Anochetus horridus
| Anochetus horridus | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Anochetus |
| Species: | A. horridus |
| Binomial name | |
| Anochetus horridus Kempf, 1964 | |
A. horridus, in spite of its habitus, has only been taken on the ground in humus or leaf litter, but it may well be an epigaeic forager, in consonance with its large eyes and long mandibles (Brown 1978).
Identification
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 5.266667° to -64.3°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Brazil (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
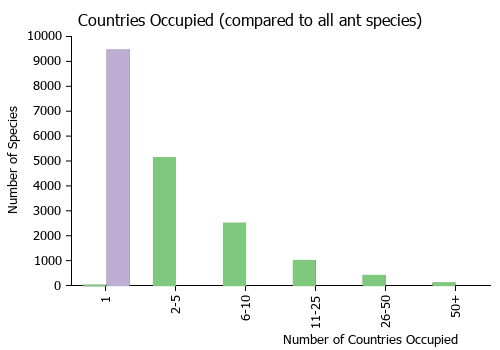
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Gibson et al. (2018) found the trap-jaws of Anochetus horridus and Anochetus emarginatus have slower strikes relative to the other species of Anochetus and Odontomachus, reaching mean maximum rotational velocity and acceleration of around 1.3 9 10 4 rad s -1 and 2 9 10 8 rad s -2, respectively. The mass-specific power output of these and other species in these two genera confirm their mandible strikes are power amplified, i.e., in addition to muscle contraction energy, the acceleration of the mandibles is enhanced by mechanical structures that are adapted for, and form part of, their trap-jaws. These species and other measured species in Anochetus and Odontomachus follow a pattern of increasingly energetic strikes with larger body mass. This relationship appears to be primarily driven by an increase in mandible mass with larger body size.
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- horridus. Anochetus (Stenomyrmex) horridus Kempf, 1964f: 239, figs. 4, 8, 12, 14 (gynecoid w.) BRAZIL (Pará, Amazonas).
- Type-material: holotype ergatoid queen, paratype workers (number not stated, “a small series”).
- Type-locality: holotype Brazil: Pará, Belém, 12-19.viii.1962, no. 2272, in soil cover (K. Lenko); paratypes: Brazil: Amazonas, on Manaus-Itacoatiara road, km, 49, 24.viii.1962, M-65 (W.L. Brown).
- Type-depositories: DZSP (holotype); MCZC, MZSP (paratypes).
- Wheeler, G.C. & Wheeler, J. 1971b: 1212 (l.).
- Status as species: Kempf, 1970b: 327; Kempf, 1972a: 22; Brown, 1978c: 557, 612; Bolton, 1995b: 64; Zabala, 2008: 131; Feitosa, 2015c: 98; Fernández & Guerrero, 2019: 516.
- Distribution: Brazil, Colombia, Venezuela.
Description
Karyotype
- See additional details at the Ant Chromosome Database.
 Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Karyotype data or Search these data. See also a list of all data tables or learn how data is managed.
- n = 23, 2n = 46, karyotype = 14M + 32A (French Guiana) (Mariano et al., 2011; Santos et al., 2010; Mariano et al., 2015) (karyotype originally as 8M+4SM+34T).
References
- Aguiar, H.J.A.C., Barros, L.A.C., Silveira, L.I., Petitclerc, F., Etienne, S., Orivel, J. 2020. Cytogenetic data for sixteen ant species from North-eastern Amazonia with phylogenetic insights into three subfamilies. Comparative Cytogenetics 14(1): 43–60 (doi:10.3897/CompCytogen.v14i1.46692).
- Albuquerque, E., Prado, L., Andrade-Silva, J., Siqueira, E., Sampaio, K., Alves, D., Brandão, C., Andrade, P., Feitosa, R., Koch, E., Delabie, J., Fernandes, I., Baccaro, F., Souza, J., Almeida, R., Silva, R. 2021. Ants of the State of Pará, Brazil: a historical and comprehensive dataset of a key biodiversity hotspot in the Amazon Basin. Zootaxa 5001, 1–83 (doi:10.11646/zootaxa.5001.1.1).
- Brown, W. L., Jr. 1978c. Contributions toward a reclassification of the Formicidae. Part VI. Ponerinae, tribe Ponerini, subtribe Odontomachiti. Section B. Genus Anochetus and bibliography. Studia Entomologica. 20:549-638. (page 612, see also)
- Camargo, K.S. de. 2011. Composicao e diversidade de "Poneromorfas" (Hymenoptera, Formicidae) em duas fitofisionomias de cerrado e padroes de distribuicao de "Poneromorfas", Pseudomyrmecinae e Cephalotini (Myrmicinae) para o Brasil. Thesis, Universidade de Brasilia.
- Franco, W., Ladino, N., Delabie, J.H.C., Dejean, A., Orivel, J., Fichaux, M., Groc, S., Leponce, M., Feitosa, R.M. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674, 509–543 (doi:10.11646/zootaxa.4674.5.2).
