptera: Formicidae). Belgian Journal of Entomology, 93: 1–60.
- Heterick, B.E. 2021. A guide to the ants of Western Australia. Part I: Systematics. Records of the Western Australian Museum, Supplement 86, 1-245 (doi:10.18195/issn.0313-122x.86.2021.001-245).
- Heterick, B.E. 2022. A guide to the ants of Western Australia. Part II: Distribution and biology. Records of the Western Australian Museum, supplement 86: 247-510 (doi:10.18195/issn.0313-122x.86.2022.247-510).
- Imai, H.T., Kihara, A., Kondoh, M., Kubota, M., Kuribayashi, S., Ogata, K., Onoyama, K., Taylor, R.W., Terayama, M., Yoshimura, M., Ugawa, Y. 2003. Ants of Japan. 224 pp, Gakken, Japan.
- Lubertazzi, D. 2019. The ants of Hispaniola. Bulletin of the Museum of Comparative Zoology, 162(2), 59-210 (doi:10.3099/mcz-43.1).
- MacGown, J.A., Wetterer, J.K. 2013. Distribution and biological notes of Strumigenys margaritae (Hymenoptera: Formicidae: Dacetini). Terrestrial Arthropod Reviews 6, 247–255 (doi:10.1163/18749836-06001066).
- Meurgey, F. 2020. Challenging the Wallacean shortfall: A total assessment of insect diversity on Guadeloupe (French West Indies), a checklist and bibliography. Insecta Mundi 786: 1–183.
- Michlewicz, M. 2022. Strumigenys emmae (Emery, 1890) (Hymenoptera: Formicidae) in Poland – first record of this pantropic ant species from Europe with remarks on its biology. Annals of the Upper Silesian Museum in Bytom, Entomology 31 (online 007): 1-5 (doi:10.5281/ZENODO.6559247).
- Oussalah, N., Marniche, F., Espadaler, X., Biche, M. 2019. Exotic ants from the Maghreb (Hymenoptera, Formicidae) with first report of the hairy alien ant Nylanderia jaegerskioeldi (Mayr) in Algeria. Arxius de Miscel·lània Zoològica, 45–58 (doi:10.32800/amz.2019.17.0045).
- Pawluk, F., Borowiec, L., Salata, S. 2022. First record of Plagiolepis alluaudi Emery, 1894 (Hymenoptera: Formicidae) from Poland. Annals of the Upper Silesian Museum in Bytom Entomology 31 (online 006) 1-5 (doi:10.5281/ZENODO.6522444).
- Purkaret, A., Repta, F., Selnekovic, D., Jancik, L., Holecova, M. 2021. Notes on Strumigenys argiola (Emery, 1869) (Hymenoptera: Formicidae) with emphasis on its distribution, ecology and behaviour. Entomofauna Carpathica 33(2): 73-88.
- Rosas-Mejía, M., Guénard, B., Aguilar-Méndez, M. J., Ghilardi, A., Vásquez-Bolaños, M., Economo, E. P., Janda, M. 2021. Alien ants (Hymenoptera: Formicidae) in Mexico: the first database of records. Biological Invasions 23(6), 1669–1680 (doi:10.1007/s10530-020-02423-1).
- Sharaf, M.R., Fisher, B.L., Aldawood, S.A. 2014. Notes on ants of the genus Strumigenys F. Smith, 1860 (Hymenoptera: Formicidae) in the Arabian Peninsula, with a key to species. Sociobiology, 61, 293-299.
- Tang, K. L., Guénard, B. 2023. Further additions to the knowledge of Strumigenys (Formicidae: Myrmicinae) within South East Asia, with the descriptions of 20 new species. European Journal of Taxonomy 907, 1–144 (doi:10.5852/ejt.2023.907.2327).
- Tang, K.L., Pierce, M.P., Guénard, B. 2019. Review of the genus Strumigenys (Hymenoptera, Formicidae, Myrmicinae) in Hong Kong with the description of three new species and the addition of five native and four introduced species records. ZooKeys 831: 1–48 (DOI 10.3897/zookeys.831.31515).
- Varghese, T. 2004. Record of Strumigenys emmae (Emery) (Formicidae: Myrmicinae) from Bangalore, Karnataka and a key to Indian species. J. Bombay Nat. Hist. Soc. 101: 170-171 (page 170, record in India)
- Wang, C., Chung, F.-Y., Lin, C.-C., Gibson, J. C., McGuire, S., Suarez, A. V., Billen, J. 2023. The spongiform tissue in Strumigenys ants contains exocrine glands. Arthropod Structure & Development 73, 101246 (doi:10.1016/j.asd.2023.101246).
