Aphaenogaster treatae
| Aphaenogaster treatae | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Stenammini |
| Genus: | Aphaenogaster |
| Species: | A. treatae |
| Binomial name | |
| Aphaenogaster treatae Forel, 1886 | |
| Synonyms | |
| |
Aphaenogaster treatae, unlike many of its North American congeners, is typically found in open habitats such as fields and pine barrens. In Florida it occurs in sandy uplands. It is often difficult to find even where it is known to occur (Deyrup, 2016). It is known to remove (Stamp & Lucas, 1990; Atchison & Lucky, 2022) and disperse (Hilley & Thiet, 2015) seeds .
Photo Gallery
Identification
Note flat lobes at the inner bases of each antennal scape.
Aphaenogaster treatae is similar to Aphaenogaster ashmeadi, but has a larger lobe at the base of the scape. The lobe is one-fourth the length of the scape. (DeMarco, 2015)
Florida
Deyrup (2016) - This is a large species resembling Aphaenogaster ashmeadi. Both species have a conspicuous lobe at the base of the antennal scape; this might serve to protect the joint at the base of the antennae, but if this is the case, one might expect to see the structure in more species of ants. In treatae, this lobe is thickened, with a distinct lateral face, while in ashmeadi the lobe is thin, although it may look thicker from some angles because its dorsal surface is convex. In peninsular Florida, treatae is reddish brown, while ashmeadi is blackish, but in the Panhandle, both species may be reddish brown.
Keys including this Species
Distribution
Florida
Deyrup (2016) - A. treatae occurs from Ontario south into Florida, west into Michigan, Oklahoma and Texas. In Florida, treatae occurs in sandy uplands from Collier County northward and west through the Panhandle, although there are many large gaps in this distribution. This species is often difficult to find even where it is known to occur.
Latitudinal Distribution Pattern
Latitudinal Range: 43.67° to 20.84539°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Nearctic Region: United States (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
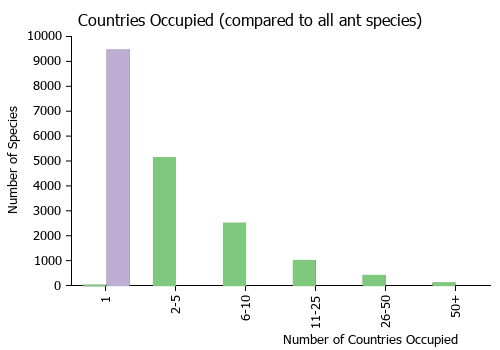
|
Biology
This species nests between clumps of grass in sandy, semi-open areas.
Florida
Deyrup (2016) - The following paragraph is summarized from Carroll (1975). Nests are in well-drained sandy soil, generally in open stands of pines and oaks. In Florida, the nest entrances are usually at the base of a clump of grasses or herbs, while in northern parts of its range, treatae is often associated with rocks or logs. Nests have one or two entrances, one of which is usually marked by a small turret of plant debris. Nests go down to approximately 45 cm, with pupae and prepupae in a superficial chamber and larvae in deeper chambers. Foraging is on the surface of the ground, always in open to partially shaded areas. Workers commonly collect arthropods, occasionally collect seeds and bits of Russula mushrooms. Alates are in nests from mid-May through mid-July. Florida colonies usually have approximately 200 workers.
In Michigan, treatae was studied in detail by Talbot (1954, 1966). Nests often had a chamber that was mostly above ground and roofed with a thatch of plant material and soil, presumably for warming pupae or prepupae when the soil temperature was cool. Talbot made a complete inventory of the individuals in 30 nests. Numbers of workers ranged from 65 to 1662, with an average of 682. The locations of nests were also plotted in a sandy, abandoned field. In a study plot 100 feet by 120 feet, there were 63 colonies, providing an average of 32 workers that might forage over every square yard. Alates fly under precisely controlled conditions, when the temperature is 78°F-88°F, the air is calm, the sun is partly or completely obscured by clouds, but the ambient light remains relatively high and there is no rain (Talbot 1966). Alates sometimes wait in the nest entrance and rush out as soon as a cloud obscures the sun. In North Carolina, Carter (1962) found treatae common throughout the state, living primarily in fields and in open forests, and only rarely in closed, mesic forests.
