Ponera xenagos
| Ponera xenagos | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Ponera |
| Species: | P. xenagos |
| Binomial name | |
| Ponera xenagos Wilson, 1957 | |
Relatively large for a Ponera, xenagos is only known from New Guinea.
Identification
Taylor (1967) - the largest known Ponera apart from Ponera alpha. Recognized by the combination of large size (head width 0.65-0.73 mm) with relatively broad head (cephalic index 84-90), vestigial medium clypeal tooth and relatively long scapes, which surpass the median occipital border by about 1/2 their maximum thickness. The eyes are small (4-5 indistinct facets) , and no antennal club is differentiated. Posterolateral propodeal angles not markedly raised, and node with more rounded contours than in alpha or selenophora, although it still maintains the same basic form with arched anterolateral and posterodorsal faces.
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 4.966666667° to -7.55°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Indo-Australian Region: New Guinea (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
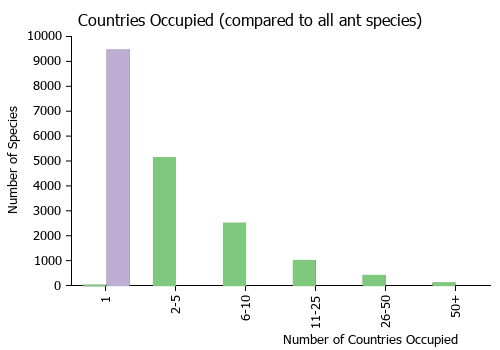
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Wilson collected specimens from partial clearings beside trails in dense midmountain rain forest. He found one nest at each site, under loose bark on a rotting stump at Tumnang, and on the upper surface of a large rotting log at Ebaabang. Both nest sites were in partial clearings at the side of native trails running through dense midmountain rainforest. My Aiyura (Taylor) samples were taken in disturbed midmountain forest, nesting in rotting logs.
|
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- xenagos. Ponera xenagos Wilson, 1957b: 385 (w.) NEW GUINEA. See also: Taylor, 1967a: 43.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Holotype. HW 0.67 mm, HL 0.77 mm, SL 0.59 mm, CI 87, SI 88, PW 0.52 mm, petiole height 0.53 mm, petiolar node length 0.27 mm, dorsal petiole width 0.42 mm. Very similar to P. selenophora Emery, differing by the following characters:
(1) Larger size.
(2) The three apical mandibular teeth occupy less than half the masticatory border, and distinct teeth are not developed on the basal half of the border as described for selenophora.
(3) The posterolateral margins of the propodeum are less pronounced; viewed from directly above they form an angle of only a little less than 90°.
(4) When viewed from directly above, the posterior margin of the petiolar node is almost perfectly straight, as opposed to the distinctly concave margin of selenophora.
(5) Pubescence is generally sparser. The anterior face of the petiolar node has pubescence only over its upper quarter, and there it is relatively sparse, whereas in selenophora it is abundant over the entire upper half.
Paratype variation. HW 0.65-0.68 mm, HL 0.75-0.80 mm, SL 0.57-0.60 mm, CI 86-90, SI 83-89, PW 0.52-0.54 mm, dorsal petiole width 0.40-0.44 mm.
Immature Forms
Taylor (1967) - Larvae from Aiyura have 3 pairs of dorsal abdominal glutinous tubercles, as usual in Indo-Australian Ponera; the worker pupae are enclosed in cocoons (other castes not available).
Type Material
N-E. NEW GUINEA: Tumnang, 1500 m. (type locality), April 14-15, 1955, holotype and eight paratype workers (Wilson, acc. no. 801); Ebabaang, 1300-1400 m., April 16-18, 1955, three paratype workers (Wilson, acc. no. 819). Both of the above localities are in the Mongi River Watershed of the Huon Peninsula. Museum of Comparative Zoology.
References
- Taylor, R. W. 1967a. A monographic revision of the ant genus Ponera Latreille (Hymenoptera: Formicidae). Pac. Insects Monogr. 13: 1-112 (page 43, see also)
- Wilson, E. O. 1957b. The tenuis and selenophora groups of the ant genus Ponera (Hymenoptera: Formicidae). Bulletin of the Museum of Comparative Zoology 116: 355-386 (page 385, worker described)
References based on Global Ant Biodiversity Informatics
- CSIRO Collection
- Janda M., G. D. Alpert, M. L. Borowiec, E. P. Economo, P. Klimes, E. Sarnat, and S. O. Shattuck. 2011. Cheklist of ants described and recorded from New Guinea and associated islands. Available on http://www.newguineants.org/. Accessed on 24th Feb. 2011.
- Lucky A., E. Sarnat, and L. Alonso. 2011. Ants of the Muller Range, Papua New Guinea, Chapter 10. In Richards, S. J. and Gamui, B. G. (editors). 2013. Rapid Biological Assessments of the Nakanai Mountains and the upper Strickland Basin: surveying the biodiversity of Papua New Guineas sublime karst environments. RAP Bulletin of Biological Assessment 60. Conservation International. Arlington, VA.
- Lucky A., K. Sagata, and E. Sarnat. 2011. Ants of the Nakanai Mountains, East New Britain Province, Papua New Guinea, Chapter 1. In Richards, S. J. and Gamui, B. G. (editors). 2013. Rapid Biological Assessments of the Nakanai Mountains and the upper Strickland Basin: surveying the biodiversity of Papua New Guineas sublime karst environments. RAP Bulletin of Biological Assessment 60. Conservation International. Arlington, VA.
- Taylor R. W. 1967. A monographic revision of the ant genus Ponera Latreille (Hymenoptera: Formicidae). Pacific Insects Monograph 13: 1-112.
- Wilson E. O. 1957. The tenuis and selenophora groups of the ant genus Ponera (Hymenoptera: Formicidae). Bulletin of the Museum of Comparative Zoology 116: 355-386.
- Wilson E. O. 1958. Studies on the ant fauna of Melanesia III. Rhytidoponera in western Melanesia and the Moluccas. IV. The tribe Ponerini. Bulletin of the Museum of Comparative Zoology 119: 303-371.
- Wilson Edward O. 1959. Adaptive Shift and Dispersal in a Tropical Ant Fauna. Evolution 13(1): 122-144
- Woodcock P., D. P. Edwards, T. M. Fayle, R. J. Newton, C. Vun Khen, S. H. Bottrell, and K. C. Hamer. 2011. The conservation value of South East Asia's highly degraded forests: evidence from leaf-litter ants. Phil. Trans. R. Soc. B. 366: 3256-3264.