- Gibson, J. C., F. J. Larabee, A. Touchard, J. Orivel, and A. V. Suarez. 2018. Mandible strike kinematics of the trap-jaw ant genus Anochetus Mayr (Hymenoptera: Formicidae). Journal of Zoology. 306:119-128. doi:10.1111/jzo.12580
- Kempf, W. W. 1964f. The ants of the genus Anochetus (Stenomyrmex) in Brazil (Hym., Formicidae). Stud. Entomol. 7: 237-246 (page 239, figs. 4, 8, 12, 14 (gynecoid worker ))
- Mariano, C.S.F., Santos, I.S., Silva, J.G., Costa, M.A., Pompolo, S.G. 2015. Citogenética e evolução do cariótipo em formigas poneromorfas. In: Delabie, J.H.C., Feitosa, R.M., Serrao, J.E., Mariano, C.S.F., Majer, J.D. (eds) As formigas poneromorfas do Brasil, 1st edn. Ilhéus, Brasil, pp 102–125 (doi:10.7476/9788574554419.0010).
- Wheeler, G. C.; Wheeler, J. 1971b. Ant larvae of the subfamily Ponerinae: second supplement. Ann. Entomol. Soc. Am. 6 64: 1197-1217 (page 1212, larva described)
References based on Global Ant Biodiversity Informatics
- Fernandes I., and J. de Souza. 2018. Dataset of long-term monitoring of ground-dwelling ants (Hymenoptera: Formicidae) in the influence areas of a hydroelectric power plant on the Madeira River in the Amazon Basin. Biodiversity Data Journal 6: e24375.
- Fernández F. 2008. Subfamilia Ponerinae s.str. Pp. 123-218 in: Jiménez, E.; Fernández, F.; Arias, T.M.; Lozano-Zambrano F. H. (eds.) 2008. Sistemática, biogeografía y conservación de las hormigas cazadoras de Colombia. Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, xiv + 609 pp.
- Fichaux M., B. Bechade, J. Donald, A. Weyna, J. H. C. Delabie, J. Murienne, C. Baraloto, and J. Orivel. 2019. Habitats shape taxonomic and functional composition of Neotropical ant assemblages. Oecologia 189(2): 501-513.
- Franco W., N. Ladino, J. H. C. Delabie, A. Dejean, J. Orivel, M. Fichaux, S. Groc, M. Leponce, and R. M. Feitosa. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674(5): 509-543.
- Groc S., J. H. C. Delabie, F. Fernandez, M. Leponce, J. Orivel, R. Silvestre, Heraldo L. Vasconcelos, and A. Dejean. 2013. Leaf-litter ant communities (Hymenoptera: Formicidae) in a pristine Guianese rainforest: stable functional structure versus high species turnover. Myrmecological News 19: 43-51.
- Groc S., J. Orivel, A. Dejean, J. Martin, M. Etienne, B. Corbara, and J. H. C. Delabie. 2009. Baseline study of the leaf-litter ant fauna in a French Guianese forest. Insect Conservation and Diversity 2: 183-193.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Pires de Prado L., R. M. Feitosa, S. Pinzon Triana, J. A. Munoz Gutierrez, G. X. Rousseau, R. Alves Silva, G. M. Siqueira, C. L. Caldas dos Santos, F. Veras Silva, T. Sanches Ranzani da Silva, A. Casadei-Ferreira, R. Rosa da Silva, and J. Andrade-Silva. 2019. An overview of the ant fauna (Hymenoptera: Formicidae) of the state of Maranhao, Brazil. Pap. Avulsos Zool. 59: e20195938.
- Santos I. S., M. A. Costa, C. S. F. Mariano, J. H. C. Delabie, V. Andrade-Souza, and J. G. Silva. 2010. A Cytogenetic Approach to the Study of Neotropical Odontomachus and Anochetus Ants (Hymenoptera: Formicidae). Ann. Entomol. Soc. Am. 103(3): 424Ð429
- Scott-Santos, C.P., F.A. Esteves, C.R.F. Brandao. 2008. Catalogue of "Poneromorph" ant type specimens (Hymenoptera, Formicidae) deposited in the Museu de Zoologia da Universidade de Sao Paulo, Brazil. Papeis Avulsos de Zoologia 48(11):75-88.
- Sosa-Calvo J. 2007. Ants of the leaf litter of two plateaus in Eastern Suriname. In Alonso, L.E. and J.H. Mol (eds.). 2007. A rapid biological assessment of the Lely and Nassau plateaus, Suriname (with additional information on the Brownsberg Plateau). RAP Bulletin of Biological Assessment 43. Conservation International, Arlington, VA, USA.
- Vasconcelos, H.L., J.M.S. Vilhena, W.E. Magnusson and A.L.K.M. Albernaz. 2006. Long-term effects of forest fragmentation on Amazonian ant communities. Journal of Biogeography 33:1348-1356