Association with Other Organisms
 Explore: Show all Associate data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Associate data or Search these data. See also a list of all data tables or learn how data is managed.
- This species is a host for the cricket Myrmecophilus pergandei (a myrmecophile) in United States.
Life History Traits
- Queen number: monogynous (Rissing and Pollock, 1988; Frumhoff & Ward, 1992)
Castes
Worker
Images from AntWeb
    
| |
| Worker. Specimen code casent0103609. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Queen
Images from AntWeb
    
| |
| Queen (alate/dealate). Specimen code casent0103608. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Male
Images from AntWeb
    
| |
| Male (alate). Specimen code casent0103605. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- treatae. Aphaenogaster treatae Forel, 1886b: xl (w.q.m.) U.S.A. (New Jersey).
- Type-material: lectotype worker (by designation of Mackay & Mackay, 2017: 425), 6 paralectotype workers, 3 paralectotype queens, 3 paralectotype males.
- Type-locality: lectotype U.S.A.: New Jersey, Vineland (M. Treat); paralectotypes with same data.
- Type-depository: MHNG.
- Combination in Stenamma (Aphaenogaster): Emery, 1895c: 302;
- combination in Aphaenogaster: Wheeler, W.M. 1913c: 114;
- combination in Aphaenogaster (Attomyrma): Emery, 1921f: 60.
- Status as species: Mayr, 1886d: 443 (in key); Cresson, 1887: 260; Dalla Torre, 1893: 107; Emery, 1895c: 302; Wheeler, W.M. 1904e: 303; Wheeler, W.M. 1905f: 383; Wheeler, W.M. 1906b: 5; Wheeler, W.M. 1910g: 565; Wheeler, W.M. 1913c: 114; Wheeler, W.M. 1916m: 586; Emery, 1921f: 60; Wheeler, G.C. & Wheeler, E.W. 1934: 6 (redescription); Dennis, 1938: 284; Wesson, L.G. & Wesson, R.G. 1940: 93; Buren, 1944a: 285; Creighton, 1950a: 153; Smith, M.R. 1951a: 798; Carter, 1962a: 6 (in list); Smith, D.R. 1979: 1363; DuBois & LaBerge, 1988: 138; Deyrup, et al. 1989: 95; Wheeler, G.C., et al. 1994: 302; Bolton, 1995b: 73; Deyrup, 2003: 44; Coovert, 2005: 51; MacGown & Forster, 2005: 71; Ellison, et al. 2012: 233; Shattuck & Cover, 2016: 14; Deyrup, 2017: 51; Mackay & Mackay, 2017: 425 (redescription).
- Senior synonym of pluteicornis: Shattuck & Cover, 2016: 14; Mackay & Mackay, 2017: 425.
- Senior synonym of wheeleri: Creighton, 1950a: 153; Smith, M.R. 1958c: 119; Bolton, 1995b: 73; Mackay & Mackay, 2017: 425.
- Material of the unavailable name alabamensis referred here by Creighton, 1950a: 153; Smith, M.R. 1958c: 119; Smith, D.R. 1979: 1363; Bolton, 1995b: 73; Shattuck & Cover, 2016: 14; Mackay & Mackay, 2017: 426
- Material of the unavailable name oklahomensis referred here by Shattuck & Cover, 2016: 13; Mackay & Mackay, 2017: 426.
- Distribution: Canada, U.S.A.
- pluteicornis. Aphaenogaster treatae subsp. pluteicornis Wheeler, G.C. & Wheeler, E.W. 1934: 7 (w.q.m.) U.S.A. (Oklahoma, Texas).
- Type-material: syntype workers (number not stated, “numerous”), 4 syntype queens, 4 syntype males.
- Type-locality: U.S.A.: Oklahoma, Poteau, 17.vi.1929 (G.C. Wheeler & E.W. Wheeler).
- Type-depository: LACM (perhaps also DRIR).
- Wheeler, G.C. & Wheeler, J. 1953b: 62 (l.).
- Combination in Aphaenogaster (Attomyrma): Creighton, 1950a: 154.
- Subspecies of treatae: Wesson, L.G. & Wesson, R.G. 1940: 93; Creighton, 1950a: 154; Smith, M.R. 1951a: 798; Smith, M.R. 1958c: 118; Smith, D.R. 1979: 1363; Bolton, 1995b: 72.