- Wang, C., Lin, C.-C., Keller, R.A., Billen, J. 2021. The ‘hairwheels’ in Strumigenys ants are not glandular. Asian Myrmecology 13: e013004 (doi:10.20362/am.013004).
- Wang, C., Sung, P.-J., Lin, C.-C., Ito, F., Billen, J. 2023. Parthenogenetic reproduction in Strumigenys ants: An update. Insects 14, 195 (doi:10.3390/insects14020195).
- Wang, W.Y., Soh, E.J.Y., Yong, G.W.J., Wong, M.K.L., Benoit Guénard, Economo, E.P., Yamane, S. 2022. Remarkable diversity in a little red dot: a comprehensive checklist of known ant species in Singapore (Hymenoptera: Formicidae) with notes on ecology and taxonomy. Asian Myrmecology 15: e015006 (doi:10.20362/am.015006).
- Wetterer, J.K. 2012. Worldwide spread of Emma's dacetine ant, Strumigenys emmae (Hymenoptera: Formicidae). Myrmecologial News 16: 69-74.
- Wetterer, J.K. 2017. Invasive ants of Bermuda revisited. Journal of Hymenoptera Research 54, 33–41 (doi:10.3897/jhr.54.11444).
- Wetterer, J.K. 2021. Ants (Hymenoptera, Formicidae) of St. Vincent, West Indies. Sociobiology 68, e6725 (doi:10.13102/sociobiology.v68i2.6725).
- Wetterer, J.K., Espadaler, X., Ashmole, N.P., Mendel, H., Cutler, C., Endeman, J. 2007. Ants (Hymenoptera: Formicidae) of the South Atlantic islands of Ascension Island, St Helena, and Tristan da Cunha. Myrmecological News 10: 29-37.
- Wheeler, W. M. 1908a. The ants of Porto Rico and the Virgin Islands. Bull. Am. Mus. Nat. Hist. 24: 117-158 (page 149, queen described)
References based on Global Ant Biodiversity Informatics
- Bolton B. 1983. The Afrotropical dacetine ants (Formicidae). Bulletin of the British Museum (Natural History). Entomology 46: 267-416.
- Bolton, B. 2000. The Ant Tribe Dacetini. Memoirs of the American Entomological Institute 65
- Brassard, F., Leong, C.-M., Chan, H.-H., Guénard, B. 2020. A new subterranean species and an updated checklist of Strumigenys (Hymenoptera, Formicidae) from Macao SAR, China, with a key to species of the Greater Bay Area. ZooKeys 970: 63–116 (doi:10.3897/zookeys.970.54958).
- Brown W. L., Jr. 1949. Revision of the ant tribe Dacetini: III. Epitritus Emery and Quadristruma new genus (Hymenoptera: Formicidae). Trans. Am. Entomol. Soc. 75: 43-51.
- Chapman, J. W., and Capco, S. R. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monogr. Inst. Sci. Technol. Manila 1: 1-327
- Donisthorpe H. 1916. Epitritus wheeleri, n. sp., an ant new to science; with notes on the genus Epitritus, Emery. Entomologist's Record and Journal of Variation 28: 121-122.
- Emery C. 1897. Formicidarum species novae vel minus cognitae in collectione Musaei Nationalis Hungarici quas in Nova-Guinea, colonia germanica, collegit L. Biró. Természetrajzi Füzetek 20: 571-599.
- Janda M., G. D. Alpert, M. L. Borowiec, E. P. Economo, P. Klimes, E. Sarnat, and S. O. Shattuck. 2011. Cheklist of ants described and recorded from New Guinea and associated islands. Available on http://www.newguineants.org/. Accessed on 24th Feb. 2011.
- Jourdan H., L. Bonnet de Larbogne, T. Potiaroa, and J. Chazeau. Invasions biologiques: le cas de la "fourmi électrique" Wasmannia auropunctata en Nouvelle Calédonie. Pp 156-164.
- Szabó J. 1909. De duabus speciebus novis Formicidarum generis Epitritus Em. Arch. Zool. (Budapest) 1: 27-28.
- Taylor R. W., and D. R. Brown. 1985. Formicoidea. Zoological Catalogue of Australia 2: 1-149.
- Viehmeyer H. 1912. Ameisen aus Deutsch Neuguinea gesammelt von Dr. O. Schlaginhaufen. Nebst einem Verzeichnisse der papuanischen Arten. Abhandlungen und Berichte des Königlichen Zoologischen und Anthropologische-Ethnographischen Museums zu Dresden 14: 1-26.
- Wetterer J. K. 2012. Worldwide spread of Emma's dacetine ant, Strumigenys emmae (Hymenoptera, Formicidae). Myrmecological News 16: 69-74.