- Junior synonym of treatae: Shattuck & Cover, 2016: 14; Mackay & Mackay, 2017: 425.
- wheeleri. Aphaenogaster treatae subsp. wheeleri Mann, 1915: 51, fig. 1a (w.q.) U.S.A. (Massachusetts).
- Type-material: syntype workers (number not stated, “numerous”), 2 syntype queens.
- Type-locality: U.S.A.: Massachusetts, Naushon I. (opposite Woods Hole), viii.1915 (F.X. Williams & W.M. Mann).
- Type-depository: USNM.
- Subspecies of treatae: Wheeler, G.C. & Wheeler, E.W. 1934: 11 (in key); Smith, M.R. 1951a: 798.
- Junior synonym of treatae: Creighton, 1950a: 153; Smith, M.R. 1958c: 119; Smith, D.R. 1979: 1363; Bolton, 1995b: 74; Mackay & Mackay, 2017: 425.
Type Material
- Aphaenogaster treatae: Syntype, workers, Vineland, New Jersey, United States, 39°29′0″N 75°2′0″W / 39.483333°N 75.033333°W, Mary Treat, CASENT0907701, Musee d'Histoire Naturelle Genève; (specimen labeled as lectotype but designation unpublished).
- Aphaenogaster treatae pluteicornis: Syntype, numerous workers, 4 queens, 4 males, a few larvae and pupae, Poteau, Oklahoma, United States, 35°03′0″N 94°37′0″W / 35.05°N 94.616667°W, 17 June 1929, G.C. & E.W. Wheeler, Los Angeles County Museum of Natural History; on a steep slope in the soil and under dead leaves beside a stone.
Note: A specimen (BMNH(E)1013809; CASENT0900414) in The Natural History Museum from Woods Hole, Massachusetts is labelled as a "cotype" but is not a true type as it is not from the type locality.
Taxonomic Notes
The subspecies A. treatae pluteicornis has been described for the population occurring from Alabama into Texas; the two intergrade in Alabama (Creighton 1950), and there is no reason to believe that this subspecies is actually a separate species (Deyrup, 2016).
Description
Karyotype
- See additional details at the Ant Chromosome Database.
 Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
- 2n = 42 (USA) (Crozier, 1977).
Name Derivation
August Forel named this species for Mary Treat, who sent him the type specimen. Forel named both Aphaenogaster mariae and A. treatae for Mary Treat in the same paper (Forel 1886).
Mary Treat (1830-1923) was an important self-taught scientist who wrote several books on natural history, including one on butterflies, her work often being published in popular magazines rather than scientific journals. In January, 1879, she published an extensive account of a colony of a slave-making Formica, with detailed descriptions of raids and some preliminary experimental manipulations. Lacking scientific credentials, and perhaps primarily interested in stimulating general interest in the natural history of ants, Treat published her paper in Harper's Monthly Magazine. Treat's observations are still useful and raise some interesting questions, assuming the account is accurate. There are plenty of startling anthropomorphisms, as one might expect for the time, considering that the first volumes of Fabre's Souvenirs Entomologiques (which mark the beginning of our modern understanding of insect behavior) were also published in 1879. Her work was noted by Charles Darwin in his book on insectivorous plants, and the two exchanged letters on the subject. Many of her studies were done in her yard and garden in New Jersey, or in the nearby Jersey Pine Barrens. A patient, intelligent, original observer and excellent writer, she might have been an academic star if she had been born at a time or place when there was less disparagement of what was called "the female intellect." (Deyrup, 2016)
References
- Atchison, R. A., Lucky, A. 2022. Diversity and resilience of seed-removing ant species in Longleaf Sandhill to frequent fire. Diversity 14, 1012 (doi:10.3390/d14121012).
- Bolton, B. 1995b. A new general catalogue of the ants of the world. Cambridge, Mass.: Harvard University Press, 504 pp. (page 73, catalogue)
- Carroll, T.M. 2011. The ants of Indiana (Hymenoptera: Formicidae). M.S. thesis, Purdue University.
- Creighton, W. S. 1950a. The ants of North America. Bulletin of the Museum of Comparative Zoology 104: 1-585 (page 153, Senior synonym of wheeleri, and material of the unavailable name alabamensis referred here.)
- Davis, T. 2009. The ants of South Carolina (thesis, Clemson University).
- DeMarco, B.B. 2015. Phylogeny of North American Aphaenogaster species (Hymenoptera: Formicidae) reconstructed with morphological and DNA data. Ph.D. thesis, Michigan State University.
- Deyrup, M.A. 2016. Ants of Florida: Identification and Natural History. CRC Press, 423 pp.
- Emery, C. 1895d. Beiträge zur Kenntniss der nordamerikanischen Ameisenfauna. (Schluss). Zool. Jahrb. Abt. Syst. Geogr. Biol. Tiere 8: 257-360 (page 302, Combination in Stenamma (Aphaenogaster))
- Emery, C. 1921c. Hymenoptera. Fam. Formicidae. Subfam. Myrmicinae. [part]. Genera Insectorum 174A:1-94 94: 1-94 + 7 (page 60, Combination in Aphaenogaster (Attomyrma))
- Forel, A. 1886b. Espèces nouvelles de fourmis américaines. Ann. Soc. Entomol. Belg. 30:xxxviii-xlix. (page xl, worker, queen, male described)
- Hill, J.G. 2015. Ants (Hymenoptera: Formicidae) of the Big Thicket Region of Texas. Midsouth Entomologist 8: 24-34.
- Hilley, E., Thiet, R. 2015. Vulnerable broom crowberry (Corema conradii) benefits from ant seed dispersal in coastal US heathlands. Plant Ecology 216, 1091–1101.
- Ipser, R.M., Brinkman, M.A., Gardner, W.A., Peeler, H.B. 2004. A survey of ground-dwelling ants (Hymenoptera: Formicidae) in Georgia. Florida Entomologist 87: 253-260.
- Ivanov, K. 2019. The ants of Ohio (Hymenoptera, Formicidae): an updated checklist. Journal of Hymenoptera Research 70: 65–87 (doi:10.3897@jhr.70.35207).
- MacGown, J.A., Booher, D., Richter, H., Wetterer, J.K., Hill, J.G. 2021. An updated list of ants of Alabama (Hymenoptera: Formicidae) with new state records. Transactions of the American Entomological Society 147: 961-981 (doi:10.3157/061.147.0409).
- Mann, W. M. 1915. A new form of a southern ant from Naushon Island, Massachusetts. Psyche (Camb.) 22: 51.
- Moura, M.N., Cardoso, D.C., Cristiano, M.P. 2020. The tight genome size of ants: diversity and evolution under ancestral state reconstruction and base composition. Zoological Journal of the Linnean Society, zlaa135 (doi:10.1093/zoolinnean/zlaa135).
- Shattuck, S.O., Cover, S. 2016. Taxonomy of some little-understood North American ants (Hymenoptera: Formicidae). Zootaxa 4175: 10–22 (doi:10.11646/zootaxa.4175.1.2).
- Stamp, N.E., Lucas, J.R. 1990. Spatial patterns and dispersal distances of explosively dispersing plants in Florida sandhill vegetation. Journal of Ecology 78, 589–600.
- Talbot, M. 1954. 1954. Populations of the ant Aphaenogaster (Attomyrma) treatae Forel on abandoned fields on the Edwin S. George Reserve. Contributions of the Laboratory of Vertebrate at the University of Michigan 69, 9 p.
- Tschinkel WR. 2011. The nest architecture of three species of north Florida Aphaenogaster ants. Journal of Insect Science 11:105.
- Waters, J.S., Keough, N.W., Burt, J., Eckel, J.D., Hutchinson, T., Ewanchuk, J., Rock, M., Markert, J.A., Axen, H.J., Gregg, D. 2022. Survey of ants (Hymenoptera, Formicidae) in the city of Providence (Rhode Island, United States) and a new northern-most record for Brachyponera chinensis (Emery, 1895). Check List 18(6), 1347–1368 (doi:10.15560/18.6.1347).
- Wheeler, W. M. 1913d. Ants collected in Georgia by Dr. J. C. Bradley and Mr. W. T. Davis. Psyche (Camb.) 20: 112-117 (page 114, Combination in Aphaenogaster)
References based on Global Ant Biodiversity Informatics
- Annotated Ant Species List Ordway-Swisher Biological Station. Downloaded at http://ordway-swisher.ufl.edu/species/os-hymenoptera.htm on 5th Oct 2010.
- Atchison R. A., J. Hulcr, and A. Lucky. 2018. Managed fire frequency significantly influences the litter arthropod community in longleaf pine flatwoods. Environmental Entomology 20(10): 1-11.
- Buren W. F. 1944. A list of Iowa ants. Iowa State College Journal of Science 18:277-312
- Callcott A. M. A., D. H. oi, H. L. Collins, D. F. Williams, and T. C. Lockley. 2000. Seasonal Studies of an Isolated Red Imported Fire Ant (Hymenoptera: Formicidae) Population in Eastern Tennessee. Environmental Entomology, 29(4): 788-794.
- Coovert G. A. 2005. The Ants of Ohio (Hymenoptera: Formicidae). Ohio Biological Survey, Inc. 15(2): 1-207.
- Coovert, G.A. 2005. The Ants of Ohio (Hymenoptera: Formicidae) Ohio Biological Survey Bulletin New Series Volume 15(2):1-196
- Davis W. T., and J. Bequaert. 1922. An annoted list of the ants of Staten Island and Long Island, N. Y. Bulletin of the Brooklyn Entomological Society 17(1): 1-25.
- DeMarco B. B., and A. I. Cognato. 2016. A multiple-gene phylogeny reveals polyphyly among eastern North American Aphaenogaster species (Hymenoptera: Formicidae). Zoologica Scripta DOI: 10.1111/zsc.12168
- Del Toro, I. 2010. PERSONAL COMMUNICATION. MUSEUM RECORDS COLLATED BY ISRAEL DEL TORO
- Deyrup M., C. Johnson, G. C. Wheeler, J. Wheeler. 1989. A preliminary list of the ants of Florida. Florida Entomologist 72: 91-101
- Deyrup M., L. Deyrup, and J. Carrel. 2013. Ant Species in the Diet of a Florida Population of Eastern Narrow-Mouthed Toads, Gastrophryne carolinensis. Southeastern Naturalist 12(2): 367-378.
- Deyrup, M. and J. Trager. 1986. Ants of the Archbold Biological Station, Highlands County, Florida (Hymenoptera: Formicidae). Florida Entomologist 69(1):206-228
- DuBois M. B. 1985. Distribution of ants in Kansas: subfamilies Ponerinae, Ecitoninae, and Myrmicinae (Hymenoptera: Formicidae). Sociobiology 11: 153-431
- DuBois M. B. 1985. Distribution of ants in Kansas: subfamilies Ponerinae, Ecitoninae, and Myrmicinae (Hymenoptera: Formicidae). Sociobiology 11: 153-432
- DuBois M. B. 1985. Distribution of ants in Kansas: subfamilies Ponerinae, Ecitoninae, and Myrmicinae (Hymenoptera: Formicidae). Sociobiology 11: 153-433
- DuBois M. B. 1985. Distribution of ants in Kansas: subfamilies Ponerinae, Ecitoninae, and Myrmicinae (Hymenoptera: Formicidae). Sociobiology 11: 153-434
- Dubois, M.B. and W.E. Laberge. 1988. An Annotated list of the ants of Illionois. pages 133-156 in Advances in Myrmecology, J. Trager
- Ellison A. M., and E. J. Farnsworth. 2014. Targeted sampling increases knowledge and improves estimates of ant species richness in Rhode Island. Northeastern Naturalist 21(1): NENHC-13NENHC-24.
- Emery C. 1895. Beiträge zur Kenntniss der nordamerikanischen Ameisenfauna. (Schluss). Zoologische Jahrbücher. Abteilung für Systematik, Geographie und Biologie der Tiere 8: 257-360.
- Forster J.A. 2005. The Ants (hymenoptera: Formicidae) of Alabama. Master of Science, Auburn University. 242 pages.
- Frye J. A., T. Frye, and T. W. Suman. 2014. The ant fauna of inland sand dune communities in Worcester County, Maryland. Northeastern Naturalist, 21(3): 446-471.
- General D. M., and L. C. Thompson. 2011. New Distributional Records of Ants in Arkansas for 2009 and 2010 with Comments on Previous Records. Journal of the Arkansas Academy of Science 65: 166-168.
- General D.M. & Thompson L.C. 2008. New Distributional Records of Ants in Arkansas for 2008. Journal of the Arkansas Academy of Science. 63: 182-184
- Graham, J.H., A.J. Krzysik, D.A. Kovacic, J.J. Duda, D.C. Freeman, J.M. Emlen, J.C. Zak, W.R. Long, M.P. Wallace, C. Chamberlin-Graham, J.P. Nutter and H.E. Balbach. 2008. Ant Community Composition across a Gradient of Disturbed Military Landscapes at Fort Benning, Georgia. Southeastern Naturalist 7(3):429-448
- Guénard B., K. A. Mccaffrey, A. Lucky, and R. R. Dunn. 2012. Ants of North Carolina: an updated list (Hymenoptera: Formicidae). Zootaxa 3552: 1-36.
- Hill J. G., K. S. Summerville, and R. L. Brown. 2008. Habitat Associations of Ant Species (Hymenoptera: Formicidae) in a Heterogeneous Mississippi Landscape. Environ. Entomol. 37(2): 453-463.
- Hill J. V. 2012. The ant (Hymenoptera: Formicidae) fauna of the cedar glades and xeric limestone prairies of the Central Basin of Tennessee. Journal of the Tennessee Academy of Science 87(3): 135.
- Hill J.G. & Brown R. L. 2010. The Ant (Hymenoptera: Formicidae) Fauna of Black Belt Prairie Remnants in Alabama and Mississippi. Southeastern Naturalist. 9: 73-84
- Hill, J.G. 2006. Ants collected at Okatibbee Lake, Lauderdale County, Mississippi
- Ivanov, K. 2019. The ants of Ohio (Hymenoptera, Formicidae): an updated checklist. Journal of Hymenoptera Research 70: 65–87.
- Ivanov K., L. Hightower, S. T. Dash, and J. B. Keiper. 2019. 150 years in the making: first comprehensive list of the ants (Hymenoptera: Formicidae) of Virginia, USA. Zootaxa 4554 (2): 532–560.
- Johnson C. 1986. A north Florida ant fauna (Hymenoptera: Formicidae). Insecta Mundi 1: 243-246
- Lubertazi, D. Personal Communication. Specimen Data from Museum of Comparative Zoology at Harvard
- Lynch J. F. 1988. An annotated checklist and key to the species of ants (Hymenoptera: Formicidae) of the Chesapeake Bay region. The Maryland Naturalist 31: 61-106
- MacGown J. A., J. G. Hill, R. L. Brown, T. L. Schiefer, J. G. Lewis. 2012. Ant diversity at Noxubee National Wildlife Refuge in Oktibbeha, Noxubee, and Winston Counties, Mississippi. Mississippi Agricultural and Forestry Experiment Station Bulletin 1197: 1-30
- MacGown J. A., J. G. Hill, and R. L. Brown. 2010. Native and exotic ant in Mississippi state parks. Proceedings: Imported Fire Ant Conference, Charleston, South Carolina, March 24-26, 2008: 74-80.
- MacGown J. A., and R. L. Brown. 2006. Survey of the ants (Hymenoptera: Formicidae) of the Tombigbee National Forest in Mississippi. Journal of the Kansas Entomological Society 79(4):325-340.
- MacGown, J.A and J.A. Forster. 2005. A preliminary list of the ants (Hymenoptera: Formicidae) of Alabama, U.S.A. Entomological News 116(2):61-74
- MacGown, J.A. and JV.G. Hill. Ants of the Great Smoky Mountains National Park (Tennessee and North Carolina).
- MacGown, J.A. and R.L. Brown. 2006. Survey of the ants (Hymenoptera: Formicidae) of the Tombigbee National Forest in Mississippi. Journal of the Kansas Entomological Society 79(4):325-340.
- MacGown, J.A., J.G. Hill, R.L. Brown and T.L. 2009. Ant Diversity at Noxubee National Wildlife Refuge in Oktibbeha, Noxubee, and Winston Counties, Mississippi Report #2009-01. Schiefer. 2009.
- MacGown. J. 2011. Ants collected during the 25th Annual Cross Expedition at Tims Ford State Park, Franklin County, Tennessee
- Mann W. M. 1915. A new form of a southern ant from Naushon Island, Massachusetts. Psyche (Cambridge) 22: 51.
- Nuhn, T.P. and C.G. Wright. 1979. An Ecological Survey of Ants (Hymenoptera: Formicidae) in a Landscaped Suburban Habitat. American Midland Naturalist 102(2):353-362
- O'Keefe S. T., J. L. Cook, T. Dudek, D. F. Wunneburger, M. D. Guzman, R. N. Coulson, and S. B. Vinson. 2000. The Distribution of Texas Ants. The Southwestern Entomologist 22: 1-92.
- Rowles, A.D. and J. Silverman. 2009. Carbohydrate supply limits invasion of natural communities by Argentine ants. Oecologia 161(1):161-171
- Smith M. R. 1935. A list of the ants of Oklahoma (Hymen.: Formicidae). Entomological News 46: 235-241.
- Smith M. R. 1962. A new species of exotic Ponera from North Carolina (Hymenoptera, Formicidae). Acta Hymenopterologica 1: 377-382.
- Spiesman B. J. and G. S. Cumming. 2008. Communities in context: the influences of multiscale environmental variation on local ant community structure. Landscape Ecol. 23: 313-325
- Sturtevant A. H. 1931. Ants collected on Cape Cod, Massachusetts. Psyche (Cambridge) 38: 73-79
- Talbot M. 1976. A list of the ants (Hymenoptera: Formicidae) of the Edwin S. George Reserve, Livingston County, Michigan. Great Lakes Entomologist 8: 245-246.
- Toennisson T. A., N. J. Sanders, W. E. Klingeman, and K. M. Vail. 2011. Influences on the Structure of Suburban Ant (Hymenoptera: Formicidae) Communities and the Abundance of Tapinoma sessile. Environ. Entomol. 40(6): 1397-1404.
- Van Pelt A. F. 1966. Activity and density of old-field ants of the Savannah River Plant, South Carolina. Journal of the Elisha Mitchell Scientific Society 82: 35-43.
- Van Pelt A., and J. B. Gentry. 1985. The ants (Hymenoptera: Formicidae) of the Savannah River Plant, South Carolina. Dept. Energy, Savannah River Ecology Lab., Aiken, SC., Report SRO-NERP-14, 56 p.
- Warren, L.O. and E.P. Rouse. 1969. The Ants of Arkansas. Bulletin of the Agricultural Experiment Station 742:1-67
- Wheeler G. C., J. N. Wheeler, and P. B. Kannowski. 1994. Checklist of the ants of Michigan (Hymenoptera: Formicidae). The Great Lakes Entomologist 26(4): 297-310
- Wheeler G. C., and E. W. Wheeler. 1934. New forms of Aphaenogaster treatae Forel from the southern United States (Hym.: Formicidae). Psyche (Cambridge) 41: 6-12.
- Wheeler G. C., and J. Wheeler J. 1989. A checklist of the ants of Oklahoma. Prairie Naturalist 21: 203-210.
- Wheeler W. M. 1904. The ants of North Carolina. Bulletin of the American Museum of Natural History. 20: 299-306.
- Wheeler W. M. 1905. An annotated list of the ants of New Jersey. Bulletin of the American Museum of Natural History. 21: 371-403.
- Wheeler W. M. 1919. A new subspecies of Aphaenogaster treatae Forel. Psyche (Cambridge) 26: 50.
- Wheeler, G.C. and J. Wheeler. 1985. A checklist of Texas ants. Prairie Naturalist 17:49-64.
- Wheeler, G.C., J. Wheeler and P.B. Kannowski. 1994. CHECKLIST OF THE ANTS OF MICHIGAN (HYMENOPTERA: FORMICIDAE). Great Lakes Entomologist 26:1:297-310
- Young J., and D. E. Howell. 1964. Ants of Oklahoma. Miscellaneous Publication. Oklahoma Agricultural Experimental Station 71: 1-42.
- Young, J. and D.E. Howell. 1964. Ants of Oklahoma. Miscellaneous Publications of Oklahoma State University MP-71
- Zettler J. A., M. D. Taylor, C. R. Allen, and T. P. Spira. 2004. Consequences of Forest Clear-Cuts for Native and Nonindigenous Ants (Hymenoptera: Formicidae). Ann. Entomol. Soc. Am. 97(3): 513-518.





